* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Methylprednisolone
Survey
Document related concepts
Compounding wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Drug discovery wikipedia , lookup
Drug design wikipedia , lookup
Prescription drug prices in the United States wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Pharmacognosy wikipedia , lookup
Dextropropoxyphene wikipedia , lookup
Prescription costs wikipedia , lookup
Dydrogesterone wikipedia , lookup
Neuropharmacology wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Psychopharmacology wikipedia , lookup
Theralizumab wikipedia , lookup
Transcript
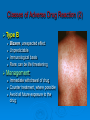
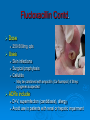
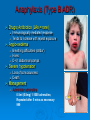
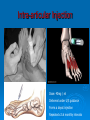
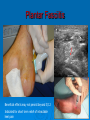
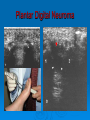
Prescription-Only Medicines now Accessible to Podiatrists The Science Behind Them Dr Jean Mooney PhD, FChS, FCPodS, FCPodMed, FHEA Pods and POMs: History ~1980 4 injectable plain local anaesthetic solutions ~1996 Statutory Instrument gave access to Further SI gave access to 2 adrenalinised local anaesthetics Topical anti-fungal agents Topical 1% corticosteroid 3-day course Ibuprofen, 200mg tds Patient Group Directions Legal framework (August 2000) Allowed Podiatrists to supply and administer specified medicines to patients who are designated as within a group as defined by the PGD e.g: Diabetics with soft tissue or bone infections POMs supplied directly to a patient without the need for a separate prescription from a prescriber. PGD allows access to POMs for specific types of patient presenting with a specific need: it is NOT a form of prescribing PGD does not require the podiatrist to have any additional qualification Employing organisation must ensure that only fully competent, trained health care professionals use PGDs. Supplementary Prescribing (2005) Voluntary prescribing partnership between IP and SP Implements an agreed patient-specific clinical management plan (CMP) CMP agreed between Podiatrist must undergo training (~6/12) to become SPs IP: doctor SP: podiatrist Patient HPC-Register annotated Allows them to prescribe or adapt dosage of POMs specified within the CMP without recourse back to IP Pods and POMs: 17.11.2006 SI extended access to the list of POMs that can be administered, sold and supplied to patients by Podiatrists Schedule 5, Articles 4(2) and 4(4) amended Part I and III of Schedule 5 to the Prescription Only Medicines (Human Use) Order 1997 Also regularised access to some Pharmacy medicines (P) for topical application e.g.: 1% Griseofulvin 1% Terbinafine 2006 Additional POMs from 17.11.06 Adrenaline 2 more plain LA solutions 4 Anti-microbial agents Levobupivacaine Ropivacaine Amoxicillin Erythromycin Flucloxacillin Silver Sulfadiazine Anti-inflammatory Methylprednisolone Further amendments allowed additional access to other medicines from 10.03.2011 Ibuprofen (400mg) Codeine phosphate Co-codamol Co-dydramol (10/500) Pre-mix injectable solutions E.g.: Depomedrone (Pre-mixed Lidocaine and Methylprednisolone) Recommendations Continue to use all means of access to POMs e.g.: PGDs Train Not easy for those in private practice Gain as a Supplementary Prescriber HPC annotation POMs LA College of Podiatrists Recommendations Codeine, Co-codamol and Co-dydramol Indicated for short term treatment of acute / moderate pain unrelieved by paracetamol, ibuprofen or aspirin Limited to a maximum of 3 days prior to direct patient review even though the pack size may exceed that dose level Essential that all Medicines are correctly labelled and supplied with an explanatory leaflet that clearly states Dosage Side effects (e.g.: constipation) Possibility of addiction or habituation Pharmacodynamics Adverse Drug Reactions Pharmacodynamics The effects of the drug on the body desired and undesired effects of the drug on body systems Intended effects Modes of action / drug-receptor interaction Doses and maximum safe doses