* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download LabNAAT
Survey
Document related concepts
Hepatitis C wikipedia , lookup
Hepatitis B wikipedia , lookup
Middle East respiratory syndrome wikipedia , lookup
Sexually transmitted infection wikipedia , lookup
Epidemiology of HIV/AIDS wikipedia , lookup
Microbicides for sexually transmitted diseases wikipedia , lookup
Transcript
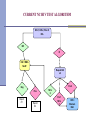
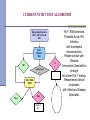
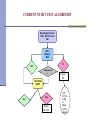
Lab Experience with HIV RNA NAAT Myra Brinson, MT(ASCP) Manager, Virology/Serology North Carolina State Laboratory of Public Health Ph: 919-807-8835 E-mail: [email protected] Discussion Topics History of HIV RNA NAAT at NC State Laboratory GenProbe APTIMA HIV Method Verification NC State Lab HIV Test Algorithm Evolution of NCSLPH HIV NAAT: 2001 to 2008 Utilization of different assays for the detection of HIV-1 RNA Different pooling algorithms Different pooling mechanisms Consistently demonstrated the ability to detect acute HIV-1 infections Pilot Study 2001 - Design All consecutive routine HIV tests submitted to the NC State Laboratory of Public Health over 4 weeks from 110 publicly funded counseling and testing sites (CTS) [n=8505] Initial Ab testing - OT Vironostika HIV-1 Viral Lysate Microelisa (State Lab) Manual pooling of Ab NR samples (State Lab) Roche Amplicor HIV-1 Monitor (UNC) – Standard and US Figure 1: Schema for pooling Ab-negative specimens Individual Specimens Intermediate Pools Master Pool (1:10) (1:90) Pilot Study 2001 - Results Acute infection: 5 per 10,000 Figure 2 : Disposition of Specimens Chronic infection: 44 per 152 with Data Incomplete or Questionable 10,000 Overall specificity: 99.99% 38 EIA Repeat Reactive Sera 8298 EIA negative & 5 EIA Unconfirmed Pos. 1 WB Negative v Estimated total testing costs/additional case diagnosed: $4109 299 Specimens Insufficient Vol. 8005 Ab Neg. Specimens Pooled Pilcher CD et al, JAMA, Vol. 288/No. 2, July 10, 2002 2 HIV+, Status Unknown 8341 Persons at Risk Estimated additional cost per specimen: $2.01 12 Previously Tested HIV+ 8505 Total Specimens 37 Newly Diagnosed WB+ Chronic Infections 1 False + RNA Test 5 RNA Positive 4 True Positive Acute Infections Screening and Tracing Active Transmission (STAT) Program – Year 1 11/01/2002 to 10/31/2003 - All consecutive routine HIV tests submitted to the State Laboratory from 110 publicly funded counseling and testing sites (CTS) [n=110,890] Initial Ab testing - bioMerieux Vironostika HIV-1 Viral Lysate Microelisa Initial manual pooling NAAT testing – bioMerieux NucliSens HIV-1 Qualitative assay Automated pooling added-Beckman Coulter bioMek FX STAT – Pooling Design Figure 1: Schema for pooling Ab-negative specimens Individual Specimens Intermediate Pools Master Pool 8-12 sera 96 sera STAT Results – Year 1 110,890 requested VCT Testing Population 1,427 Insufficient sample 213 Previously HIV + 109,250 successfully tested Population at risk 583 EIA + WB+ Long term HIV infected 107 Less sensitive EIA – by STARHS Likely recent infection 108,667 EIA -, WB – or indeterminate RNA tested 108,642 negative RNA screen HIV negative 25 individual specimens RNA positive 2 individuals EIA – RNA – through wk 12 False RNA positive; HIV negative 23 individuals confirmed HIV positive Acutely HIV infected Pilcher CD et al, New England Journal of Medicine, May 5 2005;352(18):1873-1883. Screening and Tracing Active Transmission (STAT) Program – Year 2 Nov 03 to March 05 - All consecutive routine HIV tests submitted to the State Laboratory from 110 publicly funded counseling and testing sites (CTS) [n=118,656] Initial Ab testing - bioMerieux Vironostika HIV-1 Viral Lysate Microelisa NAAT testing – GenProbe Procleix HIV-1 Assay Automated pooling – Hamilton AT Plus Detected 17 acute HIV cases (1 False Positive) STAT Overall Results - 2 years 224,108 EIA negative sera pooled and tested for HIV-1 RNA 40 True Positive Acute Infections; 3 False Positive RNA tests Acute infection: 1.8 per 10,000 Chronic infection: 65.9 per 10,000 Overall specificity: 99.8% At least 4% of the HIV-1 infected patients would have been undetected without the use of NAATs Manual vs. Automated Pooling Manual pooling very labor intensive – 30 to 45 minutes/90 samples Must be