* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Numerical methods in science
Survey
Document related concepts
Transcript
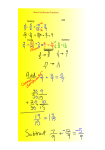
Numerical Methods in Science --How many scientists does it take to change a light bulb? --Scientists don’t change light bulbs, that’s what engineers are for. Rounding • Choose where (at which digit) you want to round. • If the NEXT digit is 5 or more, round up; otherwise round down • Rounding does not change the size of the number, just its precision. Examples • 27,454,352 • 7432 • Round to the nearest million • Round to the nearest ten • .00088536 • .0653 • Round to the nearest 100,000th • Round to the nearest 1000th Examples • 27,454,352 • 7432 • Round to the nearest million • Round to the nearest ten • .00088536 • .0653 • Round to the nearest 100,000th • Round to the nearest 1000th Examples • 27,454,352 • 7432 • Check • Check • .00088536 • .0653 • Check • Check Examples • 27,454,352 • Check • 7432 Round down • .00088536 • Check • Check • .0653 Round up • Check Round down Round down Examples • 27,454,352 =27,000,000 • (fill in 0’s to keep the same size) • 7432 • .00088536 =.00089 • (change the 8 to 9, do not fill in 0’s after a decimal!) • .0653 =7430 • (fill in 0 to keep the same size) =.065 • (do not fill in 0’s after a decimal!) Round to the nearest: 1) 2) 3) 4) 5) 6) 7) 1.22 .0004528 12,900,000 .00100 3,045,000,000 .00003 7 (tenth) (1000th) (million) (10000th) (million) (100th) (10) Significant figures • All non-zero digits are significant • Zeros – A) Leading, not significant. – B) Trapped (by SF)--significant – C) Trailing, with a decimal--significant Which digits are SF? 1) 2) 3) 4) 5) 6) 7) 1.22 .0004528 12,900,000 .00100 3,045,000,000 .00003 5.30 x 10 14 Which digits are SF? 1) 2) 3) 4) 5) 6) 7) 1.22 .0004528 12,900,000 .00100 3,045,000,000 .00003 5.30 x 10 14 Adding and subtracting 1.22 + .452 = Adding and subtracting 1.22 + .452 = 1.67 Your calculator says “1.672”, but you don’t know how many thousandths there are in the first number. Round where your knowledge ends Adding and subtracting 1) 2) 3) 4) 5) 1.22 - .047 1290 + 100 .00034 + .000038 5.30 - 2.30 153000 - 12 Adding and subtracting 1) 2) 3) 4) 5) 1.22 - .047 1290 + 100 .00034 + .000038 5.30 - 2.30 153000 - 12 = 1.17 = 1400 = .00038 = 3.00 = 153000 Multiplying and dividing Suppose there are 20,000 pairs of Nike Air Pegasus running shoes in the Denver area. Suppose each pair cost $53.47, like mine did. How much did those shoes cost? Multiplying and dividing Suppose there are 20,000 pairs of Nike Air Pegasus running shoes in the Denver area. Suppose each pair cost $53.47, like mine did. How much did those shoes cost? $1 million. Multiplying and dividing Suppose there are 20,000 pairs of Nike Air Pegasus running shoes in the Denver area. Suppose each pair cost $53.47, like mine did. How much did those shoes cost? $1 million. Not $1,069,400 Multiplying and dividing • Round to match the precision of the least number of SF in your problem. • The “20,000 pairs” is a round number, 1SF. Don’t use more than 1SF in your answer. Multiplying and dividing 1) 2) 3) 4) 5) 138422 x .047 1390 ÷ 150 .34 x .038 5.30 ÷ 23521 3x4 Multiplying and dividing 1) 2) 3) 4) 5) 138422 x .047 1390 ÷ 150 .34 x .038 5.30 ÷ 23521 3x4 = 6500 = 9.3 = .013 = .000225 = 10 A little bit of algebra • If Density = mass/volume (It does.) then: D=m/v , m=vD, and v=m/D A little bit of algebra • You will have to be able to solve for any variable in a formula. • The steps are: 1) Start with your original formula. D=m/v A little bit of algebra • You will have to be able to solve for any variable in a formula. • The steps are: 2) Multiply both sides by v (the denominator) vD=vm/v A little bit of algebra • You will have to be able to solve for any variable in a formula. • The steps are: 2) Multiply both sides by v (the denominator) vD=vm/v = m V cancels on the right A little bit of algebra • You will have to be able to solve for any variable in a formula. • The steps are: 3) Divide both sides by D m = vD D D A little bit of algebra • You will have to be able to solve for any variable in a formula. • The steps are: 3) Divide both sides by D m = vD =v D D D cancels on the right A little bit of algebra • So: D=m/v m=vD v=m/D In general: Solve by undoing • • • • • If something is added, subtract If something is subtracted, add If something is multiplied, divide If something is divided, multiply If something is raised to a power, take that root Practice, Practice, Practice! Conversions 1) Start with the measurement given. 