* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download PTH Vit D
Survey
Document related concepts
Transcript
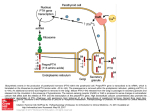
Pharmacologic Options for Osteoporosis Prevention and Treatment Alireza Khabbazi, MD BONE PHYSIOLOGY • Most common bone disease in humans • Characterized by: – Low bone mass – Microarchitectural deterioration – Compromised bone strength – Increased risk for fracture BONE PHYSIOLOGY • Bone Resorption – Osteoclast • Bone Formation – Osteoblast • Bone Multicellular Unit (BMU) – Positive < age 30 – Negative > age 30 Remodeling (Resorption) Bone Multicellular Unit (BMU) Lining cells Remodeling (Resorption) Bone Multicellular Unit (BMU) Lining cells Remodeling (Resorption) Lymphocyte CSF Macrophage Bone Multicellular Unit (BMU) Remodeling (Resorption) Lymphocyte PTH Vit D CSF Macrophage Osteoblast Bone Multicellular Unit (BMU) Remodeling (Resorption) Lymphocyte PTH Vit D CSF Macrophage Preosteoclast Osteoblast Bone Multicellular Unit (BMU) Remodeling (Resorption) Lymphocyte PTH Vit D Bone Multicellular Unit (BMU) CSF Macrophage Preosteoclast RANKL RANK IL1,TNFα,IL6 Osteoblast Osteoclast Remodeling (Resorption) Lymphocyte PTH Vit D Bone Multicellular Unit (BMU) CSF Macrophage Preosteoclast RANKL RANK IL1,TNFα,IL6 Osteoclast Osteoblast Cathepsin K Remodeling (Formation) Lymphocyte Estrogen OPG Osteoclast inhibition RANKL RANK IL1,TNFα,IL6 Osteoblast Remodeling (Formation) Lymphocyte Estrogen OPG Osteoclast inhibition RANKL RANK IL1,TNFα,IL6 Osteoblast Remodeling (Formation) Lymphocyte - PTH Vit D Estrogen Mechanical stimuli OPG Osteoclast inhibition IGF Estrogen Androgen RANKL RANK IL1,TNFα,IL6 Osteoblast Osteoblast stimulation Remodeling (Formation) Osteoblast proliferation Remodeling (Formation) Lining cells Osteocyte Osteoid Etiology and pathogenesis of Osteoporosis Pathogenesis PTH Vit D Estrogen Hyperparathyroidism Estrogen deficiency Inflammation Low Vitamin D, Ca Low physical activity Androgen deficiency Drugs toxins PTH Vit D Progestrone OPG Estrogen Androgen IGF RANKL RANK IL1,TNFα,IL6 Osteoclast Osteoblast Osteoblast Cathepsin K Pathogenesis PTH Vit D Estrogen Hyperparathyroidism Estrogen deficiency Inflammation Low Vitamin D, Ca Low physical activity Androgen deficiency Drugs toxins PTH Vit D Progestrone OPG Estrogen Androgen IGF RANKL RANK IL1,TNFα,IL6 Osteoclast Osteoblast Osteoblast Cathepsin K Pathogenesis PTH Vit D Estrogen Hyperparathyroidism Estrogen deficiency Inflammation Low Vitamin D, Ca Low physical activity Androgen deficiency Drugs toxins PTH Vit D Progestrone OPG Estrogen Androgen IGF RANKL RANK IL1,TNFα,IL6 Osteoclast Osteoblast Osteoblast Cathepsin K Pathogenesis PTH Vit D Estrogen Hyperparathyroidism Estrogen deficiency Inflammation Low Vitamin D, Ca Low physical activity Androgen deficiency Drugs toxins PTH Vit D Progestrone OPG Estrogen Androgen IGF RANKL RANK IL1,TNFα,IL6 Osteoclast Osteoblast Osteoblast Cathepsin K Pathogenesis PTH Vit D Estrogen Hyperparathyroidism Estrogen deficiency Inflammation Low Vitamin D, Ca Low physical activity Androgen deficiency Drugs toxins PTH Vit D Progestrone OPG Estrogen Androgen IGF RANKL RANK IL1,TNFα,IL6 Osteoclast Osteoblast Osteoblast Cathepsin K Pathogenesis PTH Vit D Estrogen Hyperparathyroidism Estrogen deficiency Inflammation Low Vitamin D, Ca Low physical activity Androgen deficiency Drugs toxins PTH Vit D Progestrone OPG Estrogen Androgen IGF RANKL RANK IL1,TNFα,IL6 Osteoclast Osteoblast Osteoblast Cathepsin K Pathogenesis PTH Vit D Estrogen Hyperparathyroidism Estrogen deficiency Inflammation Low Vitamin D, Ca Low physical activity Androgen deficiency Drugs toxins PTH Vit D Progestrone OPG Estrogen Androgen IGF RANKL RANK IL1,TNFα,IL6 Osteoclast Osteoblast Osteoblast Cathepsin K Pathogenesis PTH Vit D Estrogen Hyperparathyroidism Estrogen deficiency Inflammation Low Vitamin D, Ca Low physical activity Androgen deficiency Drugs toxins