* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Slide 1
Condensed matter physics wikipedia , lookup
Chemical bond wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Isotopic labeling wikipedia , lookup
Chemical element wikipedia , lookup
Abundance of the chemical elements wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
Stoichiometry wikipedia , lookup
History of chemistry wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
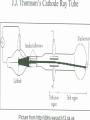
History of the Atomic Theory Law of Definite Proportions A given compound contains the same elements in exactly the same proportions by mass, regardless of the size of the sample or the source of the compound e.g. NaCl always contains 39.34% by mass of sodium and 60.66% by mass of Chlorine Law of Conservation of Mass The mass of the products of a reaction equals the mass of the reactants e.g. 32 mass units of sulfur and 32 mass units of oxygen react to form 64 mass units of sulfur dioxide Law of Multiple Proportions The mass ratio for one of the elements that combines with a fixed mass of another element can be expressed in small whole numbers e.g. 2 grams of hydrogen combine with 16 grams of oxygen to form water, but 2 grams of hydrogen combine with 32 grams of oxygen to form hydrogen peroxide 16:32 = 1:2 John Dalton http://www.slcc.edu/schools/hum_sci/physics/whatis/biography/dalton.html John Dalton’s Billiard Ball Model http://www.poolndarts.com/index.cfm/fuseaction/Catalog. Category/categoryID/77.cfm?STLCMPID=1798&engine=adwords!1994&keyword= %28billiard+ball%29&match_type= Dalton’s Atomic Theory 1. All matter is made of indivisible and indestructible atoms. 2. All atoms of a given element are identical in their physical and chemical properties. 3. Atoms of different elements differ in their physical and chemical properties. 4. Atoms of different elements combine in simple whole-number ratios to form compounds. 5. Chemical reactions consist of the combination, separation, or rearrangement of atoms. J. J. Thomson http://www.aip.org/history/electron/jjhome.htm Plum Pudding model http://www.nationmaster.com/encyclopedia/Plum-pudding-model Ernest Rutherford http://www.nationmaster.com/encyclopedia/Ernest-Rutherford Rutherford’s Gold Foil Experiment Niels Bohr http://nobelprize.org/physics/laureates/1922/ James Chadwick http://www.light-science.com/chadwick.html Model of the Atom http://www.nationmaster.com/encyclopedia/Image:Atom-diagram.png Louis deBroglie and Erwin Schrodinger http://www.britannica.com/nobel/micro/87_61.html http://www.britannica.com/nobel/micro/531_80.html