Undesired / unwanted / unexpected effects = Adverse drug reactions (ADRs) Classes of Adverse Drug Reaction (1) Type A: Addative effects Dose related Predictable effect Not usually severe Management: Dose modification Classes of Adverse Drug Reaction (2) Type B Bizarre, unexpected effect Unpredictable Immunological basis Rare: can be life threatening Management: Immediate withdrawal of drug Counter treatment, where possible Avoid all future exposure to the drug Comparison: Type A and Type B ADRS Other Types of Adverse Drug Reaction Type C Type D Delayed effects Drug does not ‘kick in’ when expected Type E Chronic administration Habituation; tolerance; dependence Exclusion Effects of drug withdrawal Type F Failure of therapy Often due to drug interactions Frequency of ADR, by Class of Drug ADR-Risk Patients Elderly Very young Renal disease Liver disease Genetic predisposition ADRs attributed to Celecoxib (COX2 inhibitor) in 6/12 period ADR avoidance Use prescribed medications only when necessary Patients should be warned of the possibility of ADR occurring In the lowest dose, to achieve required effect For the shortest time, to maintain the required effect Package advice leaflet BNF Yellow card system Report all suspected ADRs Pharmacokinetics Drug Interactions Pharmacokinetics The effects of the body on the drug How the body deals with the drug What the body does to the drug • Absorption • Metabolism • Excretion Drug interactions Drug Interactions (DIs) Effect or action occurring in the body Due to taking two or more drugs, or one drug+ Beneficial / desired Adverse / unwanted OTC medicines Vitamin and mineral supplements Medicinal herbs Foods Does not occur when taking either alone DIs usually inadvertent, e.g.: POM + OTC Drugs supplied by more than one prescriber The more drugs that are taken, the more likely that DI will occur Often under-reported as they mimic exaggerated action of drug Drug Interactions Desired effects: potentiating effect of drugs used in combination Undesired effects: one drug mitigates the effect of another absorption by small intestine metabolism to non-active substances by liver (non-active substances excreted via the kidney: may still carry some drug effect) DIs also occur when drug constituents interact with e.g.: foods, antacids, vitamin, mineral or herbal supplements e.g.: Erythromycin reduces the effectiveness of oral contraceptives Most DIs arise due to effects on Cytochrome P450 enzyme system e.g.: codeine combined with paracetamol gives greater pain relief e.g.: Antacids can bind with antibiotics preventing blood uptake It is essential that the name and dosage of all medications (including OTCs) are identified before supplying a POM to a patient Full medical history Antibiotics Antibiotics (ABx) Substances that kill or inhibit a range of MOs Dose: usually minimum 5-7 days Any MO-derived substance that antagonizes growth of another MO in high dilution to ensure full MO kill wound swab BEFORE starting ABx review patient after 3 days to check response to AB treatment Classified By manufacture • natural, semi-synthetic or synthetic analogues of natural compounds Spectrum of biological effect • Bactericidal / Bacteriostatic Susceptibility of a range of MOs to ABx effect • Broad / Medium / Narrow spectrum Amoxicillin Flucloxacillin Erythromycin Silver Sulfadiazine Available to HPC POM-annotated Podiatrists since Nov 2006 Amoxicillin Beta-lactam penicillin-type antibiotic with moderatespectrum of activity Bacteriolytic Inhibits synthesis of G+ve and G-ve bacterial cell walls Good absorption with oral administration MO resistance is common MOs produce beta-lactamase and degrade amoxicillin Often formulated in combination with clavulanic acid (Coamoxiclav / Augmentin) to overcome MO resistance Amoxicillin Contd: Dose: Uses 250mg / 500mg tds Skin infections (No longer recommended for prevention of bacterial endocarditis) Side effects (ADRs) D+V Non allergic rashes • Affects 3-10% of children Anaphylaxis Flucloxacillin Beta-lactam penicillin-type antibiotic with