* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Tetracycline Antibiotics
Survey
Document related concepts
Peptide synthesis wikipedia , lookup
Butyric acid wikipedia , lookup
Epitranscriptome wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Point mutation wikipedia , lookup
Genetic code wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Protein structure prediction wikipedia , lookup
Proteolysis wikipedia , lookup
Metalloprotein wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Transcript
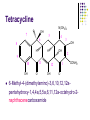
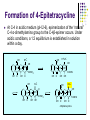
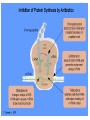
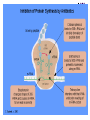
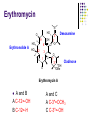
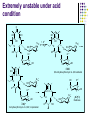
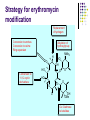
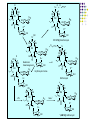
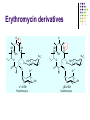
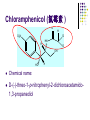
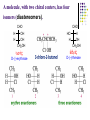
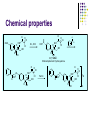
Chapter 8 Antibiotics Section 2. Tetracyclines Section 3. Aminoglycoside Section 4. Macrolides Section 5. Chloramphenicol Antibiotics as disturber with the biosynthesis of protein These antibiotics all target the bacterial ribosome and interfere in the process of translation of the messenger RNA into protein and thus block a fundamental process in bacterial metabolism. Inhibitors of 30s Ribosomal subunit: Aminoglycosides and Tetracyclines Inhibitors of the 50s Ribosomal subunit: Macrolides and Chloramphenicol Tetracycline Antibiotics Tetracyclines are produced by actinomyces (放线菌), which have broad-antibacterial spectrum. The basic skeleton of tetracyclines is naphthacene ring. Tetracyclines differing from each other chemically only by substituent variation at positions 5,6 and 7. R4 H N O 2 HO R2 H R1 R3 O OH H H O OH H R4 R3 R2 R1 8 7 9 D 10 N 6 CH 11 OH O OH 5 BH 12 OH N 4 OH A 1 OH 3 2 CONH2 O ÍÁùËØ£¨ Oxytetracycline£© R1 = -OH R2 = -OH R3 = -CH3 R4 = -H ½ðùËØ£¨ Chlotetracycline£© R1 = -H R2 = -OH R3 = -CH3 R4 = -Cl ËÄ»· ËØ£¨ Tetracycline£© R1 = -H R2 = -OH R3 = -CH3 R4 = -H Tetracycline pharmacophore and numbering Positions at the “bottom” of the molecule (10, 11, 1) and most of ring A (positions 2, 3, and 4) represent the invariant pharmacophore region of the molecule, where modifications are not tolerated without loss of antibiotic activity. Mechanism of Action: Tetracyclines inhibit bacterial protein synthesis by blocking the attachment of the t-RNA-amino acid to the ribosome. Tetracyclines can also inhibit protein synthesis in the host, but are less likely to reach the concentration required because eukaryotic (真核状态的) cells do not have a tetracycline uptake mechanism. Tetracycline OH 7 6 8 H N(CH3)2 5 4 H OH 9 11 10 OH O 1 12 OH 3 OH 2 CONH2 O 6-Methyl-4-(dimethylamino)-3,6,10,12,12apentahydroxy-1,4,4a,5,5a,6,11,12a-octahydro-2naphthacenecarboxamide Stability under acid condition The tetracycline molecule, as well as those that contain the 6β-hydroxy group, is labile to acid and base degradation. At pH 2.0, tetracycline eliminates a molecule of water with concomitant aromatization of ring C to form anhydrotetracycline. OH2+ N OH OH H H OH H+ OH N H H CONH2 OH O CONH2 OH O OH O OH O N + H N OH H OH OH - H+ H OH CONH2 OH O OH O - H2O OH CONH2 OH OH O O ÍÑË®Îï Formation of 4-Epitetracycline At C-4 in acidic medium (pH 2-6), epimerization of the “natural” C-4 α-dimethylamino group to the C-4β-epimer occurs. Under acidic conditions, a 1:2 equilibrium is established in solution within a day. H+N OH N OH OH OH H OH H H OH H CONH2 CONH2 OH O OH OH O O N OH OH OH N OH O H H OH OH H H OH CONH2 OH O OH OH CONH2 OH O OH O 4-Epitetracycline Stability under base condition In basic medium, ring