* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download NO 2
Survey
Document related concepts
Transcript
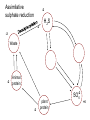
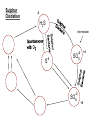
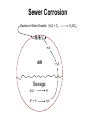
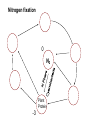
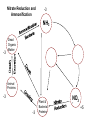
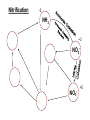
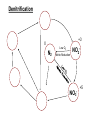
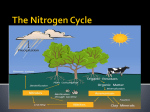
Biogeochemical Cycles • Objective to understand the principles and importance of biological systems in the cycling of key elements and nutrients • Reference Butcher S.S. et al Global Biogeochemical Cycles Carbon Cycling • The Major Carbon Reservoirs CO2 in Air CO2 in water Soil Humics Coal/Oil Carbonate mineral Marine Biota Terrestrial Biosphere 750 Pg 38,000 Pg 1500 Pg 10,000 Pg 107 Pg 3 Pg 560 Pg P = Peta = (1015) • The Major Carbon Fluxes Biological Assimilation Biological Dissimilation Fossil Fuel burning 170 Pg 170 Pg 5 Pg Sulphur Cycle Differences from N cycle More valences spontaneous reactions Important Oxidation States Form Example S2- sulphur proteins sulphur sulphite sulphate S0 SO32SO42- Oxidation State -2 0 +4 +6 Assimilative sulphate reduction -2 H2S -2 Waste -2 Animal protein -2 -2 plant protein SO4 +6 Assimilative Sulphate reduction Sulphate v. stable Incorporated into protein Many organisms in assimilative reduction SO4 R-SH Decay H2S Desulphurylation Dissimilatory Sulphur Reduction -2 H2S intermediate SO3 SO42- 2- +4 +6 Dissimilatory Sulphate Reduction Sulphate stable in high pE environments If – – – no oxygen energy source (eg, H2 or acetate) Sulphate reducing bacteria (SRB) then SO42- H2S SO4 electron acceptor Competes with methanogens Sulphate reduction and sulphide very important H2S is: – toxic to bacteria, algae, workers (ppm) – smelly – reactive with metals gives FeS – precursor for acid formation (when oxidised by Thiobacillus) – cause of steel and concrete corrosion H2S Equilibria and pH HS- + H+ H2S(aq) pKa = 7.1 S2- + 2 H+ pKa = 14 H2S also a gas Only H2S toxic/smelly, others dissolved pH >8 little H2S pH <7 mostly H2S Sulphur Oxidation -2 H2S intermediate +4 -2 S 0 SO 3 -2 SO4 +6 Sulphur oxidation H2S S , spontaneous in O2 – Not fast (only HS- reacts) H S and S electron donors 2 Variety of organisms – Sulphur Bacteria (Beggiatoa; Thiobaccillus, Thiothrix) require oxygen – Photosynthetic (Green and Purple sulphur bacteria) anaerobic Waste Stabilization Ponds (WSP) Sewer Corrosion Bacteria in Water Droplets (H2S + O2 H2S AIR H2S Sewage SO42- S2- S2- + H+ H2S H2SO4 ) Nitrogen cycle Less spontaneous reactions than S Form Species NH3 ammonia proteins N2 NO NO2NO3- Nitric oxide Nitrite Nitrate Oxidation State -3 0 +2 +3 +5 Nitrogen fixation 0 N2 Plant Protein -3 Nitrogen fixation Energy intensive Reduction of nitrogen Major path biological – – bacteria free-living (Azospirillum) and symbiont (Rhizobium) Blue green algae Also lightening and feritilizers Nitrate Reduction and Ammonification -3 NH3 -3 Dead Organic Matter Animal Proteins -3 -3 Plant & Bacterial Proteins NO3 - +5 Organic Nitrogen Transformations Assimilative Nitrate reduction N incorporated into organic nitrogen. minor pathway Bacteria, fungi, algae Ammonification Organic nitrogen into ammonium ammonium taken up by plants Organic nitrogen measured as Kjeldahl-N (TKN) Organic N is digested by H2SO4 to release N as ammonia. Ammonia then measured (distillation) Nitrification -3 NH3 +3 NO2- NO3 - +5 Nitrification Two Stage Process – 1 Ammonia oxidation (Nitrosomonas and Nitrosospira) 2NH3 + 3O2 2NO2- + 2H+ + 2H2O – 2 Nitrite oxidation (Nitrobacter) 2NO2- + O2 2NO3- Measurement of ammonia – – Nesslers method Use Nesslers reagent to get a brown colour Borate method NH3 + H3BO3 NH4+ + H2BO3H2BO3- + H+ H3BO3 Denitrification +3 0 Low O2 N2 Nitrite Reduction NO2- NO3 - +5 Denitrification Dissimilatory Reduction of NO3- and NO2- to N2 – Variety of bacteria – Occurs in absence of oxygen – Small amounts of NO and N2O also formed Nitrate and Nitrite measured – ion chromatography (Dionex) - best Nitrite – Indirect Colorimetric method - tedious Nitrate – UV absorbtion - interfering species – Additional reduction methods – ion specific electrodes