narrow spectrum of activity Inhibits synthesis of bacterial cell walls Used to treat infections caused by susceptible G+ve bacteria Active against beta-lactamase MOs, such as Staph aureus Not effective against G-ve organisms or non-beta lactamase producing G+ves Ineffective against MRSA MO Resistance Flucloxacillin Contd. Dose 250-500mg qds Uses Skin infections Surgical prophylaxis Cellulitis • May be combined with ampicillin (Co-fluampicil) if Strep pyogenes suspected ADRs include D+V, superinfection (candidiasis), allergy Avoid use in patients with renal or hepatic impairment Erythromycin Bactericidal macrolide antibiotic Slightly wider antimicrobial spectrum than penicillins Often used in subjects with penicillin allergy Unknown mechanism of activity Taken up by macrophages so concentrates in area of infection Indicated for skin infections Metabolised in the liver Erythromycin Contd. Dose ADRS include D+V, nausea and abdo cramps Cardiac arrhythmias and deafness Allergies 250mg qds Non acid-stable (give after meals) Clarythromycin is acid-stable To be avoided in infancy, pregnancy and lactation Not used in conjunction with many drugs e.g.: Warfarin, OCs, corticosteroids, simvastatin, antimigraine drugs, verapamil, terfenadine, theophilline, clindamycin Silver Sulfadiazine Topical agent Antibacterial: broad-spectrum activity in chronic wounds G+ve and G-ve bacteria (including Pseudomonas aeruginosa) Some yeasts and fungi Poor penetration on normal skin Up to 1% show hypersensitivity reaction, e.g.: 1% cream Sulfonamide and Silver Rashes; erythema multiforme Skin discolouration (argyria) Avoid in late pregnancy / infancy Avoid in patients with G6PD deficiency May increase wound healing times Not recommended by Cochrane review LET’S See you again in 15 mins Pain control Analgesia Anaesthesia Analgesics Analgesic = painkiller Act at PNS and / or CNS membrane receptors Include Paracetamol (acetaminophen in US), NSAIDs, e.g.: Salicylates (aspirin), Ibuprofen Opioids, including Morphine and Codeine CoP advice: an = without; algos = pain NB: Anaesthetics = without sensation Max administration = 3 days, then direct patient review Analgesic choice is determined by Severity of pain Pain type, e.g.: neuropathic pain is more responsive to tricyclic antidepressants and anticonvulsants (e.g.: gaba-pentin) Codeine phosphate Opiate drug Actions Weak to mid-range opioid Makes up 3% of opium CSN and PNS action Analgesic, anti-tussive, anti-diarrhoeal Side effects (especially in overdose) Gut immobility Respiratory suppression Tolerance, habituation, addiction, coma, death Codeine is metabolised to morphine • 5% show rapid metabolism to morphine ‘High’ • Avoid use during lactation Codeine contd. Unwanted side effects include Euphoria, itching, nausea, vomiting, drowsiness, orthostatic hypotension, urinary retention, depression, constipation, and paradoxical coughing Hives and rashes due to allergic reaction Long-term administration causes erectile dysfunction and hypogonadism (especially in white males) Sugar cravings • Induces hypoglycaemia (the ‘munchies’) • Was once used to control diabetes, as was morphine Co-dydramol Compound analgesic Dihydro-codeine tartrate 7.5 / 10 / 20 / 30mg + Paracetamol 500mg Used to relieve moderate pain Side effects Allergic reactions - urticaria, breathing difficulty, increased sweating, facial flushing, mouth ulcers. Abdominal pain GIT upsets: abdominal pain, nausea, heartburn, constipation, loss of appetite, dry mouth, Blood problems - anaemia, nose bleeds, increased risk of infection, bruising. Co-dydramol Side Effects Contd UT upsets - pain or difficulty in passing urine. Nervous system - confusion, drowsiness, dizziness, mood changes, depression, hallucinations, restlessness, excitation, fits, painful eyes, headache, sleeping problems, Tolerance or dependence. Eyes - blurred or double vision, extremely small pupils. Other - trembling, tiredness. weakness, malaise, low body temperature, muscle stiffness, changes in libido. Co-Codamol Compound Codeine phosphate 8 / 