particularly diligent in pipetting technique to avoid cross contamination of samples 3 of 4 false positives occurred during the period of manual pooling Automated pooling instrumentation, maintenance, and pipet tips can be expensive Much more efficient – 10-15 minutes/90 samples Improved control of process – decreased human error Positive specimen identification - barcodes STAT Project Continues GenProbe Procleix HIV-1 Assay withdrawn from market due to patent dispute with Chiron March 05 – Nov 07 bioMerieux Nuclisens Easy Q HIV-1 NAAT EasyMag Automated Extraction/ Real-Time assay Initial Ab testing - bioMerieux Vironostika HIV-1 Viral Lysate Microelisa Automated pooling - Hamilton AT Plus Continued to detect acute HIV cases, although assay sensitivity was somewhat of concern 2008 – Changes to HIV Testing CATALYSTS bioMerieux Vironostika HIV-1 Viral Lysate Microelisa discontinued Contract for HIV NAAT out for bid – opportunity to bring on a more sensitive assay New LIMS Increase in HIV Antibody Screens 2001-2007 180,000 160,000 140,000 # of Tests 120,000 100,000 80,000 180,000 60,000 134,241 91,668 99,551 112,716 146,116 112,904 40,000 20,000 0 2001 2002 2003 Year 2004 2005 2006 2007 New HIV Antibody Assay Bio-Rad Genetic Systems HIV-1/HIV-2 Plus O EIA Automation - 3 Evolis instruments More sensitive assay – detects both IgM and IgG Ability to detect HIV-2 and Group O New NAAT Assay and Pooling Algorithm GenProbe APTIMA HIV- 1 NAAT RNA assay Hamilton STARlet robotic pipetting instrument Reduced pool size (80 samples/pool) Increased sensitivity for HIV-1 NAAT New Pooling Strategy 80 HIV-1/HIV-2 Plus O Nonreactive Samples 1 B Pool (containing 120 µl x 10 A Pools) = 1200 µl 10 A Pools (containing 20 µl x 8 samples) = 160 µl X X X X X X X X X X X X X X X X NAAT Questions??? Cost of NAAT: $37-$50/test, based upon 4,000/year test volume Two dedicated staff for pooling and NAAT testing – pool daily and test 2-3 times/week Currently pool all EIA negative samples vs. separating into low and high risk groups NAAT has also been useful for resolution of EIA reactive/WB negative or indeterminate samples Increased TAT – 2-3 days to 2-3 weeks GenProbe APTIMA HIV-1 NAAT Method Verification Off-label use of an FDAapproved assay * Plasma vs. serum * Waterbaths vs.SB100s CAP Molecular Verification Checklist *Accuracy *Precision *Reportable Range *Limit of Detection *Analytical Sensitivity *Analytical Specificity *Specificity/Cross Reactivity Accuracy 18 Nonreactive and 7 Reactive B Pools previously tested by current method (bioMerieux Nuclisens EasyQ) 17 of 18 known NR samples tested NR by APTIMA 7 of 7 known R samples tested R by APTIMA One discrepant sample QNS to resolve 96% (24/25) Precision-Reproducibility Tested several replicates of pooled NR sera and a R sera (170 copies /ml) over multiple days 170 copies/ml- prepared by diluting a sample of known copy number from a purchased linearity panel (HIV RNA Linearity Panel, PRD801, BBI Diagnostics) in pooled NR sera NR Intra-Assay 49% CV; Inter-Assay 51% CV R Intra-Assay 4% CV; Inter-assay R: 8% CV Reportable Range 2,900,000 (2.9 x 106) copy/ml sample from the purchased linearity panel Serially diluted in pooled NR sera to yield approx. log concentrations of 1 x 106, 105, 104, 103, 102, and 10 copies/ml Observed typical reaction curve for molecular assays Precipitous drop-off in signal strength from 100 to 10 copies/ml Limit of Detection Serially diluted the 170 copies/ml sample from the purchased linearity panel in pooled NR sera to yield samples of approx. concentration of 85, 43, 21, 11, 5, and 3 copies/ml Tested five replicates of the seven dilutions 100% at ≥43 copies/ml HIV-1 RNA 80% at 5 to 21 copies/ml HIV-1 RNA Analytical Sensitivity/Specificity Calculated by using accuracy sample results Analytical Sensitivity: 100% 7 of 7 known R samples tested R by APTIMA Analytical Specificity: 96% 17 of 18 known NR samples tested NR by APTIMA Specificity/Cross Reactivity Spiked pooled NR sera with other blood-borne viral pathogens that might be present in tested patient population: HSV-1, HSV-2, CMV, HAV, HBV, and HCV Required the purchase of HBV viral isolate from ATCC; other viruses obtained locally Virus concentrations tested were similar to average concentrations of HIV virus expected to be present