2) Multiply it by a fraction called a conversion factor. It has three properties: --The units you start with go on the bottom (You want them to cancel) --The units you want go on the top (You want to end up with them next) --The numbers make the top and the bottom equal (So the fraction is equal to 1, it won't change the value of the measurement) 3) Cancel your units, multiply the numerators, and divide by the denominator 4) Repeat if necessary For example: • 74.32 mm = _______ m For example: • 74.32 mm = _______ m • 74.32 mm Start with the measurement given. For example: • 74.32 mm = _______ m • 74.32 mm x ____________ = Multiply it by a fraction called a conversion factor. For example: • 74.32 mm = _______ m • 74.32 mm x ____________ = mm --The units you start with go on the bottom (You want them to cancel) For example: • 74.32 mm = _______ m • 74.32 mm x ________m___ = mm --The units you want go on the top (You want to end up with them next) For example: • 74.32 mm = _______ m • 74.32 mm x __1 x 10-3 m___ = 1 mm --The numbers make the top and the bottom equal (So the fraction is equal to 1, it won't change the value of the measurement) For example: • 74.32 mm = _______ m • 74.32 mm x __1 x 10-3 m___=7.432x10-2m 1 mm (or .07432m) 3) Cancel your units, multiply the numerators, and divide by the denominator Convert 1) 1.26 cm = _____m 2) 5.28 m = ______ mm 3) .00084 km = _______ mm 4) 8.00 mm = _______nm Metric System prefixes • • • • • • • • • • Prefix giga mega kilo deka deci centi milli micro nano Symbol G M k dk d c m m n Meaning 109 (1 000 000 000) 106 (1 000 000) 103 (1 000) 101 (10) 10-1 (0.1) 10-2 (0.01) 10-3 (0.001) 10-6 (0.000 001) 10-9 (0.000 000 001) SI System • • • • • --the International system --used by scientists worldwide --more consistent than the English system --defines seven standard units --allows combinations for derived units • (it is no more precise or accurate than any other system) Measurement Unit Symbol • • • • • • • Length Mass Time electric current temperature amount of substance luminous intensity meter kilogram second ampere kelvin mole candela m kg s A K mol cd Commonly Used Derived Units • • • • • • Area Volume Velocity Acceleration Density Dynamic viscosity Commonly Used Derived Units • • • • • • Area =length x width (in m2) Volume =area x height (in m3) Velocity =length / time (in m/s) Acceleration =velocity / time (in m/s2 ) Density =mass / volume (in kg/m3) Dynamic viscosity (Just kidding, it’s not common) For a chemist • Mass: gram, kilogram, milligram • Length: centimeter, meter, millimeter, nanometer • Volume: milliliter, liter, cubic meter • Time: second, minute, hour Making measurements • Read the numbers • Count the marks • Estimate one final digit. 7 3 10 15 7 6 6 2 9 10 8 4 5 1 8 5 9 2 50 40 30 1 2 3 4 5 6 1 2 3 4 5 6 10 20 30 40 50 60 Scientific Notation • For any real number, A, there is some a and b, such that: • A= a x 10b • a is between 1 and 10 • b is a whole number Examples • 27,000,000 • 7430 • .00089 • .065 Examples • 27000000 = 2.7 x 10 7 • .00089 = 8.9 x 10 -4 • 7430 = 7.43 x 10 3 • .065 = 6.5 x 10 -2 Examples • 5.8 x 10 4 • 2.17 x 10 8 • 1.20 x 10 -4 • 5.05 x 10 -3 Examples • 5.8 x 10 4 =58000 • 1.20 x 10 -4 =.000120 • 2.17 x 10 8 = 21,700,000 • 5.05 x 10 -3 = .00505 Put into scientific notation 1) 2) 3) 4) 5) 6) 7) 1.22 .0004528 12,900,000 .00100 3,045,000,000 .00003 5 Take out of scientific notation 1) 2) 3) 4) 5) 6) 7) 1.82 x 10 -5 4.28 x 10 4 1.60 x 10 -6 1.030 x 10 7 7.045 x 10 -3 9 x 10 0 4 x 10 1 Graphing • A graph shows a picture of what a set of numbers represent. • The representation must be honest Pie Graphs • Used when the total of all of the numbers is some whole value—this is for all of my AP Chemistry students AP Chemistry Scores, Denver South High School 2004-2008 No recommendation 2 10 Possibly qualified 14 Qualified 14 Extremely well qualified Extremely well qualified Well Qualified Qualified 2 3 Possibly qualified 4 No recommendation 5 12 Well Qualified 1 Bar Graphs • Used when the categories don’t add up to any definite total A&P Grades, last names K-P Percent grade 120 100 80 60 40 20 0 1 2 3 4 5 6 7 Student number 8 9 10 Line Graphs • Used when both sets of data are numbers Mass of Lead iodide recovered 0.14 Mass, in grams 0.12 0.1 0.08 0.06 0.04 0.02 0 0 2 4 6 8 Milliliters of lead solution 10 12