12.8 / 15 / 30mg + Paracetamol 500 / 1000mg For analgesic the relief of mild – moderate pain, where paracetamol alone, or NSAIDS (aspirin, ibuprofen, naproxen) does not control the pain Co-codamol Contd. Side effects include Allergic reactions: Shortness of breath Hypersensitivity, pruritis, Rashes, CNS effects: Confusion, Loss of short term memory, Dizziness, Fainting, Drowsiness, Sedation, Euphoria, dysphoria, addiction. Blood changes: bleeding gums, easy bruising GIT effects: Abdominal pain, Nausea / vomiting, Constipation Others: Dry mouth; Paracetamol (Acetaminophen) OTC analgesic and antipyretic Relief of minor aches and pains • COX2 inhibitor COX + arachidonic acid prostaglandin • Reduces Prostaglandin E2 lowers temperature • Modulates endogenous canabinoid system pain awareness reduced • Inhibits sodium channels in pain fibres Constituent of many cold and ‘flu relief remedies Does not cause gastric irritation Does not have marked anti-platelet effect Used in combination with opioid analgesics to control more severe pain, e.g.: post surgery Paracetamol contd. Onset of analgesia is approximately 11 minutes after oral administration Half-life = 1–4 hours. Metabolised by liver Recommend dose = 1g tds 3g daily • 2g daily maximum for heavy drinkers • 325mg tds in USA Acute overdose causes potentially fatal liver damage • First aid = activated charcoal • Paracetamol toxicity is foremost cause acute liver failure • Rare individuals develop irreversible liver damage at normal dose Risk of overdose increased by alcohol consumption Local Anaesthetics Lidocaine hydrochloride (Xylocaine) Bupivacaine hydrochloride (Marcain) Lidocaine hydrochloride + 1:200,000 adrenaline Bupivacaine hydrochloride + 1:200,000 adrenaline Mepivacaine (Scandonest) Prilocaine (Citanest) Levo-Bupivacaine (Chirocaine) Ropivacaine (Naropin) LAs prevent generation of nerve impulses (action potentials) in pain fibres Injected LA diffuses into nerve fibre LA molecule blocks Na+channels in nerve fibre membrane Nerve impulse cannot be generated LA gradually diffuses out of the nerve fibre Nerve function returns to normal Impulse can be generated and propagated LA taken up from site of injection into general circulation LA metabolised in liver and excreted via kidney Onset of Action Lidocaine hydrochloride = 5 mins Bupivacaine hydrochloride = 20 mins Mepivacaine = 10 mins Prilocaine = 10 mins Levo-Bupivacaine = 20 mins Ropivacaine = 10-30 mins Duration of Action Lidocaine = 1-2 hours Bupivacaine = 6-8 hours Bupivacaine + 1:200,000 adrenaline = 12-16 hours Mepivacaine = 2-4 hours Prilocaine = 2-4 hours Levo-Bupivacaine = 5-15 hours Lidocaine + 1:200,000 adrenaline = 2-4 hours Up to 30 hours post-op analgesia Ropivacaine = 4-8 hours Up to 24 hours post-op analgesia Cautions Do not inject adrenalinised solutions into the distal foot Causes ischaemia ‘Chemical tourniquet’ Ischaemic effect persists for duration of anaesthesia Avoid adrenalinised solutions in patients taking Beta-blockers MAOIs Tri-cyclic anti-depressants Calculation (in mg) of total LA dose administered from drug labelled as % solution Percentage Mass 1% solution = 10mg of drug in 1ml 2% solution = 20 mg of drug in 1ml 3% solution = 30mg of drug in 1ml THUS 3.5ml of 1% soln delivers 35mg of drug 8.3ml of 2% soln delivers 166mg of drug 5.6ml of 3% soln delivers 168mg of drug Maximum safe doses 70Kg or >70Kg person Lidocaine Bupivacaine / Levobupivacaine 150mg (2mg / Kg) 30ml of 0.5% OR 60ml of 0.25% soln Mepivacaine OR Prilocaine 200mg (3mg / Kg) 20ml of 1% OR 10ml of 2% soln 400mg (6mg /Kg) 13ml of 3% soln Ropivacaine ~250mg (4mg/Kg) 50ml of 0.5% OR 33ml of 0.75% soln Maximum Safe Dose for patient <70kg MSD of 1% Lidocaine for 68kg person? MSD of 3% Mepivacaine for 61kg person? MSD for 70kg = 200mg MSD for 68kg in mg = 68/70*200 = 195mg MSD of 1% Lidocaine soln for 68kg, in ml = 195/10 = 19.5ml MSD for 70kg = 400mg MSD for 61kg in mg = 61/70*400 = 349mg MSD of 3% Mepivacaine soln for 61kg, in ml = 349/30 = 11.6ml MSD of 0.5% Bupivacaine for 58kg person? MSD