PTH Vit D Progestrone OPG Estrogen Androgen IGF RANKL RANK IL1,TNFα,IL6 Osteoclast Osteoblast Osteoblast Cathepsin K Drugs used for osteoporosis treatment Odanacatib Denosumab Strontium Teriparatide Treatment PTH Estrogen PTH Vit D + OPG _ + _ _ Estrogen Androgen IGF RANKL RANK IL1,TNFα,IL6 Progestrone Osteoclast + Osteoblast _ Cathepsin K SERMs Biphosphonates Calcitonine Ca, Vit D Exercise Osteoblast Drugs used for osteoporosis • Antiresorptive drugs - Bisphosphonates Calcitonin Estrogen SERM’s Denosumab • Bone formation stimulators - PTH (Teriparatide) • Dual action - Strontium Vitamin D and calcium Vitamin D and calcium • Daily 800 IU of vitamin D is associated with a reduction in hip fractures of 30% and in nonvertebral fractures of 14% • Fall reduction in the elderly • Therapeutic dose of vitamin D: 600-800 unit Vitamin D and calcium • Calcium: 1000-1300mg • Avoid taking more than 500 mg of calcium in one dose. • Take one dose before bedtime to prevent bone loss at night. If more is needed, take several doses throughout the day. • Calcium supplements should be taken with meals to boost their absorption. Vitamin D and calcium • Certain substances can hinder absorption of calcium: foods rich in fibres and fat, zinc, iron, spinach, coffee, alcohol and antacids. Therefore, calcium should not be taken together with these. • Calcium may interfere with certain drugs, including: thyroid medications, tetracycline, anticonvulsants and corticosteroids. Therefore, these should always be taken separately. Vitamin D and calcium • There is no need to worry about development of kidney stones if the correct dosage in the suitable form of calcium is taken together with sufficient fluid. • Calcium supplements can cause gas, abdominal distension and constipation in some individuals. In this situation, it is reasonable to switch to a different preparation. Bisphosphonates Bisphosphonates • The most commonly prescribed therapy for OP prevention and management • Mechanism: inhibits bone resorption by attaching to bony surfaces undergoing active resorption and inhibiting action of osteoclasts - Farensyl pyrophosphate synthase -GTPase -Attachment of osteoclast Bisphosphonates-place in treatment • Prevention and treatment of postmenopausal osteoporosis • Osteoporosis in men • Prevention and treatment of GIOP Bisphosphonates • Alendronate use for 10 years cause a continuous increase in vertebral (13.7%) and hip trochanter (10.3%) bone mineral density (BMD) • Decrease incidence of vertebral, hip, and all non-vertebral fractures by 50% • 90% reduction of multiple radiographic vertebral fractures at year 3 Bisphosphonates-Adverse events • Gastrointestinal problems, such as difficulty swallowing, gastric ulcers, and inflammation of the esophagus. • Hypocalcemia (18%) • Hypophosphatemia (10%) • Musculoskeletal pain, cramps Bisphosphonates-Adverse events • Atrial fibrillation (3-50/100000): zoledronic acid • Osteonecrosis of the jaw (1/100000): IV bisphosphonates • Atypical femur fracture Bisphosphonates-Contraindications • Abnormalities of the esophagus which delay esophageal emptying, such as stricture or achalasia • Inability to stand or sit upright for at least 30 minutes • Patients at increased risk of aspiration • Hypocalcemia • Pregnancy • Renal insufficiency (Not recommended if CrCl < 30-35 ml/min) Bisphosphonates-Dosing • Alendronate: 10 mg/day or 70 mg once weekly at least 30 minutes before eating or drinking • Risedronate: 5 mg/day, 35 mg once weekly, or 150 mg once monthly • Ibandronate: 150 mg once monthly at least 60 minutes before eating or drinking • Zoledronate: 5 mg administered intravenously (IV) once yearly Bisphosphonates-Duration of therapy • The optimal duration: 5 years Repair (up to 12 months) Rebuilding (6–36 months) Maintenance (24–60 months) • After 7 years of therapy the bone mass still increased by about 1% a year. However, a drug holiday after 5 years of