* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 2
Energy efficiency in transport wikipedia , lookup
Internal energy wikipedia , lookup
Alternative energy wikipedia , lookup
Negawatt power wikipedia , lookup
Energy policy of the European Union wikipedia , lookup
Conservation of energy wikipedia , lookup
Low-carbon economy wikipedia , lookup
Energy Independence and Security Act of 2007 wikipedia , lookup
Energy in the United Kingdom wikipedia , lookup
Artificial photosynthesis wikipedia , lookup
Water splitting wikipedia , lookup
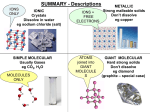
Chapter 2 Science, Systems, Matter, and Energy TYPES AND STRUCTURE OF MATTER Elements and Compounds Matter exists in chemical forms as elements and compounds. • Elements (represented on the periodic table) are the building blocks of matter. • Compounds: two or more different elements held together in fixed proportions by chemical bonds. Basic Chemistry Matter, Mass, and Weight Matter: Anything that occupies space and has mass Mass: The amount of matter in an object Weight: The gravitational force acting on an object of a given mass Elements and Atoms Elements: The simplest type of matter with unique chemical properties Atoms: Smallest particle of an element that has chemical characteristics of that element Atoms Figure 2-4 Atomic Structure Atoms: composed of subatomic particles Neutrons: no electrical charge Protons: positive charge Electrons: negative charge Nucleus Formed by protons and neutrons Most of volume of atom occupied by electrons Atomic Number and Mass Number Atomic Number: Equal to number of protons in each atom which equals the number of electrons Mass Number: Number of protons plus number of neutrons Isotopes and Atomic Mass Isotopes: Two or more forms of same element with same number of protons and electrons but different neutron number 3 types of hydrogen Denoted by using symbol of element preceded by mass number as 1H, 2H, 3H Atomic Mass: Average mass of naturally occurring isotopes Molecules and Compounds Molecules: Two or more atoms form a covalent bond Example: Water Compounds: A substance composed of two or more different types if atoms chemically combined Example: Hydrogen Molecule Molecular Mass: Determined by adding up atomic masses of its atoms or ions Example: NaCl (22.99 + 35.45) Covalent Bonding Atoms share one or more pairs of electrons Single covalent: Electron pair between 2 atoms Double covalent: Two atoms share 4 electrons Nonpolar covalent: Electrons shared equally Polar covalent: Electrons not shared equally Electrons and Chemical Bonding Ion: When an atom loses or gains electrons and become charged Cation: Positively charged ion Anion: Negatively charged ion Ionic Bonding Cations and anions are attracted to each other Ions An ion is an atom or group of atoms with one or more net positive or negative electrical charges. The number of positive or negative charges on an ion is shown as a superscript after the symbol for an atom or group of atoms Hydrogen ions (H+), Hydroxide ions (OH-) Sodium ions (Na+), Chloride ions (Cl-) Compounds and Chemical Formulas Chemical formulas are shorthand ways to show the atoms and ions in a chemical compound. Combining Hydrogen ions (H+) and Hydroxide ions (OH-) makes the compound H2O (dihydrogen oxide, a.k.a. water). Combining Sodium ions (Na+) and Chloride ions (Cl-) makes the compound NaCl (sodium chloride a.k.a. salt). Synthesis and Decomposition Reactions Synthesis Reactions Two or more reactants chemically combine to form a larger product Decomposition Reactions Reverse of synthesis reactions Acids and Bases; Salts and Buffers Acid: A proton donor or any substance that releases hydrogen ions Bases: A proton acceptor or any substance that binds to or accepts hydrogen ions Buffers: Able to maintain the pH The pH Scale Refers to the Hydrogen ion concentration in a solution Neutral: pH of 7 or equal hydrogen and hydroxide ions Acidic: a greater concentration of hydrogen ions Alkaline or basic: a greater concentration of hydroxide ions The pH (potential of Hydrogen) is the concentration of hydrogen ions in one liter of solution. Figure 2-5 Chemistry Inorganic Chemistry: Generally substances that do not contain carbon Water Oxygen Organic Chemistry: Study of carboncontaining substances Organic Compounds: Carbon Rules Organic compounds contain carbon atoms combined with one another and with various other atoms such as H+, N+, or Cl-. Contain at least two carbon atoms combined with each other and with atoms. Methane (CH4) is the only exception. All other compounds are inorganic. Organic Chemistry Carbohydrates Composed of carbon, hydrogen, oxygen Lipids Composed mostly of carbon, hydrogen, oxygen Proteins Composed of carbon, hydrogen, oxygen,nitrogen Nucleic Acids: DNA and RNA Composed of