* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
International investment agreement wikipedia , lookup
Corporate venture capital wikipedia , lookup
Socially responsible investing wikipedia , lookup
Early history of private equity wikipedia , lookup
Investment banking wikipedia , lookup
Environmental, social and corporate governance wikipedia , lookup
History of investment banking in the United States wikipedia , lookup
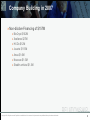
KPCB Pandemic and Biodefense Fund »Thomas P Monath MD Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. PPB Fund Mission & Goals Invest in innovations to address gaps in global pandemic preparedness » Rapid “point of care” diagnostics » Vaccine science and production » Therapeutic drug discovery and development » Platform and broad spectrum technologies Investment Criteria & Goals » Back technology platforms applicable across multiple diseases » Build sustainable companies in sizable markets » Invest in companies with suites of products (no single customer) » Catalyze innovation in treating & managing emerging infectious diseases beyond influenza » Double-bottom line thesis Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. 2 The Journey 2H05 2006 2007 2008 »SAG Summit »Boil the ocean »Identify preparedness gaps »Build the network »Develop strategic map »Raise fund »Build KPCB team »Screen >100 Companies »Invest based on strategic map »Passed BARDA »Screen 100 more companies »Build companies & management »Secured nondilutive funding »Expand & refocus strategy »Build companies & management »More non-dilutive funding »Screen fewer companies »Invest to fill gaps Anza »Hire CEOs: HX, BRCX, Xcellerex »China Strategy Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. »Hire CEOs: 3-V, Juvaris 3 Portfolio Goals September 2006 Category Innovations Needed (Sept ’06) Co’s Evaluated (Sept ’06) Potential Investment or MTI Forecast Portfolio Mix (Sept ’06) Vaccines, Adjuvants & Enabling Technology -Cell production -Adjuvant -Broad spectrum -Platforms 40 - Xcellerex - Novavax - Juvaris 5 Antivirals, therapeutics -Better drugs (efficacy & broad spectrum) -New Mechanisms -Cytokine storm 30 - BioCryst - Stealth 3 Diagnostics -Rapid diagnostic -Molecular Dx 30 - HX Dx 2 Antibodies -Combination therapies -Conserved antigens -Polyclonal 10 - Spaltudaq 1 Surveillance, Masks, Ventilators -Early detection -Ventilators -Masks 10 -Voxiva 1 Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. 4 Learning & Strategic Expansion » Platforms and broad-spectrum technologies favored » Biodefense » Not primary focus of investment » Important secondary programs, stabilization, POC, no-dilutive funding » Surveillance » No investment case » Antibody technologies– none found » Monoclonal escape variants » Flu genetic plasticity » Uncertain monetization, ROI » Cytokine storm– none found » Learning ignited expansion of strategy to other technology gaps in emerging infectious disease with large markets » Hospital Acquired Infections » Chronic infectious agents Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. 5 Major Gaps in Flu (and Biodefense) --What should we invest in? BROAD-SPECTRUM COUNTERMEASURES » Vaccine platform technologies, plug-and-play » Multivalent and “universal” vaccines » Broad-spectrum and novel-class antibacterial drugs » Broad-spectrum and novel-class antiviral drugs » Human polyclonal antibody technologies (multivalent) » Immune modulation for broad protection and treatment » Immune agonists (innate immunity, adjuvants) » Immune antagonists (cytokine storm, inflammation, oxidative stress ) »Improved delivery systems » Oral, skin, self-administered » Flexible, affordable and faster manufacturing systems » Vaccines, antibodies » POC diagnostics and surveillance tools » Patient care (ventilators) Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. 6 Investment Progress & Expanded Portfolio Co’s Evaluated to Date Investment or MTI Expected Portfolio Sept ’06 Portfolio Mix Dec ’07 Category Innovations Needed Vaccines, Adjuvants, and Enabling Technologies • Recombinant production • Adjuvants • Broad spectrum • High potency • Hospital-acquired infections • Therapeutic vaccines • Other high-value vaccines 75+ • Xcellerex • Novavax • Juvaris • Anza • Novadigm 5 5 Antivirals, Antibiotics • Better drugs (resistance, efficacy & broad spectrum) • New Mechanisms 40+ • BioCryst • Stealth 3 2 Diagnostics • Rapid diagnostic • Molecular Dx 35+ • HX Dx 2 1 Other Preparedness Gaps • Ventilator innovation • Antimicrobial Masks • Enterprise Preparedness 35+ • Breathe • SafeLife • Preparis 1-2 1-2 Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. 