* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Local anaesthetics
Pharmaceutical industry wikipedia , lookup
Drug discovery wikipedia , lookup
Pharmacognosy wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Toxicodynamics wikipedia , lookup
Prescription costs wikipedia , lookup
Discovery and development of proton pump inhibitors wikipedia , lookup
Drug design wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Drug interaction wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
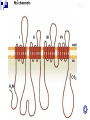
LOCAL ANAESTHETICS DR.SUDHIR MUBARAK AL KABEER HOSPITAL DEFINITION They are defined as drugs that can produce reversible inhibition of excitation & conduction in perpipheral nerve fibres & nerve endings & thus produce a loss of sensation in a circumscribed area of the body HISTORY Captain James cook – puffer fishtetrodotoxin – 18 th century 1884 – Cocaine – Freud & Koller – used first for corneal anaesthesia 1905 – Procaine – Einhorn – first synthetic local anaesthetic 1943- Lidocaine – Lofgren Chemical Structure Physical Properties (structure) Ester: R3 R 1—COO—R 2 —N R 1 — Lipophilic aromatic residue. R4 Amide: R3 R 1—NHCO—R 2—N R 2 — Aliphatic intermediate connector. R 3, R 4 — Alkyl groups or Amino groups R4 Example: C 2H 5 H2 N— —COO—(CH 2) 2—N C 2H 5 Changes to any part of the molecule lead to alterations in activity & toxicity Increases in the length of the intermediate alcohol group, up to a critical length, result in greater anaesthetic potency Beyond this critical length, increased toxicity results Compounds with an ethyl ester, such as procaine, exhibit the least toxicity The length of the two terminal groups on the tertiary amino-N group are similarly important The addition of a butyl group to mepivacaine results in bupivacaine, which differs by, a. increased lipid solubility & protein binding b. greater potency c. a longer duration of action ESTERS COCAINE PROCAINE CHLORPORCAINE TETRACAINE (AMETHOCAINE) BENZOCAINE AMIDES LIDOCAINE PRILOCAINE MEPIVACAINE BUPIVACAINE ROPIVACAINE ETIDOCAINE CINCHOCAINE (DIBUCAINE) NEURONAL TRANSMISSION SODIUM & POTASSIUM CHANNELS Voltage sensitive sodium channel It sorrounds aqueous pore Three subunits- α β1 β2 α unit –largest- MW 260 kDa It’s a single long peptide containing four hydrophobic regions – I,II,III,IV They are connected to each other by intracellular bridges Each region concists of six membrane spanning segments( S1- S6) S4 segment is the voltage sensor The intracellular bridge between III & IV is the Inactivation gate This gate is responsible for occluding Na+ channel & hence inactivation. Potassium channel is similar but with 4 subunits- 14 types of k+ channels are present Acid base considerations related to mode of action Rules Acid drugs – become more NON ionized in acidic pH Basic drugs – become more NON ionized in basic pH (alkaline pH) Acid Drug Basic Drug Acid pH Environment NON-ionized IONIZED Basic pH Environment IONIZED NON-ionized Rules Hydrophilic = Ionized molecules (charged) Lipophilic = Non-ionized molecules (non charged) Lipophilic molecules penetrate membranes Hydrophilic molecules dissolve in water Depending if a molecule is ionized or not determines if it can pass through a cell membrane MEMBRANE Non-Ionized Molecule CELL Ionized Molecule Basic Drug Basic drugs – become more NON ionized in basic pH I pH 2 N I pH 6 N I pH 8 What’s the pKa for this drug? N I pH 9 base pH = pKa + log acid = Ionized molecules = Non ionized molecules MODE OF ACTION Most LA are tertiary amine Bases(B) which are administered as water soluble hydrochlorides(B.Hcl). After injection ,the base is liberated by Alkaline pH of the ECF B.HCL + HCO3 - = B + H2CO3 + CLSo the LA is present in both ionised (BH+) & non –ionised forms(B) in tissues The proportion of each depends on Pka of drug and the ph medium into which it is adimistered The non ionised base( B) only can diffuse through the nerve sheath,perinural tissues & neurilemma to reach neuroplasm In the neuroplasm it gets again ionised by H+ ions B + H + = BH+ This BH+ the sodium channel from INSIDE They are believed to interact with phenyalanine & tyrosine residues in the S6 segment of region IV Thus they block Na+ channels and prevent depolarisation ( Phase 0) Benzocaine - Membrane expansion- causing swelling of lipoprotein matrix of Na+ channel . Tetrodotoxin & saxitoxin- directly block Na+ channel from the exterior of the membrane,close to the external pore PHYSIOCHEMICAL PROPERTIES LIPID SOLUBILITY TISSUE PROTEIN BINDING pKa STEREO SPECIFICITY VOSODILATATION MINIMUM CONCENTRATION