* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download The AL-1 Story
Drug design wikipedia , lookup
Clinical trial wikipedia , lookup
Drug discovery wikipedia , lookup
Drug interaction wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Prescription costs wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
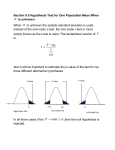
The Stavudine Story Chemical name: 2'-3'-didehydro-2'-3'-dideoxythymidine (d4T) Commercial name: Zerit®) International Non-proprietary name (通用名) : Stavudine Mechanism of action Stavudine is an analog of thymidine. It is phosphorylated by cellular kinases into active triphosphate. Stavudine triphosphate inhibits the HIV revers transcriptase by competing with natural substrate, thymidine triphosphate. It also causes termination of DNA synthesis by incorporating into it. Simultaneous use of AZT is not recommended, as it can inhibit the intracellular phosphorylation of stavudine. Other anti-HIV drugs do not possess this property. Discovery of d4T. Stavudine (d4T) was initially synthesized by Dr. Jerome Horowitz of the Michigan Cancer Foundation (Karmanos Cancer Institute) in 1966. Discovery that d4T would treat HIV/AIDS Dr. Tai-Shun Lin (Yale) and Dr. William Prusoff (Yale) first discovered d4T's capability to treat HIV/AIDS. Yale University holds the key use patent. The patent number in the USA is 4,978,655 (Patent #4,978,655). The fourth antiretroviral drug on the market, its patent expired in the United States on 2008-06-25. Professors. William Prusoff and Tai-Shun Lin Professors Prusoff and Wang at Yale in August 2009 Stavudine was approved by the U.S. FDA in Jun 24, 1994 for adults and in Sep 6, 1996 for pediatric use and again as an extended-release version for once-a-day dosing in 2001. Stavudine sales from $100-700m /year, and had generated $276M for Yale University Yale Univerisity-the hood where D4T was synthesized Idoxuridine It was discovered by Prof. William Prussof at Yale University and was introduced in 1963 for the treatment of herpes keratitis (角膜炎). It is the first FDA approved antiviral drug for human use. The Onrigin Story US brand names: Cloretazine, Onrigin Code name: VNP40101M Chemical structure names: 1,2-bis(methylsulfonyl)-1-(2chloroethyl)-2-(methylaminocarbonyl)hydrazine Professor Alan Sartorelli and Wang, Summer 2007 Trials Indication Dates Status Phase III trial with standard remissioninduction therapy AML and MDS December 2008 Ongoing Phase I/II trial in combination with daunorubicin and Aracytine Previously untreated adult AML with unfavorable cytogenetics March 2009 Ongoing Phase I/II trial in combination with cytarabine Elderly AML and MDS May 2008 Ongoing Phase I/II trial in combination with temozolomide Brain tumors, adult September 2007 Ongoing Phase III trial in combination with AraC AML, relapsed March 2005 Closed Phase II single agent trial AML, elderly poor-risk May 2006 Closed Phase II single agent trial Small cell lung cancer September 2005 Closed Phase I trial in combination with stem cell transplantation Hematologic malignancies November 2007 Closed Phase II single agent trial Brain tumors, adult June 2004 Completed Phase II single agent trial AML and high-risk myelodysplastic syndromes, elderly; AML, relapsed March 2004 Completed Phase I/II single agent trial Chronic lymphocytic leukemia July 2005 Completed Phase I trial Brain tumors, pediatric April 2005 Completed Phase I trial in combination with temozolomide Hematologic malignancies October 2004 Completed Phase I trial in combination with Ara-C Hematologic malignancies July 2003 Completed Phase I single agent trial Solid tumors February 2003 Completed Phase I single agent trial Hematologic malignancies August 2002 Completed Phase I single agent trial Solid tumors June 2001 Completed Sept. 1, 2009 NEW HAVEN, Conn. (TheStreet) -- The FDA's cancer drug advisory panel voted unanimously against approval of Vion's leukemia drug Onrigin. The panel recommended that a new randomized controlled trial be conducted before the drug is approved. The same panel rejected Genzyme's leukemia drug during a morning session for similar reasons. The Memantine Story (1-Amino-3,5-dimethyladamantadine Hydrochloride) (Axura® and Akatinol® by Merz, Namenda® by Forest and Ebixa® by Lundbeck). The first in a novel class of Alzheimer's disease medications acting on the NMDA receptor. Memantine is a low-affinity voltage-dependent uncompetitive antagonist at glutamatergic NMDA receptors. By binding to the NMDA receptor with a higher affinity than Mg2+ ions, memantine is able to inhibit the prolonged influx of Ca2+ ions. NH 2HCl H3 C CH 3 The early development of Memantine as an Alzheimer's disease treatment was carried by the biotech company Neurobiological Technologies led by a team including Professors Stuart Lipton, Jonathan Stamler, Yuqiang Wang and Dr. James W. Larrick From year 2007, memantine sales >$1000 M worldwide. Prof. Stuart Lipton, MD; PhD Burham Institute Prof. Jonathan Stamler, MD; PhD, Duke University James Larrick, MD; PhD, Panorama Research, Inc Prof. Yuqiang Wang, PhD, Jinan University The AL-1 Story O O O S O S CH2 HO CH2OH AL-1, synthesized by Professor Yuqiang Wang, is under development as an anti-diabetic drug currently. AL-1 is dual-functional, i.e., lowering blood glucose and protecting islet betacells. Significant Hypoglycemic Effect in STZ-induced Diabetic Rats Blood glucose level (mmol/L) Group day 0 day 7 Change (%) Normal mice 7.92 ± 2.87 6.45 ± 1.73 -18.5 Diabetic mice 24.93 ± 4.97 23.60 ± 3.06 -5.3 AL-1(80 mg/kg) 19.53 ± 1.90 6.63 ± 2.4 -66.1 Andro (50 mg/kg) 22.15 ± 0.35 18.6 0± 0.28 -16.0 Glibenclamide (1.2 mg/kg) 26.90 ± 1.56 9.25 ± 0.64 -65.6 *Rats were given ip STZ (60 mg/kg) once. On day 3rd (designated as Day 0), drugs were given po and continuously daily for 7 days. aP<0.01 compared to solvents, and bP < 0.05 compared to day 0 (by oneway ANOVA analysis and Student’s t-test ). AL-1 Protects Pancreatic -cells Normal Diabetic islet AL-1(20mg/Kg) Andro(50mg/Kg) AL-1(80mg/Kg) Glib (1.2mg/Kg) (×400) AL-1 Protects Functions of -cells Normal islet Diabetic islet AL-1(20mg/Kg) AL-1(80mg/Kg) Andro(50mg/Kg) Glib (1.2mg/Kg) Development of AL-1: Needs >10M RMB to get an IND; 5-10M RMB to be a new drug Commercial potential: 10,00M RMB in China