* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download therapeutic range - Home - KSU Faculty Member websites
Survey
Document related concepts
Orphan drug wikipedia , lookup
Compounding wikipedia , lookup
Polysubstance dependence wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Psychedelic therapy wikipedia , lookup
Plateau principle wikipedia , lookup
Drug design wikipedia , lookup
Psychopharmacology wikipedia , lookup
Neuropharmacology wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Drug discovery wikipedia , lookup
Pharmacognosy wikipedia , lookup
Prescription drug prices in the United States wikipedia , lookup
Theralizumab wikipedia , lookup
Prescription costs wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Transcript
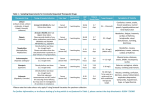
Pharmaceutical Pharmacokinetic process process Drug Therapy Pharmacodynamic Therapeutic process process Monitoring the Therapeutic Effects of Drugs :- 1-Monitoring the therapeutic event in the individual:A-Anticonvulsant drug therapy : seizure frequency Patients should be encouraged to keep a diary documenting the occurrence of seizure in order to assess the success of treatment. B-Anticholiesterase in myasthenia gravis : improvement in muscle power Patient can assess his own muscle power by simple quantitative test. In case of respiratory paralysis , test of pulmonary function e.g. vital capacity. Fatigue (Ptosis) in a patient with MG C-Drugs for angina pectoris : frequency of attacks Ability of GTN to prevent anticipated attack or shorten its duration. In assessment of βadrenoceptor blockers or calcium channel blockers , the fall in the number of GTN tablets a patient has to take may be used as a measure of efficacy. D-Diuretics in the treatment of oedema : body weight Urine volume can be used , but unreliable because of errors in recording or incontinence. Daily measurement of body weight is more reliable. E-The treatment of ulcerative colitis with glucocorticoids or 5aminosalicylic acid:Improvement in the systemic effects e.g. lessening of fever, general malaise, , increase of appetite & vigor. Reduction in the number & severity of attacks of diarrhoea, with reduction in the amount of blood, mucus & pus. 2-Monitoring the effects of drug therapy in the population:A-Immunization:-the effect of imunizing the population against poliomyelitis can be monitored by surveying the occurrence of this disease in the population. B-Currently recommended procedures in preventive medicine:By encouraging people to change their life style the rate of certain disease will fall i-Stopping smokingCHD, peripheral vascular diseases, chronic bronchitis, bronchial carcinoma. ii-Losing weightdiabetes mellitus, CHD, hypertension , osteoarthritis iii-High -fiber dietdiverticular disease, irritable bowel syndrome. vi-Reducing cholesterol & saturated fats in the diet:-CHD C-Some longterm prophylactic drug therapies widely prescribed:i-Prevention of pregnancies-oral contraceptives. ii-Secondary prevention of myocardial infarctionaspirin, βadrenoceptor blockers. iii-Hypertension & its complicationsantihypertensive drug therapy. iv-Diabetes mellitus & its complicationsdiet, insulin & oral hypoglycaemic agents. 2- Monitoring the Pharmacodynamic Effects of Drugs:- In some cases it is not possible to measure the therapeutic outcome directly. A pharmacodynamic effect, called surrogate marker, is measured. A surrogate marker is defined as an endpoint that is measured in place of the true end point & that relates in someway to the end point of primary interest. Surrogate markers are cheaper & easier to measure than true end point & can be measured more quickly A-Insulin therapy in diabetes mellitus: effect on blood glucose • Diabetes ketoacidosis is managed with low- dose continuous IV infusion of insulin & frequent monitoring of blood glucose. • Measurement of blood glucose allows alteration of the dose of insulin to maintain the blood glucose within reasonable limits to avoid hyperglycaemia or hypoglycaemia [410mmol/L] Measurement of urinary glucose concentration in patients who cannot measure their own glucose can be used to guide therapy. [aim is to reduce glycosuria to 14-28 mmol/L. Idea about how good overall control has been during past week or month can be obtained by looking at past records or by estimation of HbAIC [raised concentration suggests poor blood glucose control over the preceding 2 or 3 months] [normal<6%]. B-Anticoagulant