C of tetracycline is opened to form isotetracycline. O- N OH OH H H OH N OH OHH OH CONH2 OH O OH CONH2 O OH N H OH OH OH OH -O O O OH OH O N H OH OH O CONH2 O O O- O OH OH OH O CONH2 N O O CONH2 O Formation of metal chelates OH OH H H OH CONH2 OH O OH N(CH3)2 OH O Mn+ N(CH3)2 OH H H OH CONH2 OH O O O Mn+ Stable chelate complexes are formed by the tetracyclines with many metals, including calcium, magnesium, and iron. Such chelates are usually very insoluble in water. The affinity of tetracyclines for calcium causes them to incorporated into newly forming bones and teeth as tetracycline-calcium orthophosphated complexes. Deposits of these antibiotics in teeth cause a yellow discoloration. The tetracyclines are distributed into the milk of lactating mothers and will cross the placental barrier into the fetus. The possible effects of these agents on bones and teeth of the child should be considered before their use during pregnancy or in children under 8 years of age. Aminoglycoside Antibiotics The aminoglycoside class of antibiotics contains a pharmacophoric 1,3-diaminoinositol (1,3-二氨 基肌醇) derivatives H2N NH NH H2N OH HO HO NH OH Streptamine (链霉胺) NH2 NH HO HO HN NH2 OH HO HO HO 2-Deoxystreptamine Spectinamine (2-脱氧链霉胺) (放线菌胺) NH OH Chemistry (N-Methyl-LGlucosmine) HO HOH2C HO H2N NH NHCH3 O NH O HO OHC (Streptide) OH CH3O O HO NH OH NH2 NH (L-Streptose) Aminoglycosides are so named because their structures consist of amino sugars linked glycosidically. All have at least one aminohexose, and some have a pentose lacking an amino group. Caution ! It should be remember that penicillin and aminoglycoside antibiotics must never be physically mixted, because both are chemically inactivated to a significant degree on mixting. Chemistry Aminoglycosides are strong basic compounds that exist as polycations at physiological pH. Their inorganic acid salts are very soluble in water. All are available as sulfates. The high water solubility of the aminoglycosides no doubt contributes to their pharmacokinetic properties. They distribute well into most body fluids but not into the ventral nervous system, bone, or fatty or connective tissues. They tend to concentrate in the kidneys and excreted by glomerular filtration. Aminoglycosides are apparently not metabolized in vivo. Spectrum of activity Aminoglycosides are used for treatment of serious systemic infections caused by aerobic Gram-negative bacilli. Aerobic G-N and G-P cocci tend to be less sensitive; thus the β–lactams and other antibiotics tend to be preferred for the treatment of infections caused by these organisms. Anaerobic bacteria are invariably resistant to the aminoglycosides. Streptomycin is the most effective of the group for the chemotherapy of tuberculosis. Under certain circumstances, aminoglycoside and β– lactams antibiotics exert a synergistic action in vivo against some bacterial strains when the two are administered jointly. Mechanism of Action The mechanism of action of these antibiotics believed that they can inhibit the biosynthesis of protein of bacteria. At less than toxic doses, they bind to the protein portion of the 30S ribosomal subunit leading to mistranslation of RNA templates and the consequent insertion and wrong amino acids and formation so-called nonsense proteins. Toxicity Their undesirable side effects: severe ototoxicity and nephrotoxicity. 