FREQUENCY DEPENDANT BLOCKADE DIFFERENTIAL BLOCKADE LIPID SOLUBILITY There is a close relation between lipid solubility and potency Bupivicaine is approx 4 times more potent than lidocaine because of lipid solubility Etidocaine(5000) > Bupivacine(4000)> Ropivacaine>tetracine> lidocaine(150)> prilocaine=mepivacine> procaine (1) Tissue protein binding It primarily affects the Duration of LA Etidocaine(96%) > Bupivacine(95%)> Ropivacaine(94%)>mepivacine>tetracine> lidocaine(65%)> prilocaine> procaine (6%) pKa Value- disscociation constant This is the most important factor determining the rapidity and onset of action Low pKa – more non-ionised- rapid onset Procaine (8.9)> Tetracaine(8.5) > Bupivacine(8.1) = Ropivacaine> lidocaine = prilocaine = etidocaine(7.7)> Mepivacaine(7.6) High pKa value – better differential blockade Onset time & Duration Agent Onset (mins) Duration Effect of Adreanaline Bupivacaine 20-30 8-9 hrs X1 Chlorprocaine Fast in 3% 30-60 mins Etidocaine 15-20 6-8 hrs X 1.5 Lidocaine 20 60 mins X4 Mepivacaine 20 2 hrs X2 Prilocaine 20 2 hrs X2 Ropivacaine 20 8-9 hrs X1 STEREO SPECIFICITY Most ester LA are (procaine,chlorprocaine,) are achiral compounds Most amides are chiral drugs ( exception- lidocaine ) Most used clinically are Recemic mixtures But recently s-enantiomers are produced(S-bupivacinechirocaine) They have enhanced vasoconstricion So longer acting But can cause less duration & intensity of block Also has less cardiotoxicity Frequency or use dependent blocakde Local anesthetics block trains of action potentials >> single action potentials The more frequent the A.P, the more often Na+ channels are open. Also, affinity of the binding site increases. Magnitude and rate of block increase Depolarized membranes are more sensitive to local anesthetic action The more depolarized the membrane, the more Na+ channels are open Differential nerve blocakde LA cause differential nerve block pain > temperature > touch > deep pressure > motor Small diameter, unmyelinated C fibers (pain and autonomic) and Aδ fibers (pain) blocked before large diameter, myelinated Aα, Aβ or Aγ nerves (motor, limb position) Factors Causing Differential Nerve Block Critical length – About 3 nodes of Ranvier need to be blocked to completely prevent a.p. conduction down a nerve The larger the fiber, the greater the distance between nodes and the larger the area that needs to be exposed to LA Inhibition of >70% of Na+ channels will reduce the a.p. size Progressive blockade as you move down the axon results in failure to conduct after a sufficient length is exposed to drug Pain fibers and sympathetic fibers fire in high frequency bursts, whereas motor fibers fire at lower frequencies. LA with high use dependence (bupivacaine) therefore provide a better preferential block of pain and autonomic over motor function Axons at the periphery of a nerve (outside) are blocked more readily than those in the core because they are exposed to higher concentrations Proximal tissues are blocked more readily because their nerves lie in the perimeter of the nerve bundle In large mixed nerves, motor fibers lie on the outside Minimum concentration( Cm ) It is the minimum concentration of LA necessary to produce conduction blockade of nerve impulses It is analogous to MAC Larger nerve fibres – more Cm An increased tissue pH or high frequency nerve stimulation will decrease Cm vasodilatation Cocaine – vasoconstrictor ( moffett’s solution) cocaine 10% 1 ml + adrenaline 1ml 1:1000 + NaH(CO3)2 2ml 8.4%. Vosodilatation Pro >pri> l > M > B > R S-isomers less vasodilataion SYSTEMIC ABSORPTION It depends on Dose Vasoconstrictor presence Site of injection Intercostal block > caudal > paracervical> epidural> brachial plexus > intrathecal PLASMA PROTEIN BINDING & PLACENTAL TRANSFER Esters are not significantly protein bound(5-10% or less) Amides are highly protein bound by α1-acid glycoprotein There order is B>R>M>L>Pri In Pregnancy there is increased sensitivity to LA High protein bound drugs have low UV:M ratio For bupivacaine 0.2( so less transfer) For prilocaine 0.5 ( sp more transfer) PHARMACKOKINETICS Plasma concentration declines in a Biexponential manner Rapid distribution phase( 1- 3 mins) brain,myocardium,lungs,liver Followed by slower decline phase( muscle & fat) Terminal half life of most ester anaesthetics is short- 10 mins due to rapid hydrolysis by plasma & tissue cholinesterases The amides half life range from 100 mins -200 mins Volume of distribution> total body water Plasma clearance is comparable with liver blood flow Low cardiac