for 70kg = 150mg MSD for 58kg in mg = 58/70*150 = 120mg MSD of 0.5% Bupivacaine soln for 58kg, in ml = 120/5 = 24.25ml ADRs of LA Toxicity (Type A ADR) High plasma concentration • Actual overdose • Relative overdose Faint Vasovagal attack • Psychosomatic effect Hypersensitivity reactions Rare with amide-type LAs Toxic Effect of LAs CNS effects Inebriation, Lightheaded-ness, Drowsiness Numbness of tongue / peri-oral tissues, Paraestheiae Restlessness, Nausea + vomiting, Blurred vision Muscle twitching, Tremors, Convulsions Respiratory failure, Coma Cardiovascular effects Myocardial depression Peripheral vasodilatation Hypotension and Bradycardia Arrhythmias and Cardiac arrest Be cautious in using LAs on these patients Children, elderly or debilitated patients Impaired cardiac conduction Cardiovascular disease Hypovolaemia Shock Impaired respiratory function Epilepsy Myaesthenia gravis Contra-Indications to LA Inflamed / infected tissues Reduced anaesthetic effect Increased rate of absorption predisposes to toxicity Patients with heart block Adrenalinised LA solutions Never into a digit • Risk of ischaemic necrosis Not with severe hypotension Not with unstable cardiac rhythm (e.g.: uncontrolled AF) Not with MAOIs and tricyclic antidepressants Drug Interactions and Local Anaesthetics Lidocaine + Cimetidine Lidocaine / Bupivacaine / Levo-bupivacaine / Prilocaine / Ropivacaine + Propanolol / Amiodarone Increased plasma concentration of lidocaine Lidocaine + Loop and Thiazide Diuretics Increased myocardial depression Lidocaine + antivirals Lidocaine metabolism reduced / plasma concentration increased Lidocaine effectiveness reduced Mepivacaine + opioid sedatives Increased risk of LA toxicity Drug Interactions and Local Anaesthetics, Contd Lidocaine + bupivacaine Prilocaine + dapsone Methaemoglobinaemia Ropivacaine + Fluvoxamine (Anti-depressant) Increased risk of LA toxicity Total dose should not exceed combined MSDs Ropivaciane metabolism inhibited Levo-bupivacaine + TCAs or MAOIs Increased risk of LA toxicity Anaphylaxis (Type B ADR) Drugs: Antibiotics (LAs = rare) Angio-oedema Breathing difficulties (stridor) Hives D+V; abdominal cramps Severe hypotension Immunologically mediated response Tends to increase with repeat exposure Loss of consciousness Death Management Administer adrenaline • 0.5ml (0.5mg) 1:1000 adrenaline, • Repeated after 5 mins as necessary • 999 Anti-Inflammatory Agents NSAIDs Corticosteroids Ibuprofen Iso-butyl-propanoic-phenolic acid OTC Non-steroidal anti-inflammatory agent (NSAID) Common adverse side effects include: Used to control pain that has an inflammatory component Mild, short-lasting anti-platelet effect (cf aspirin) Vasodilatory action GIT: Nausea, Indigestion, GIT ulceration/bleeding, Raised liver enzymes, Diarrhoea, Constipation, Cardiovascular effects: Epistaxis, Hypertension, Increased risk of myocardial infarction, Priapism Neurological: Dizziness, Hearing loss, Tinnitus Others: Skin rashes, Fluid retention, Spontaneous abortion All SEs minimised by low-dose administration Ibuprofen Contd. Action: Non-selective inhibition of • COX-2 (prevents degradation of arachidonic acid to prostaglandin) • COX-1 (prevents platelet aggregation) Off label • Treatment of acne • Prophylaxis of Alzheimer's disease and Parkinson’s diseases (low dose, long term) Dose-dependent duration of action (4-8 hrs) Self-medication: Max 1200mg (400mg tds) daily Prescribed: Max 3200mg (800mg qds) daily Stable in solution: topical gel Corticosteroids Anti-inflammatory effects of corticosteroid Modifies gene transcription ‘Switches off’ pro-inflammatory genes OR: ‘Switches on’ anti-inflammatory genes Reduces formation of pro-inflammatory mediator chemicals, e.g.: cytokines Local pain reduction Reduction of local swelling Reduction of local erythema and tissue irritation Anti-inflammatory Effects of Glucocorticoid ‘Dermatitis’ and Skin Inflammation Topical application 1% hydrocortisone acetate cream, e.g. HC45 Daktacort Standardized unit of application = fingertip unit FTU. One FTU = amount of topical steroid squeezed from the tip of the index finger to dipj One FTU will treat an area of skin twice the size of an adult's hand. Methylprednisolone acetate Synthetic corticosteroid Pharmacological effects by topical, inhaled, injected, or systemic delivery Glucocorticoid action Reduces normal cellular wall adhesion Reduces normal collagen production Hypertensive Immunosuppressive Diabetogenic Anti-inflammatory Intra-articular Injection Dose: 40mg / ml Delivered under U/S guidance Forms a depot injection Repeated x3 at monthly intervals Plantar Fasciitis P Beneficial effects may not persist beyond 3/12 Indicated for short term relief of intractable heel pain Plantar Digital Neuroma CS Drug Interactions 1 Systemic effects of corticosteroids are increased (or their hepatic metabolism is reduced) when administered with Erythromycin Clarithromycin Ketoconazole (Nizoral Oestrogens, including OCs and HRT Lower doses of corticosteroids may be indicated in these cases The doses of both methylprednisolone and cyclosporin may need to be reduced to if they are administered concurrently, to avoid increased side effects of either drug Cyclosporin reduces the hepatic metabolism of methylprednisolone Methylprednisolone reduces the metabolism of cyclosporin CS Drug Interactions 2 Increase or decreases the effect of warfarin Phenobarbital, Phenytoin and Rifampicin may increase corticosteroid metabolism, reducing corticosteroid effects. Anti-coagulated patients on corticosteroids should be monitored and therapy adjusted to achieve the appropriate levels of anti-coagulation Dose of methylprednisolone may need to be increased The effects of CS in pregnancy and lactation have not been fully evaluated Systemic side effects of corticosteroid therapy 1 Vary from mild temporary to severe and permanent body wide effects: Fluid retention, weight gain and central obesity Hypertension Potassium depletion Headache Muscle weakness Facial puffiness (moon face) Hirsuites Thinning of the skin Glaucoma Cataracts Incidence or exacerbation of diabetes Irregular menses Growth retardation in children Convulsions Systemic side effects of corticosteroid therapy 2 Psychic disturbances (depression, euphoria, mood swings, psychoses) Suppression of adrenal cortex activity, causing Addisonian crisis if the corticosteroid therapy is stopped abruptly Masked signs of infection Impaired immune response to infection Increased susceptibility to infection Exacerbations of viral infections Development of e.g.: small pox if live vaccines administered Reactivation of dormant TB and malaria Loss of vaccine-induced immunity False negative results from the TB (Heaf) test Impaired calcium absorption causing osteoporosis and fractures Aseptic necrosis of joints Adrenaline Epinephrine (Adrenaline) Hormone secreted by the adrenal medulla Sympathetic neurotransmitter Catecholamine Nonselective agonist of all (α1, α2, β1, β2, and β3) adrenergic receptors Participates in the fight or flight response Increases blood glucose levels • α-adrenergic receptors: inhibits pancreatic insulin secretion, stimulates pancreatic gluconeogenisis, and skeletal glcyolyisis • β-adrenergic receptors: triggers pancreatic glucagon secretion. increases pituitary ACTH secretion, and increases adipose lypolysis Increases heart rate Constricts blood vessels and increases blood pressure Dilates bronchi and bronchioles Increases skeletal muscle contraction Epinephrine (Adrenaline) Contd. Adverse reactions to adrenaline include Drug of choice for treatment of anaphylaxis Palpitations, tachycardia, arrhythmia, anxiety, headache, tremor, hypertension and acute pulmonary oedema Contraindicated in people on non-selective betablockers Administered as 1:1000 dilution 0.5mg / ml IM, repeated if necessary 5mins later Vasoconstrictor action in LA Pre-mix 1:200,000 dilution Reduced dose / prolonged action CI for LA is distal part of the foot CI for patients on MAOIs And we are looking forward to Independent Prescribing! Thank you for your kind attention [email protected]