alendronate therapy is advisable to avoid any possible microdamage accumulation, at least in low-risk patients. Other medications Raloxifene • Mechanism: tissue-selective activity, acts as an estrogen agonist on bone - Estrogen antagonist on breast, uterus • Approved only for the prevention and treatment of postmenopausal osteoporosis. Raloxifene • Reduce the incidence of vertebral fractures by 30-50% • Reduction of the risk of invasive breast cancer in postmenopausal women with osteoporosis • Reduction in the risk of invasive breast cancer in postmenopausal women at high risk of invasive breast cancer. Raloxifene: Adverse events • Frequency > 10% – Hot flashes – Arthralgia – Sinusitis • Frequency 1-10% – – – – – Chest pain Insomnia Migraines Peripheral edema Diaphoresis • An increased risk of DVT: risk is similar to reported risk of HRT Raloxifene: Containdications • • • • History of DVT/PTE or at high risk Cardiovascular disease History of uterine/cervical carcinoma Discontinue at least 72 hours prior to and during prolonged immobilization Raloxifene-Dosing • For prevention and treatment – 60 mg PO once daily • Can be taken any time of day without regard to meals Calcitonin • Approved for treatment of postmenopausal osteoporosis • Mechanism: – Peptide composed of 32 amino acids which binds to osteoclasts and inhibits bone resorption Calcitonin-Dosing • Nasal 200 international units daily • Contraindications – Clinical allergy to calcitonin-salmon • Precautions – Nasal ulcerations – Tachyphylaxis (parenteral dosage forms) • Drug interactions (DI) – No formal studies designed to evaluate DI Teriparatide • Prevention and treatment of postmenopausal osteoporosis • Treatment of osteoporosis in men • Treatment of GIOP Teriparatide • The BMD of the lumbar spine increased by 12.2% in the women receiving teriparatide and by 5.6% in the women receiving alendronate (at 14 months) Body et al. 2002 • Teriparatide was found to reduce vertebral fractures by 65% and nonvertebral fractures by 53% Teriparatide-Place in Therapy • Women or men with severe osteoporosis and at least one fragility fracture • Patients who are refractory to or unable to tolerate bisphosphonate In patients considered to be bisphosphonate failures PTH may be started approximately 3 months after bisphosphonates are discontinued Teriparatide-Adverse events • • • • • Hypercalcemia (11%) Dizziness (9%) Leg cramps (3%) Hyperuricemia Increased risk of osteosarcoma (rats) Teriparatide-Dosing • 20 µg daily as a daily subcutaneous injection for a maximum 2 years • Anti resorptive therapy may be considered after discontinuation of PTH to maintain gains in BMD acquired with PTH alone in those at high risk for subsequent fracture Teriparatide-contraindicated • Patients with an increased risk of osteosarcoma – Paget’s disease of bone – Prior radiation therapy to skeleton – unexplained elevations of ALP • • • • Bone metastases Hypercalcemia History of skeletal malignancy Pregnancy/nursing Denosumab • Reduces bone resorption by preventing RANK ligand, which is produced by the osteoblast, from attaching to its receptor on the osteoclast • The BMD increase 3-6.7% in the spine and 1.9-3.6% at the hips (1 years) Mc Clung et al. 2006 • The BMD increase up to 18.5% in the spine and 8.2% at the hips (6 years) Papapoulos et al. 2013 Denosumab A 3-year study of postmenopausal women with osteoporosis with the use of denosumab versus placebo - 68% relative decrease in the risk of new radiographic vertebral fracture - 40% relative decrease in the risk of hip fracture - 20% relative decrease in the risk of nonvertebral fracture Denosumab • Approval for the treatment of OP in - Postmenopausal women with a high risk of fracture, such as those with a history of fracture, women with multiple risk factors for fracture - Patients who failed or are intolerant of other available therapies Denosumab • The recommended dosing of denosumab is 60 