carbon, hydrogen, oxygen, nitrogen, phosphorus Adenosine Triphosphate (ATP) Composed of adenosine and three phosphate groups Carbohydrates Monosaccharides or building blocks Simple sugars: glucose, fructose, galactose Disaccharides Two simple sugars bound together by dehydration: sucrose, lactose, maltose Polysaccharides Long chains of many monosaccharides: glycogen in animals; starch and cellulose in plants Lipids Lipids: Can be dissolved in nonpolar organic solvents as alcohol or acetone but relatively insoluble in water Fats, Oils and Waxes. Triglycerides: composed of glycerol and fatty acids Phospholipids: Important structural component of cell membranes Steroids: Cholesterol, bile salts, estrogen, testosterone Fat-soluble Vitamins Proteins Amino acids: The building blocks of protein Peptide bonds: Covalent bonds formed between amino acids during protein synthesis Structure Primary, secondary, tertiary, quartenary Enzymes: Protein catalysts Lock-and-key model Active site Coenzymes Protein Structure and Enzyme Action Nucleic Acids: DNA and RNA DNA: Deoxyribonucleic acid Genetic material of cells copied from one generation to next Composed of 2 strands of nucleotides • Each nucleotide contains one of the organic bases of adenine or guanine which are purines and thymine or cystosine which are pyrimidines RNA: Ribonucleic acid Similar to a single strand of DNA • Four different nucleotides make up organic bases except thymine is replaced with uracil (pyrimidine) DNA Structure Water Inorganic Stabilizes body temperature Protection Necessary for many chemical reactions of life Mixing Medium Mixture: Substance physically but not chemically combined • Solution: Liquid, gas, or solid uniformly distributed Solvent: What dissolves the solute Solute: What is to be dissolved Acids and Bases; Salts and Buffers Acid: A proton donor or any substance that releases hydrogen ions Bases: A proton acceptor or any substance that binds to or accepts hydrogen ions Salts: A cation consisting of other than a hydrogen ion and other than an anion or hydroxide ion Buffers: A solution of a conjugate acidbase pair in which acid and base The pH Scale Refers to the Hydrogen ion concentration in a solution Neutral: pH of 7 or equal hydrogen and hydroxide ions Acidic: a greater concentration of hydrogen ions Alkaline or basic: a greater concentration of hydroxide ions Energy Energy: The capacity to do work Potential Energy: Stored energy Kinetic Energy: Does work and moves matter Mechanical Energy: Energy resulting from the position or movement of objects Chemical Energy: Form of potential energy in the chemical bonds of a substance Heat Energy: Energy that flows between objects of different temperatures Energy and Chemical Reactions Speed of Chemical Reactions Activation Energy: Minimum energy reactants must have to start a chemical reaction Catalysts: Substances that increase the rate of chemical reactions without being permanently changed or depleted Enzymes: Increase the rate of chemical reactions by lowering the activation energy necessary for reaction to begin Activation Energy and Enzymes Cells: The Fundamental Units of Life Cells are the basic structural and functional units of all forms of life. Prokaryotic cells (bacteria) lack a distinct nucleus. Eukaryotic cells (plants and animals) have a distinct nucleus. Figure 2-6 Animation: Prokaryotic and Eukaryotic Cells PLAY ANIMATION A human body contains trillions of cells, each with an identical set of genes. There is a nucleus inside each human cell (except red blood cells). Each cell nucleus has an identical set of chromosomes, which are found in pairs. A specific pair of chromosomes contains one chromosome from each parent. Each chromosome contains a long DNA molecule in the form of a coiled double helix. Genes are segments of DNA on chromosomes that contain instructions to make proteins—the building blocks of life. The genes in each cell are coded by sequences of nucleotides in their DNA molecules. Stepped Art Fig. 2-7, p. 38 All Living Organisms are made up of Macromolecules: Complex Carbohydrates Proteins Nucleic Acids Lipids Figure 2-7 States of Matter The atoms, ions, and molecules that make up matter are found in three physical states: solid, liquid, gaseous. A fourth state, plasma, is a high energy mixture of positively charged ions and negatively charged electrons. The sun and stars consist mostly of plasma. Scientists have made artificial plasma (used in TV screens, gas discharge lasers, florescent light). Matter Quality Matter can be classified as having high or low quality depending on how useful it is to us as a resource. High quality matter is concentrated and easily extracted. low quality matter is more widely dispersed and more difficult to extract. Figure 2-8 CHANGES IN MATTER Matter