effect with warfarin : effect on prothrombin time If the international normalized ratio [INR] is kept within accepted therapeutic limits the chance of reducing the likelihood of a pulmonary embolus in a patient with deep vein thrombosis is increased. C-Allopurinol in gout : effect on blood uric acid Allopurinol reduces serum uric acid by inhibiting the conversion of hypoxanthin to uric acid Fall of uric acid concentration is accompanied by decreased incidence of acute attacks of gouty arthritis & prevention of gouty renal disease. Neglected chronic gout with tophi D-Carbimazole in hyperthyroidism : effect on thyroid hormones Serum concentrations of T3 & TSH are measured. Therapeutic effect of carbimazole can be detected by a fall in serum T3 , over treatment will cause a fall in serum T3 below normal & an increase in serum TSH above normal. E-Bronchodilator therapy in bronchial asthma: effect on FEV1 & peak flow rate The effects of salbutamol inhalor or slow-release theophylline can be objectively assessed by measuring their effect on FEV1 or by measuring the peak flow rate recorded during maximal expiration. Assessment of salbutmol inhalation is after 10-15min, ipratropium 45-60min. F-Cancer chemotherapy: effect of tumour markers:The effectiveness of cytotoxic drug therapy with methotrexate can be monitored by measuring serum concentration of human chorionic gonadotrophin G-Lowering the blood pressure by antihypertensive drugs. H-The measurement of intraocular pressure in patients with glaucoma. Therapeutic drug monitoring is a tool that can guide the clinician to provide effective and safe drug therapy in the individual patient. Monitoring can be used to confirm a plasma concentration which is above or below the therapeutic range ,thus minimizing the time which elapses before corrective measures can be implemented in the patient Therapeutic drug monitoring is the measurement of the serum level of a drug and the coordination of this serum level with a serum therapeutic range. The serum therapeutic range is the concentration range where the drug has been shown to be efficacious without causing toxic effects in most people. Recommended therapeutic ranges can generally be found in the product inserts for drugs that require monitoring. They are also available in books such as the Physicians Desk Reference, and articles in the primary medical journals. Criteria which determine whether measuring the plasma concentration of a particular drug may be useful in practice:1-Drugs for which the relationship between dose & plasma concentration is unpredictable:-the plasma concentration that will result from a given dose in a given individual is unpredictable e.g. phenytoin. Factors which may alter the relationship between the dose & plasma concentration include:-renal & hepatic failure , diarrhoea , drug interactions & thyroid dysfunction 2-Drugs for which there is a good relationship between plasma concentration & effect:This criterion leads to the concept of ‘therapeutic range’ which is derived from plasma concentration measurements in large number of patients. Drugs with an irreversible or ‘hit & run’ actions e.g. MAOI & alkylating agents are not suitable. The development of tolerance may also restrict the use of the plasma level estimation. 3-Drugs with a low therapeutic index:measurement of plasma concentrations of such drugs will allow dosage alterations to be made in order to produce optimal therapeutic effect or to avoid toxic effects. 4-Drugs which are not metabolized to active metabolites:-e.g. procainamide is metabolized to N-acetylprocainamide which is equally potent . The extent of metabolism varies according to the acetylator status[10-40%]. Thus measuring plasma procainamide concentration alone gives imprecise guide to therapy. Drugs for which one or two enantiomers comprise a large proportion of the desired pharmacological response 5-Drugs for which there is difficulty in measuring or interpreting the clinical evidence of therapeutic or toxic effects:Nausea & vomiting occur in both digitalis toxicity & congestive heart failure. Phenytoin toxicity can occasionally cause an increase rather than a decrease in the frequency of epileptic seizures. Renal failure may occur in patients with G-negative septicaemia because of the disease or because of the adverse effects of the gentamicine used to treat it. If the relationship between the plasma concentration of a drug and its adverse effects is known, it can be used to distinguish between drug induced and disease-induced effects. 