* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Metal carbonyl wikipedia , lookup
Elias James Corey wikipedia , lookup
Aromaticity wikipedia , lookup
Organosulfur compounds wikipedia , lookup
Hydroformylation wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Asymmetric induction wikipedia , lookup
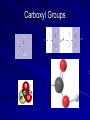
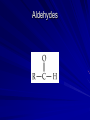
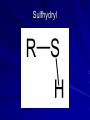
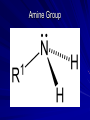
Macromolecules What are macromolecules? What makes up Macromolecules? CHNOPS The Six Most Abundant Elements of Life What makes something Organic? If it contains the element Carbon (C) and Hydrogen (H) Ex of an organic molecule… – HYDROCARBONS are important chemicals in biology…they contain only C and Hydrogen bonded in different proportions Hydrocarbons Name and Comments Molecular Structural Formula Formula H (a) Methane CH4 H C H H (b) Ethane H H C2H6 H C C H H H (c) Ethene (ethylene) H C2H4 H C C H H Ball-andStick Model SpaceFilling Model Functional Groups In organic chemistry, functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. Carboxyl Groups H H O O O H C H C C C H OH OH H + H+ C O Hydroxyl Group H H H C C H H Ethanol OH Carbonyl Group O C Carbonyl group consists of a carbon atom joined to an oxygen atom by a double bond (symbolized as C=O). Like hydroxyl groups, carbonyl groups contribute to making molecules water-soluble. All sugar molecules have one carbonyl group, in addition to hydroxyl groups on the other carbon atoms. Two Forms of Carbonyl Groups: Aldehydes and Ketones O H H C C H C H H H O O H C H Propanal, an aldehyde C C H H C H H Acetone, a ketone Aldehyde groups, where the C=O group is at the end of an organic molecule. A hydrogen atom is also located on the same carbon atom. Keto groups, where the C=O group is located within an organic molecule. All sugars have either a keto or an aldehyde group. An aldehyde and a ketone may be structural isomers with different properties, as is the case for acetone and propanal. Aldehydes Functional Groups Phosphate O O P OH OH Sulfhydryl Amine Group Putting things together…let’s make some MACROMOLECULES Monomers & Polymers: • monomers • smaller building blocks of the macromolecules •POLYMERS: • proteins: •Mono=Amino acids • nucleic acid: •Mono= nucleotides • carbohydrate: •Mono= monosaccharides • lipid: •Mono=Fatty acids Putting together / Taking apart macromolecules: macromolecules: monomers <--> polymers Dehydration Synthesis Hydrolysis • • • • fig 4.3 Synthesis = dehydration synthesis Removal of OH + H forms water, H2O Leaves bonding sites that join monomer units. 19 Aug. 2009 Chemistry.ppt 20 Disassembly = hydrolysis Break bond between monomer units. Add OH + H to fill bonding sites. 19 Aug. 2009 Chemistry.ppt 21 Fill in Chart… For each Macromolecule – Made of (elements) – Basic Unit (monomer) – Function – Where found – Examples Proteins - Composition Proteins can be made up of any or all of… – Carbon – Hydrogen – Nitrogen – Oxygen – Sulfur