* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Lesson 4 Evaluation of Measurements
Location arithmetic wikipedia , lookup
Bra–ket notation wikipedia , lookup
Abuse of notation wikipedia , lookup
History of mathematical notation wikipedia , lookup
Positional notation wikipedia , lookup
Big O notation wikipedia , lookup
Musical notation wikipedia , lookup
Large numbers wikipedia , lookup
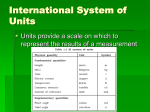
Homework Read Pgs. 11-13 Chapter 1 Problems 20, 24, 26,30 Worksheet Scientific Measurement (2A Extra Practice Problems) Significant figures Addition and Subtraction The answer must have the same number of decimal places as the factor with the least number of decimal places. Significant Figures Example of Addition: 1250 + 23.98 1273.98 (this is what a calculator will show) 1274 (this is the answer corrected for significant figures, rounded to the 1’s place) Significant figures Multiplication and division The answer must have the same number of significant figures as the factor with the least number of significant figures. Significant figures Example of Division: 15.375 / 5.0 = 3.075 (this is what a calculator will show) Since the denominator only has 2 significant figures the answer is rounded to 3.1 Significant Figures 8.654 m x 0.34 m = 2.10 cm x 0.50 cm = 10.4815 ml ÷ 8.4 ml = 0.365 m ÷ 0.050 m = textbook HW 20a. 3 b. 4 c. 4 d. 1 e. 5 24a. 0.5 b. 401.4 c. 0.2684 d. 7.8 26a. 132.5 g b. 298.69 cm c. 13 lb d. 350 oz 30. only c is exact Scientific Measurement 1 a. 3 2 a. 133 g e. 50.8 dm b. 4 b. 109 mL f. 2.86x103 cal c. 7 c. 13 cm d. 2 d. 14 g/mL e. 3 f. 6 3 a. 1.0 e. 740. 4a. 145g e. 1.30x102dm b. 40.1 f. 80 c. 6.2x10-5 d. 1.5 b. 64mL f. 16 cm c. 91.7 cm2 g. 6000cal d. 4.3g/cm3 Unit 1 Read pages 8-9 Unit 1 Sci. Notation and % Error Chapter 1 Problems 20, 24, 26,30 Quiz: Oct 18/19 (Thursday / Friday) Numbers and Measurement Chemistry requires us to make accurate measurements that are often very small or very large. To more easily handle these very large and small numbers, we use scientific notation. Scientific Notation Measurements are written as the product of two numbers A coefficient – number between 1 and 10 10 raised to a power – the exponent indicates the number of times the coefficient must be multiplied or divided by 10. Scientific Notation Write the following in scientific notation: 6,954,000 175.983 Write the following in standard numerical form: 6.75 x 10-3 1.865 x 102 Calculations in Scientific Notation Multiplication Multiply coefficients Add exponents Perform the following calculations: 7.2 x 102 · 5.02 x 10-3 1.0 x 102 · 2.6 x 108 Calculations in Scientific Notation Division Divide coefficients Subtract the exponent of the denominator from the exponent of the numerator. Perform the following calculations: 8.4 x 103 / 2.1 x 10-2 7.25 x 104 / 5.0 x 102 Calculations in Scientific Notation Addition and subtraction Make the exponent of both numbers the same Align decimal points, and add coefficients The exponent of the result will be the same as for the measurements Calculations in Scientific Notation Perform the following calculations: 1. 6.3 x 104 + 2.1 x 10-3 2. 7.563 x 102 - 1.77789 x 10-3 Calculations in Scientific Notation 1. 1.5 x 106 + 2.7 x 103 2. 6.38 x 10-3 – 3.8 x 10-4 International System of Units Units of Measurement Quantity SI base unit or derived unit Symbol Length meter m Volume cubic meter m3 Mass kilogram kg Density grams per cubic centimeter g/cm3 Temperature Kelvin K Time second s Pressure Pascal Pa Energy joule J Amount of Substance mole mol Luminous Intensity candela cd Electric Current ampere a SI Units 103 for kilo106 for mega109 for giga10-1 for deci10-2 for centi10-3 for milli10-6 for micro10-9 for nano10-12 for pico- k M G d c m μ n p Accuracy: Evaluations of Measurements Accepted value: True or correct value based on reliable source Experimental value: measured by you during the experiment Error Difference between the accepted value and the experimental value Take the absolute value % Error % Error = Error / accepted value * 100 % Error = Actual – Experimental x 100 Actual What is the % error? A student measures a volume as 25.0mL, whereas the correct volume is 23.2mL. Density Which is heavier – a pound of popcorn or a pound of cheese? They would have the same mass!! However, if you had equal VOLUMES of popcorn and cheese, the cheese would have more mass. Density A cube of gold-colored metal with a volume of 64 cm3 has a mass of 980. grams. The density of pure gold is 19.3 g/cm3. Is the metal pure gold? Unit 1 HW: Read pages 9-10 Unit 1: Density and Temperature More Conversion Problems Temperature The temperature of an object determines the flow of heat transfer. Celsius scale uses the freezing point of water as 0 and the boiling point of water as 100. Kelvin scale uses 273 for the freezing point of water, and 373 as the boiling point. (Gabriel) Fahrenheit Scale German physicist Developed scale in 1714 Hg was used in thermometer Freeze pt. water = 32˚F Boiling pt. water = 212˚F 0˚F = freeze pt. of water, salt and dry ice (CO2) Why use Hg in a thermometer? For every degree the mercury’s temp. increases, the difference in which the Hg expands is NOTICABLE and CONSTANT. Alcohol thermometer used in Alaska because Hg will freeze! (Anders) Celsius Scale Developed in 1742 Swedish Astronomer Freezing pt. of water = 0˚C Boiling pt. of water = 100˚C Uses Hg in thermometer 9 F C 32 5 (Lord William) Kelvin Scale Developed in 1848 Based on absolute zero = zero heat energy = all motion stops 0 Kelvin = -273.15˚C No degree on scale (Kelvin units) Kelvin is a theoretical scale because it does not compare the temp. to FP or BP of water. Based on lowest temperature possible – No negative numbers on scale Compare the 3 temperature scales