6- Drugs with steep dose response curve for which a small increase in dose can result in a marked increase in desired or undesired response e.g. theophylline. Drugs for which TDM might not be indicated include those characterized by a wide therapeutic index or those for which response can be easily monitored by clinical signs. • Pharmacokinetic factors that cause variability in plasma drug concentration are: • •drug-drug interactions • •patient disease state • •physiological states such as age, weight, sex • •drug absorption variation • •differences in the ability of a patient to metabolize and eliminate the drug Which drugs fulfill these criteria? Drugs that fulfill a sufficient number of criteria to make it worth measuring their plasma concentrations in monitoring therapy are listed in A Some drugs fulfill four of these criteria , but have not been shown conclusively to fulfill the most important criterion [ that there should be a good relationship between plasma concentration and the therapeutic or toxic effects] These drugs are listed in B. Indications for measuring plasma drug concentrations:when in practice such measurement is likely to contribute significantly to patient care? 1-Individualizing therapy:- to aim for a particular range within which therapeutic effect is likely to occur & toxic effects are likely to be minimized ,which is useful in:A-At the beginning of therapy when the relationship between dose & plasma concentration in the individual is uncertain. B-When rapid changes in renal function alter the relationship between dos & the plasma concentration , this is particulary important in case of digoxin, lithium, aminoglycoside antibiotics. C-When another drug alter the relationship between dose & plasma concentration e.g. plasma concentration of lithium is increased by thiazide. 2-In the diagnosis of suspected toxicity:-plasma concentration above the therapeutic range will undoubtly reinforce one’s suspicions of toxicity 3-Measuring compliance:- it is the extent to which the patient follows a prescribed drug regimen Extent of compliance varies from 10-92% Factors which affect compliance:1-The nature of the treatment a-The complexity of the prescribed regimen b-Adverse effects 2-Characteristics of the patient 3-The behavior of the doctor In general, TDM should not be performed until plasma drug concentration has reached steady state in the patient. Peak and trough concentrations are different from one another for drugs with a short half life compared to the dosing interval e.g. antibiotics, for such drugs , both a peak and a trough sample should be collected. Steady-state drug concentrations provide the best correlation between serum drug concentrations and clinical status Pharmacokinetic principles can be used to predict the concentration required to achieve a particular degree of therapeutic effect The target concentration strategy is a process for optimizing the dose in an individual . 1- choose the target concentration,TC 2-Predict Vd & Cl with adjustment for factors such as weight & renal function. 3-Give a loading dose or maintenance dose calculated from TC, Vd & Cl. 4-Measure the patient response & drug concentration. 5-Revise Vd and/or Cl based on the measured concentration. 6-Repeat steps 3-5 adjusting the predicted dose to achieve TC. Trough samples are generally recommended for consistency across time if a single samples are to be collected . Trough samples should be collected as close to but before a dose. Peak should be determined after drug absorption[generally 2-4hours for oral administration], for im,sc,0.5-1hour Most orally administered drugs are rapidly absorbed and reach peak serum concentrations within 1-2 hours The Effect of Sample Times on Parameter Values Patients receiving a drug at a dosing interval longer than the half-life of the drug will exhibit large fluctuations between the peak and trough levels. Shorter intervals lead to less variation Peak and trough concentrations will not differ substantially for drugs whose halflives is much longer than the dosing interval e.g. phenobarbitone, for such drugs a single sample is generally sufficient A single sample is also indicated for slow release preparation, and following a loading dose, in case of toxicity , a single peak sample It is important to avoid withdrawing blood until