7 Indications and platform breadth Category Investment Vaccines, Adjuvants, and Enabling Technologies • Xcellerex • Novavax • Juvaris • Anza • Novadigm Antivirals, Antibiotics • BioCryst • Stealth •Trius Diagnostics • HX Dx Other Preparedness Gaps • Breathe • SafeLife • Preparis Indications Biomanufacturing VLPs (Flu-RSV-Zoster) Adjuvant, immunotherapy, biodefense Bacterial vector, HCV, Biodefense MRSA, Candida Peramivir Host targets antivirals Novel broad antibiotic, MRSA POC Diagnostics Miniaturized, affordable respirator Improved masks Enterprise preparedness Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. 8 Company Building in 2007 » Non-dilutive Financing of $157M » BioCryst $102M » Xcellerex $27M » HX Dx $12M » Juvaris $11.5M » Anza $1.6M » Novavax $1.5M » Stealth antiviral $1.3M Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. 9 What We Have Learned »Vaccines – Proof in seasonal flu necessary; platform technologies key; “holy grail” of universal flu vaccine--still too early to tell »Adjuvants – Important for supply & efficacy »Diagnostics – Point of care is key; patent landscape messy »Antiviral therapeutics – Few novel opportunities, so incubated our own »Antibiotics- Major need for improved antibiotics, antimicrobial resistance increasing »Manufacturing – Commitment to global/local supply »Surveillance – Non-profit and government activity »Biodefense – limited investment case, secondary programs Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. 10 Our Methods »Theme days » Antibody Day, Diagnostics Day, Adeno Day » Side-by-side comparison with technical advisors » Built network (Scott Layne SAB HX) »Build a great team » Weekly meetings & formal updates »VC Partnering, leverage the investment » $200M$500M » BioCryst - Baker Investments, TPG » Stealth (antiviral) – The Column Group » Xcellerex – Vantage Point » Anza – Versant & Sofinnova » Novadigm - Domain Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. 11 Future Steps »Build portfolio companies and launch products » Bringing meaningful therapies to patients – top priority » Leveraging product platforms across new indications » Facilitate networking across the portfolio (i.e. China, Xcellerex-Novavax) » Optimizing non-dilutive financing (HHS, BARDA, NGO) » Continue to build management teams and drive clinical success »Balancing the portfolio » Invest later stage – near-term revenue opportunities (within 12 months) » Opportunistic investment in technologies to address remaining gaps » Expand disease focus to include other emerging infectious disease Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. 12 Funding NEIDL » Direct funding from grants and contracts » Partnering with industry » Key to NIAID translational research and BARDA advanced development contracts » NEIDL offers unique facilities and core competencies » Difficult for industry to find USG collaborators, competition for rare resources » Direct funding will depend on commercial applications » Biotech industry dependent on private financing » Costs of product development are very high » Mitigate risk from non-dilutive funding sources very attractive to VCs »Biodefense as an area for investment » Problems: Bubble revenues, lower return on investment, single customer » Platforms, broad spectrum and commercial indications are key » Can be a stabilizing factor, contribute to overhead costs » Can help to build core competencies » Can obtain POC for commercial technologies Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. 13 Key factors attractive to investors » » » » » » » » Commercial opportunity, market size Novel, disruptive, not fast follower Clear differentiation from competition Strong IP Opportunity for non-dilutive funding to mitigate risk Strong preclinical surrogates for efficacy Great team Clear product development plans and value creation Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. 14 NEIDL Opportunity Product development » Internal R&D, discovery, new proprietary technology » Selective in licensing » Collaborations, partnerships with industry Services »Serological testing, reagents »Analytical development »Animal models, unique pathophysiological measurements »Preclinical development »NEIDL offers unique facilities and core competencies Copyright 2006, All rights reserved. Duplication or redistribution of the contents of this presentation are prohibited without prior written authorization. 15