in patient serum samples Samples were run twice, on consecutive days No cross-reactivity or interference with the six bloodborne viral pathogens Evaluation Questions??? Duration: Verification required approx. six weeks to complete Cost: Test kits provided by GenProbe BBI Linearity Panel - $1,380 ATCC HBV - $188 Availability of NR and R comparison samples - in our study, we were able to use master pool samples run previously by existing assay - may have to purchase additional panels CURRENT NC HIV TEST ALGORITHM HIV-1, HIV-2, Plus O EIA NR R HIV-1 RNA NAAT Repeat EIA x2 Rx2 Neg Pos Report #1 HIV-1 (Groups M & O) and HIV-2 antibodies were not detected. HIV1 RNA not detected. NR x 2 Rx1 NR x 1 HIV-1 Western Blot CURRENT NC HIV TEST ALGORITHM HIV-1, HIV-2, Plus O EIA NR R HIV-1 RNA NAAT Repeat EIA x2 Rx2 Neg Pos Report #2 NR x 2 Rx1 NR x 1 HIV-1 Western Blot HIV-1 RNA detected. Possible Acute HIV Infection. Please consult with Disease Intervention Specialist to arrange for further HIV-1 testing. Recommend clinical evaluation with Infectious Disease Specialist. CURRENT NC HIV TEST ALGORITHM HIV-1, HIV-2, Plus O EIA NR R HIV-1 RNA NAAT Repeat EIA x2 Rx2 Neg Pos Report #1 Report #2 NR x 2 Rx1 NR x 1 HIV-1 Western Blot CURRENT NC HIV TEST ALGORITHM HIV-1, HIV-2, Plus O EIA NR Red, Green, or Blue Dot Samples* R HIV-1 RNA NAAT Repeat EIA x2 *For individually tested samples, repeat any Pos result: If retest is Pos = Report Pos If retest is Neg, repeat again: 2nd retest Neg = Report Neg 2nd retest Pos = Report Pos Rx2 Neg Pos* Report #1 Report #2 NR x 2 Rx1 NR x 1 HIV-1 Western Blot CURRENT NC HIV TEST ALGORITHM Repeatedly Reactive HIV-1, HIV-2, Plus O EIA HIV-1 Western Blot R NR Indeterminat e HIV-1 RNA NAAT Neg Pos Report #3 HIV-1 (Groups M & O) Antibody was detected. Please consult with Disease Intervention Specialist to determine if further testing is warranted to rule out possible co-infection with HIV-2, based upon epidemiological information. CURRENT NC HIV TEST ALGORITHM Repeatedly Reactive HIV-1, HIV-2, Plus O EIA HIV-1 Western Blot R NR Indeterminat e Report #3 HIV-1 RNA NAAT Neg Pos CDC for HIV2 WB or Rapid, by DIS request HIV-1 (Groups M & O) Antibody was detected. Please consult with Disease Intervention Specialist to determine if further testing is warranted to rule out possible co-infection with HIV-2, based upon epidemiological information. CURRENT NC HIV TEST ALGORITHM Repeatedly Reactive HIV-1, HIV-2, Plus O EIA HIV-1 Western Blot R NR Indeterminat e HIV-1 RNA NAAT Neg Pos Report #4 HIV-1 RNA detected. Possible Acute HIV Infection with incomplete seroconversion. Please consult with Disease Intervention Specialist to arrange for further HIV-1 testing. Recommend clinical evaluation with Infectious Disease Specialist. CURRENT NC HIV TEST ALGORITHM Repeatedly Reactive HIV-1, HIV-2, Plus O EIA HIV-1 Western Blot R NR Indeterminat e HIV-1 RNA NAAT Neg Pos Report #5 HIV-1 RNA detected. Probable Acute HIV Infection with incomplete seroconversion. Please consult with Disease Intervention Specialist to arrange for further HIV-1 testing. Recommend clinical evaluation with Infectious Disease Specialist. CURRENT NC HIV TEST ALGORITHM Repeatedly Reactive HIV-1, HIV-2, Plus O EIA HIV-1 Western Blot R NR Indeterminat e *For individually tested samples, repeat any Pos result: If retest is Pos = Report Pos If retest is Neg, repeat again: 2nd retest Neg = Report Neg 2nd retest Pos = Report Pos HIV-1 RNA NAAT Neg Pos* Report #4 or #5 CURRENT NC HIV TEST ALGORITHM Repeatedly Reactive HIV-1, HIV-2, Plus O EIA HIV-1 Western Blot R NR Indeterminat e Report #3 HIV-1 RNA NAAT Neg Pos Report #4 or #5 CDC for HIV2 WB or Rapid, by DIS request CURRENT NC HIV TEST ALGORITHM Repeatedly Reactive HIV-1, HIV-2, Plus O EIA HIV-1 Western Blot NR or Ind HIV-1 RNA NAAT Neg HIV-1/HIV-2 Rapid NR Report #6 or #7 R HIV-2 HIV-1/HIV-2 infection status is Inconclusive. Please submit another sample for further HIV-1/HIV-2 testing. CURRENT NC HIV TEST ALGORITHM Repeatedly Reactive HIV-1, HIV-2, Plus O EIA HIV-1 Western Blot NR or Ind HIV-1 RNA NAAT Neg HIV-1/HIV-2 Rapid NR R HIV-2 HIV-1 (Groups M or O) was not detected. HIV-2 infection status is inconclusive. Specimen referred to CDC for further HIV-2 testing. CDC for HIV-2 WB Confirmation Report #8 or #9 Questions???