absorption is complete [2h after an oral dose] Some drugs take several hours to distribute into tissues e.g. digoxin [6h after last dose] & lithium[24h after last dose] When steady state has been reached , a sample obtained near the mid point of the dosing interval will usually be close to the mean steady concentration. SAMPLING TIMES AND TARGET RANGES FOR TDM ASSAYS .TABLE 1 DRUG MINIMUM PLASMA/SERUM VOLUME (mL ) Amitriptyline (+ nortriptyline) APPROXIMATE TIME TO STEADY STATE (DAYS) RECOMMENDED SAMPLING TIME TARGET RANGE 10-7 pre-dose 250-100µg/L Caffeine 0.2 2 2-1h post-dose 36-12mg/L Carbamazepine 0.5 4-2 pre-dose 12-4mg/L Clomipramine (+norclomipramine) 2.0 42-28 pre-dose 800-150µg/L Desipramine 2.0 14-10 pre-dose 250-100µg/L Digoxin 0.5 7-5 24-6h post-dose 2.0-0.8µg/L Dothiepin (+ nordothiepin) 2.0 7 pre-dose 300-100µg/L Ethosuximide 0.5 14-7 pre-dose* 100-40mg/L Fluoxetine (+ norfluoxetine ) 2.0 42-28 pre-dose 800-150µg/L Gabapentin 0.5 2-1 pre-dose 20-2mg/L Imipramine (+ desipramine) 2.0 7 pre-dose 300-150µg/L Lamotrigine 0.5 6-4 pre-dose 4-1mg/L Nortriptyline 2.0 14-10 pre-dose 150-50µg/L Phenobarbitone 0.5 20-10 pre-dose* 40-15mg/L Phenytoin 0.5 35-7 pre-dose* 20-10mg/L Theophylline - adults (1 ) Theophylline - neonates (2 ) 0.5 0.5 2 2 4-2h post-dose 4-2h post-dose 20-10mg/L 10-5mg/L Valproic acid (3 ) 0.5 3-2 pre-dose up to 100 mg/L The process of therapeutic monitoring takes effort. First pharmacist and physician develop initial dosing recommendations. Involvement of clinical pharmacist in therapeutic drug monitoring & interpretation. The management of therapy using plasma concentration • • • • • • Information required Time of sample in relation to last dose Duration of treatment with the current dose Dosing schedule Patient - Age, weight, sex, height, smoker Clinical - Drug requirements, clinical status (renal serum creatinine; cardiac - cardiac output, liver, etc • Other drug therapy • Relevant disease states • Reason for request e.g. lack of effect, routine monitoring, suspected toxicity Calculate initial loading dose or maintenance regimen and make recommendations • Then organize sample collection and analysis . Accurate Timing in necessary • Second, someone (nurse, med tech) must take the blood. • Someone (lab tech, you) must assay the drug concentration in the blood. • Evaluate pharmacokinetically the analytical result and recalculate dosing regimen recommendations Organize further samples if necessary, repeat as necessary • You must communicate your interpretation and your recommendations for dosage regimen change to the physician. This will allow for informed dosage decisions. • You must follow through to ensure proper changes have been made. • You must continue the process throughout therapy. Therapeutic drug monitoring, in many cases, will be part of your practice. It can be very rewarding. A retrospective survey carried out at the Massachusetts General Hospital showed that whilst prior to the use of digoxin monitoring 13.9% of all patients receiving this drug showed evidence of intoxication , following introduction of monitoring this fell to 5.9%. A significant difference with regard to length of stay in the hospital between patients on gentamicin who were monitored and their dosage regulated consequently versus those who were not (DeStache, 1990) Effect of artifact on TDM results Artifact Sequela Drug All drugs Serum separator tubes Aminogl Glass tubes ycosides Digitalis Red stoppers Silicon gel can bind drug,decreasing concentration Glass bind drug, decreasing concentration Stoppers may bind drug, decreasing concentration Identification and quantization performed on an emergency basis of toxic substances in patients after toxic episode Why Measure Drug Levels? Indications of “Drug screen” 1-It is carried out for drugs or toxins for which plasma concentration data may influence treatment. e.g. phenobarbitone, ethanol, ethylene glycol, isopropyl alcohol, paraquat, salicylates, thiophylline, Why Measure Drug Levels? 2-Multiple drug ingestion 3-History unreliable [Patient may try to hide drug poisoning by withholding or altering information.] 4-Patient in delirium or coma of unclear aetiology. 5-Determine drug abuse Objectives:1-Identification of specific drug 2-To indicate when antagonists to be used 3-Aid in the estimation of prognosis Assay Techniques Spectrophotometric Immunoassay Chromatographic Awareness of relative specifity, sensitivity & precision of these methods. Difficulties & limitations 1-Some compounds may be active at low concentration which are not detectable by available techniques. 