mg subcutaneously every 6 months. • Unlike the bisphosphonates, denosumab may be used in those with renal impairment provided that monitoring of calcium, phosphorus, and magnesium is carried out Denosumab-adverse events • • • • Back pain (34.7%) Musculoskeletal pain (7.6%) High cholesterol (7.2%) Cystitis (5.9%) Strontium • Consisting of two atoms of stable strontium and an organic moiety (ranelic acid) • Strontium (2 gm/d) for three years: - Increased in BMD at the LS as 8% - Decrease in risk of new vertebral fractures, hip fracture and nonvertebral fracture by 40%, 40% and 15% respectively Strontium • Diarrhea • Drug rash with eosinophilia systemic symptoms and Stevens-Johnson syndrome (very rarely) • Thromboembolic events should be used with caution in patients • Strontium Cardiovascular risk who have had a thrombosis, and the treatment must Strontium should not be be used in patients be stopped in prolonged decubitus situations and in with IHD, peripheral arterial diseases, CVA, patients with skin reactions un controlled HTN Odanakatib • Selective catepsin K inhibitor • Dose: 50 mg/week PO • In a phase III trial the BMD of the lumbar spine increased by 5.5% and the hip increased by 3.2% Engelke et al. J Bone Miner Res. 2014 Monoclonal antibody against Sclerostin Who should be treated • All postmenopausal women who have had an osteoporotic vertebral fracture • T-score ≤-2 • T-score from -1.5 to -2 plus at least one of the following risk factors for fracture: thinness, history of fragility fracture (other than skull, facial bone, ankle, finger, and toe) since menopause, and history of hip fracture in a parent. Who should be treated-NOF guideline • Postmenopausal women and men age 50 and older who present with - T ≤-2.5 at the femoral neck or spine - Hip or vertebral fracture - T= -1.0 to -2.5 at the femoral neck or spine and a 10-year probability of a hip fracture ≥3% or a 10-year probability of a major osteoporosis-related fracture ≥20% FRAX Selection of drug • Bisphosphonates recommended as firstline therapy for postmenopausal osteoporosis. • Oral bisphosphonates are preferred • For individuals with gastrointestinal intolerance: IV bisphosphonates Selection of drug • Raloxifene was reserved for patients who cannot tolerate any bisphosphonates or for women with osteoporosis and increased risk of invasive breast cancer. Selection of drug • Denosumab could be used as initial therapy in certain patients at high risk for fracture, such as older patients who have difficulty with the dosing requirements of oral bisphosphonates. • Denosumab may have a role in patients who are intolerant of or unresponsive to other therapies and in those with impaired renal function. Selection of drug • Teriparatide should be reserved for treating women at high fracture risk, including those with very low bone mineral density (T-score worse than -3.0) with a previous vertebral fracture. Follow up strategies • Repeat DXA every 2 year using the same machine and technician if possible • The lowest significant change is 3-4% at spine, 4-6% at hip and 2% at elbow • If desired response not achieved - Re-evaluate the adherence to treatment regimen - Reconsider secondary OP - Consider changing patient medication Combination therapy • Combination of bisphosphonateraloxifene therapy is not recommended as the additional BMD benefits are small and there is no proven additional fracture benefit. • Combination therapy using PTH with raloxifene may enhance the bone forming effects of PTH. Combination therapy • Combination of PTH with alendronate blunts the effect of PTH and is not recommended • Teriparatide combined with denosumab increased BMD more than either agent alone and more than has been reported with other approved Tsai JN, Uihlein AV, Lee H, et al. Teriparatide and denosumab, alone or combined, in women with postmenopausal osteoporosis: the DATA randomised trial. Lancet 2013; 382:50–56.