can change from one physical form to another or change its chemical composition. When a physical or chemical change occurs, no atoms are created or destroyed. • Law of conservation of matter. Physical change maintains original chemical composition. Chemical change involves a chemical reaction which changes the arrangement of the elements or compounds involved. • Chemical equations are used to represent the reaction. Chemical Change Energy is given off during the reaction as a product. Types of Pollutants Factors that determine the severity of a pollutant’s effects: chemical nature, concentration, and persistence. Pollutants are classified based on their persistence: Degradable pollutants Biodegradable pollutants Slowly degradable pollutants Nondegradable pollutants Nuclear Changes in Atoms Natural radioactive decay: unstable isotopes spontaneously emit fast moving chunks of matter (alpha or beta particles), high-energy radiation (gamma rays), or both at a fixed rate. Radiation is commonly used in energy production and medical applications. The rate of decay is expressed as a half-life (the time needed for one-half of the nuclei to decay to form a different isotope). Animation: Positron-Emission Tomography PLAY ANIMATION Animation: Half-Life PLAY ANIMATION Nuclear Changes: Fission Nuclear fission: nuclei of certain isotopes with large mass numbers are split apart into lighter nuclei when struck by neutrons. Figure 2-9 Uranium-235 Uranium-235 Uranium-235 Energy Fission fragment Uranium-235 n n Neutron n n Energy n Uranium-235 Uranium-235 Energy n Fission fragment Uranium-235 Energy Uranium-235 Uranium-235 Uranium-235 Stepped Art Fig. 2-6, p. 28 Nuclear Changes: Fusion Nuclear fusion: two isotopes of light elements are forced together at extremely high temperatures until they fuse to form a heavier nucleus. Figure 2-10 Video: Nuclear Energy PLAY VIDEO From ABC News, Environmental Science in the Headlines, 2005 DVD. ENERGY Energy is the ability to do work and transfer heat. Kinetic energy – energy in motion • heat, electromagnetic radiation Potential energy – stored for possible use • batteries, glucose molecules Electromagnetic Spectrum Many different forms of electromagnetic radiation exist, each having a different wavelength and energy content. Figure 2-11 Electromagnetic Spectrum Organisms vary in their ability to sense different parts of the spectrum. Figure 2-12 Animation: Visible Light PLAY ANIMATION Source of Energy Electricity Very high temperature heat (greater than 2,500°C) Nuclear fission (uranium) Nuclear fusion (deuterium) Concentrated sunlight High-velocity wind Relative Energy Tasks Energy Quality (usefulness) Very high-temperature heat (greater than 2,500°C) for industrial processes and producing electricity to run electrical devices (lights, motors) High-temperature heat (1,000–2,500°C) Hydrogen gas Natural gas Gasoline Coal Food Mechanical motion to move vehicles and other things) High-temperature heat (1,000–2,500°C) for industrial processes and producing electricity Normal sunlight Moderate-velocity wind High-velocity water flow Concentrated geothermal energy Moderate-temperature heat (100–1,000°C) Wood and crop wastes Moderate-temperature heat (100–1,000°C) for industrial processes, cooking, producing steam, electricity, and hot water Dispersed geothermal energy Low-temperature heat (100°C or lower) Low-temperature heat (100°C or less) for space heating Fig. 2-13, p. 44 ENERGY LAWS: TWO RULES WE CANNOT BREAK The first law of thermodynamics: we cannot create or destroy energy. We can change energy from one form to another. The second law of thermodynamics: energy quality always decreases. When energy changes from one form to another, it is always degraded to a more dispersed form. Energy efficiency is a measure of how much useful work is accomplished before it changes to its next form. Chemical energy (photosynthesis) Solar energy Waste Heat Mechanical energy (moving, thinking, living) Chemical energy (food) Waste Heat Waste Heat Waste Heat Fig. 2-14, p. 45 SUSTAINABILITY AND MATTER AND ENERGY LAWS Unsustainable High-Throughput Economies: Working in Straight Lines Converts resources to goods in a manner that promotes waste and pollution. Figure 2-15 Sustainable Low-Throughput Economies: Learning from Nature Matter-Recycling-and-Reuse Economies: Working in Circles Mimics nature by recycling and reusing, thus reducing pollutants and waste. It is not sustainable for growing populations. Inputs (from environment) Energy Matter System Throughputs Outputs (into environment) Energy conservation Waste and pollution Low-quality Energy (heat) Sustainable low-waste economy Pollution control Matter Feedback Waste and pollution Recycle and reuse Energy Feedback Fig. 2-16, p. 47 Animation: Economic Types PLAY ANIMATION