2-Some compounds are heat labile and break apart before detection e.g LSD, psilocybin 3-Some compounds are distributed to compartments other than the tested compartment e.g. LSD, pesticides 4-Some evaporate in extraction or concentration e.g. solvents Sampling:1-Blood:- Blood– Specimen most often used for medicolegal cases Useful in case of paracetamol, salicylates, carboxyhaemoglobin, methanol, ethylene glycol • – Disadvantages: invasive, drugs only detected for short time, analysis of blood is labor intensive and relatively expensive Advantage: when individual suspected of being intoxicated on alcohol (exact concentration determined and related to levels determined to cause impairment) BAC(mg/L) Clinical effect 50-100 Sedation,↑reaction times 100-200 Impaired motor function, slurred speech, ataxia 200-300 Emesis, stupor 300-400 coma >500 Respiratory depression, death 2-Urine:performed when the blood concentration of the compound is too low for detection by conventional measures , rapidly eliminated or have high volume of distribution e.g. phenothiazines, benzodiazepines, antihistamines, TCA • • Advantages: most widely used, readily available, non-invasive, drugs/metabolites tend to concentrate in urine, easily handled and analyzed, little cost • • Disadvantages: drugs/metabolites only indicate past use, subject to adulteration or substitution, short detection times (12 hours to 2-3 weeks—2-5 days for most drugs • In an attempt to avoid detection of illicit drug use, some people who are asked to give a urine sample for drug screening will attempt to substitute the urine of another person or adulterate their urine specimens, using either commercially available adulterants or home-brew recipes. 3-Hair:-cocaine, opiates, once incorporated into hair , drugs remain there indefinitely (previous 3 months), due to absence of metabolism in hair and uniform growth rate(1 cm/month) Strand of hair cut into sections gives information in the pattern of drug use • – Advantages:Longer detection times (weeks and months), impossible to adulterate or substitute, easily collected • – Disadvantages: much higher cost, very recent or infrequent drug use likely not detected, alcohol use cannot be detected, not all donors have long enough hair, exposed to environment (passive contamination) • – Research is showing that darkly pigmented hair more readily incorporates and retains drugs than does light pigmented hair. 4-Sweat :-detects cocaine, heroin 5- Saliva:-Marijuana , cocaine , opiates • – Rarely used • – FDA has approved a saliva test for monitoring alcohol use 6-Breath:– Not as good as blood, but useful when ethanol abuse suspected. – Advantages:readily available, noninvasive. – Disadvantage:must have a calibrated breath alcohol testing device with qualified personnel to perform test • Results Interpretation • Time of sample collection, time of administration of toxin,time of appearance of toxic effects or death should be interpreted in relation to pharmacokinetic parameters of toxin absorption, distribution, metabolism and elimination. When did the donor use this substance? Specimen dependent Urine: drugs detectable for about 2-5 days Exceptions: cannabinoids, long-acting barbituarates, benzodiazepines Cannabinoid: light user=10 days; chronic user=month or more Benzodiazepines: 15 days or more Did donor use an illicit substance or can the result be innocently explained? passive exposure, available by prescription How much of the drug did the donor use? – Nearly impossible to answer when urine is the specimen • Differing levels of hydration – Not enough known about hair yet – Blood: estimate of drug can calculated by employing published pharmacokinetic data Is the donor continuing to use the substance? – The shorter the detection time for a substance, the easier the answer to this question becomes. – If the donor’s urine is retested seven to 10 days after initial test and the analyte is a substance with a short detection time, a second positive test can be presumed to mean that the donor is continuing to use the drug. Two components make up the process of therapeutic drug monitoring:•Assays for determination of the drug concentration in plasma. •Interpretation and application of the resulting concentration data to develop a safe and effective drug regimen.