18 of 21 actress showing “qianshou guanyin” were caused deafness by aminoglycosides. Streptomycin(链霉素) HO HO HO O NHCH3 OH H N 2 CHO HN O O O HO NH OH H N OH NH2 NH Streptomycin is the first aminoglycosides isolated from Streptomyces griseus. There are three basic centers in the structure. Clinical Use Streptomycin was the first aminoglycoside isolated and the first antibiotic with potent activity against Mycobacterium tuberculosis and this antibiotic continues to be used to treat tuberculosis, but as a result of the development of resistance, now in combination therapy with other antibiotics. Streptomycin can also be used for the treatment of tularemia(野兔病), plague(瘟疫) and leprosy(麻风病). The aminoglycosides are highly water soluble and poorly absorbed orally. These antibiotics are therefore primarily delivered by intramuscular injection or intravenously. Macrolide Antibiotics Macrolide Antibiotics Naturally occurring macrolide antibiotics are grouped into three major groups of 12-, 14-, and 16-membered macrolides with the aglycone consisting of 12-, 14-, and 16-atom cyclic lactone rings, respectively. For example, erythromycin A is a 14-membered macrolide (a 14atom cyclic lactone ring) and possesses desosamine and cladinose glycosidically linked to C-5 and C-3, respectively. Mechanism of action The mechanism of action of macrolides is that: it inhibits bacteria by interfering with programmed ribosomal protein biosynthesis by inhibiting translocation of amino acid m-RNA following binding to the 50s subunit. Erythromycin (红霉素) Erythromycin is an orally effective antibiotic discovered in 1952 in the metabolic products of a strain of Streptomyces eryyhreus(红色链丝菌), it includes Erythromycin A, B, and C. The component A is used in clinic primarily. It is active for most G-P and some G-N. Erythromycin N HO O HO Erythronolide A HO 12 3' OH 1' 9 6 O O 3 O 1 O O O 1" 3" Desosamine Cladinose OH OMe Erythromycin A A and B A C-12=-OH B C-12=-H A and C A C-3"=OCH3 C C-3"=-OH Extremely unstable under acid condition O OH OH OH OH N O O O O HO 1. H+ 2. - H2O O OH N O O O O O O OH O HO O O OH O ÍÑË®Îï 8,9-Anhydroerythromycin A -6,9-hemiketal HO O O N O O O O HO OH O O + O O O N O O O OH ÂÝÐýͪ Anhydroerythromycin A -6,9-9,12-spiroketal O O OH HO O OH ¿ËÀ-¶¨ ÌÇ Cladinose Simply modification of erythromycin -Ester Pro-drug O OH OH OH N O O O O RO O O O OH ºì ùËØ̼ËáÒÒ õ¥ Ery thromy cin Ethy lcarbonate ºì ùËØÓ²Ö¬Ëáõ¥ Ery thromy cin Stearate çúÒÒ ºì ùËØ Ery thromy cin Ethy lsuccinate ÒÀÍкì ùËØ Ery thromy cin Estolate R = -COOCH2CH3 R = -CO(CH2)16CH3 R = -CO(CH2)2OCOCH2CH3 R = -COOCH2CH3, C12H25SO3H Strategy for erythromycin modification Replacement of hydrogen Conversion to amines Conversion to oxime Ring expansion Alkylation of hydroxylgroup H O NMe2 HO OH HO Conversion to 11,12-cyclic derivatives O O HO O O O O OH OMe Cut Cladinose to ketolides O N OH OH OH OH N O O HO O O O O OH OH O N O O O HO O OH O O N O OH O O OH O ÂÞºì ùËØ¡¡ Roxithromycin OH OH O OH O O O HO O O HN OH OH O O HO O OH O O N OH O HO O O O O Erythromycin Oxime O OH O Dirithromycin O O OH O O N O N O Beckmann Rearraangement OH O N N OH HN OH OH OH OH OH »¹ Ô- N O O OH O HO O O N O ¼×»ù»¯ O O HO O O O O O OH O OH °¢Æë ùËØ¡¡ Azithromycin Erythromycin derivatives O F OH O OH OH OH OH N O O O O HO O O O O O O ·ú ºì ùËØ Flunithromycin N OH O O HO O O O ¿ËÀ-ùËØ Clarithromycin OH Telithromycin Telithromycin is the first ketolide(3-keto macrolide derivatives). It is prepared by removing the cladinose sugar from the C-3 position of the erythronolide skeleton and oxidizing the remaining hydroxyl group to a keto group. N N N NMe2 HO O O N O OMe 9 6 11 12 O 3 O 1 O O Telithromycin O In addition to the C-3 ketone, telithromycin has an aromatic Nsubstituted carbamate extension at position C-11 and C-12. This ring has an imidazo-pyridyl group attachment. Telithromycin possesses a 6-OCH3 group (like clarithromycin), avoiding internal kemiketalization with the 3keto function and giving the ketolide molecule excellent acid stability. The ketolides are very active against respiratory pathogens, including erythromycin-resistant strains Chloramphenicol Antibiotics Chloramphenicol (氯霉素 ) O H O2N Cl H HN Cl OH HO H Chemical name: D-(-)-threo-1-p-nitrophenyl-2-dichloroacetamido1,3-propanediol A molecule, with two chiral centers, has four isomers (diastereomers). CHO H OH H OH CH2OH CHO HO H H OH CH2OH ³àÞºÌÇ D-(-)-erythrose ËÕ°¢ÌÇ D-(-)-threose NO2 HO H C 1 C 2 NO2 H H NHCOCHCl2 Cl2CHCOHN CH2OH 1 C 2 OH H CH2OH 1R, 2R (-) D-(-)-Threo C NO2 1S, 2S (+) ¡¡ ¡¡ ¡¡ ¡¡ L-(+)-Threo H H C 1 C 2 NO2 OH HO NHCOCHCl2 Cl2CHCOHN CH2OH 1 C 2 H H CH2OH 1S, 2R (+) ¡¡ d-(+)-Erythro¡¡ C 1R, 2S (+) ¡¡ ¡¡ ¡¡ ¡¡ ¡¡ L-(-)-Erythro Chloramphenicol is an antibiotic produced by Streptomyces venezuelae and other soil bacteria that was first discovered in 1947 and is now exclusively produced synthetically. With two chiral centers it is one of four diastereomers only one of which (1R, 2R) is active. Chemical properties O O2N H H O Cl HN Zn, HCl Cl HO H N H Cl Cl OH H OH ôÇ°· ÑÜÉúÎï Chloramphenicol Hydroxyamine OH O N O O Cl NH OH H H H HO O Cl NH N Cl OH H H OH FeCl3 O H HO H Cl NH Cl Fe OH H OH 3 Chloramphenicol is bacteriostatic by inhibition of protein biosynthesis. Its toxicities prevent Chloramphenicol from being more widely used. The major adverse effect of chloramphenicol is a risk of fatal irreversible aplastic anemia that occurs after therapy and does not appear to be related to dose or administration route. Reversible bone marrow suppression and several other adverse effects including gastrointestinal problems, headache, and mild depression have also been noted. Usage Despite potentially serious limitations, Chloramphenicol is an excellent drug when used carefully. Its special value is in typhoid (伤寒) and paratyphoid fever(副 伤寒), Haemophilus infection , pneumococcal (肺炎球菌) and meningococcal meningitis(脑膜炎) in βlactam allergic patients, anaerobic(厌氧菌) infection , rickettsial infections, and so on. Synthesis O2N O2N Br2, C6H5Cl (CH2)6N4, C6H5Cl O2N Br .(CH2)6N4 Br O O C2H5OH,HCl, H2O O2N O Ac2O, AcONa O2N O N H NH2.HCl O O p-Nitro- -aminophenylacetone HCHO, C2H5OH O H Al[OCH(CH3)2]3, HOCH(CH3)2 H OH (±)-thero-1-p-nitrophenyl-2acetamidopropane-1,3-diol O p-Nitro- -acetamido--hydroxyphenylpropanone HO O2N HCl, H2O 15% NaOH HO O2N H NH2 H OH H OH (±)-thero-1-p-nitrophenyl-2aminopropane-1,3-diol O H NH2 OH OH H D-(-)-thero-1-p-nitrophenyl2-aminopropane-1,3-diol Resolution H NH2 .HCl O2N O H N H N H pH = 2~7.5 HO O2N HO O2N Cl2CHCOOCH3, CH3OH O2N H HN OH H OH H Cl Cl Chloramphenicol Palmitate (棕榈氯霉素) O O2N H H Cl NH Cl O H OH O -C15H31 ×ØéµÂÈùËØ Chloramphenicol Palmitate Chloramphenicol Palmitate is the palmitic acid ester of chloramphenicol. It is a tasteless prodrug of chloramphenicol intended for pediatric use. The ester must hydrolyze in vivo following oral absorption to provide the active form. Chloramphenicol Sodium Succinate (琥珀氯霉素钠) O H O2 N H Cl NH Cl O H Na OH O OH O çúçêÂÈùËØ Chloramphenicol Succinate Chloramphenicol sodium succinate is the watersoluble sodium salt of the hemisuccinate ester of chloramphenicol. Because of the low solubility of chloramphenicol, the sodium succinate is preferred for intravenous administration. The availability of chloramphenicol from the ester following intravenous administration is estimated to be 70 to 75%. Summary Tetracyclines Aminoglycosides Macrolides O OH OH OH N O O O O Erythromycin Structure modification of semi-synthetic erythromycin Chloramphenicol Mechanism of action HO O O OH O O O2N H HO HN H H Cl Cl OH Question: 1. Why is the erythromycin A unstable in acidic condition? 2. What is the difference of the action mechanism of antibiotics? Assignment: 1.Read textbook pp334-355,360-361 2.Do homework Exercises of medicinal chemistry p96 Type A and药物化学学习指导,第八章