output and hepatic cirrhosis will decrease there clearance METABOLISM & ELIMINATION Ester metabolism Most esters broken down by esterase enzymes Procaine- PABA – diethyamino ethanol – diethyglycine Exceptionally, COCAINE is resistant to hydrolysis by ChE. It is metabolised in liver Metabolites- Norcocaine, Ecgonine, benzoylated analouges They may be responsible for stumulant effects on CNS Amide metabolism Extensively metabolised in LIVER by AMIDASES Prilocaine- rapidly metabolised in liver and some extent in kidney & lung Its principle metabolite are N-Propylamine & oToludine – Methaemoglibinmia(> 600 mg) S-prilocaine produces less o-Toludine Bupivacaine- low hepatic clearance- slowly metabolised in liver Pipecolic acid & pipeco xylidine – metabolites lidocaine Convulsive properties Lidociane dealkylated Monoethy Glycine-xylidine + acetaldehyde hydrolysed N-Ethyl Glycine + 2,6 Xylidine 4-OH- 2,6 Xylidine (urine) • Glycine-Xylidine, a minor metabolite is a CNS depressant – has long half life Pulmonary Extraction Pulmonary extraction from the venous circulation limits the amount of local anesthetic (lidocaine , bupivacaine ), & prilocaine (Citanest)) that will reach the systemic circulation Bupivacaine : dose-dependent, first pass extraction (saturable, uptake) Propranolol inhibits bupivacaine extraction Propranolol reduces lidocaine & bupivacaine plasma clearance DRUGS COCAINE Has central & Peripheral effects Both effects are caused by inhibition of uptake1 in central & peripheral nerve endings Tachycardia , arrhthmias, vosoconstriction, pupillary dilatation & other sympathomimetic effects Corneal anaesthesia Nasal anaesthesia ( Moffets solution) C.I with TCA’s , pressor drugs Dependence & abuse Procaine Procainamide – not broken down by ChE. So its used for its antiarrhythmic property Class Ia – increases APD Vosodilator – Rx of vascular spasm caused by inadverdent intra-arterial injection Cardioplegia- CPB Benzocaine ( does not ionise, acts by ME) Topical ,ear drops,ointments Lidocaine Most commonly used LA Dose limits : the maximum recommended doses in the adult are, a. plain ~ 3 mg/kg b. with adrenaline ~ 7 mg/kg 2-4% - gels,ointments,creams 10 % - sprays 1-2 % epidural Hyperbaric solutions( dextrose) 5% - Intrathecal Hypobaric solutions( water) Bupivacaine 0.25% to 0.75% 0.5% heavy ( hyperbaric) most commonly used drug in UK intrathecally. the maximum recommended doses in the adult are, a. plain ~ 2 mg/kg b. with adrenaline ~ 2 mg/kg NB: this equates to ~ 25 ml of 0.5% in a 70 kg adult Chlorprocaine Used mostly in USA Used in OBS practice- 3% solution Differs from procaine by additon of chlorine atom , so its 4 times more quickly hydrolysed & has more rapid onset Preservative – neurotoxic Ropivacaine similar to bupivacaine, less cardiotoxic Etidocaine Amide derived from lidocaine It produces a more profound effect on motor than sensory nerves TOXICITY Toxicity 1.Caused by overdosage 2.As a part of Therapeutic procedure 3.Caused by Added vasoconstrictor 4.Specific Effects overdosage CNS TOXICITY Biphasic effects Small dose (2-4 mg/kg) will have anticonvulsant effects-Rx status epilepticus Large dose will cause numbeness of tongue & mouth,light headedness,visual disturbances,slurring of speech,muscular twitching & tremors,restlessness & irrational conversation Grand mal convulsions can occur( 10 mics/ml-lidocaine , 2 mics/ml – bupivacaine) Hypoxia & acidosis potentiates convulsions Prociane is relatively free from convulsant activity Initial effects- depression of inhibitory cortical pathways Profound effects- cortical & medullary depression CVS TOXICITY It follows CNS toxicity Hypotension, bradycardia, bradyarrhythmias, & cardiac arrest Bupivacine is more carditoxic It acts on K+ & Ca2+ channels in addtion to Na+ channels. Myocardial conduction is depressed Widening of QRS complex & distortion of ST- segemnts High doses can cause ventricular arrhythmia Tachycardia can enhance frequency dependent block of cardiac sodium channels & cause more cardiotoxicity S-bupivacaine & S-Ropivacaine have less carditoxicity As a part of theraupatic procedure Intercostal block > caudal > paracervical> epidural> brachial plexus > intrathecal Caused by vasoconstrictor Sympathomimetic amines will cause cardiac arrhythmias and hypertension Should not be added to digital nerve block,penile block . Specific effects Allergic responses- currently rare Skin rash Anaphylactic reactions Esters – PABA – more often cause this PABA antagonises sulphonamides Methaemoglobinaemia – o-toludine ADDITIVES & MIXTURES pH adjustment – NaOH & HCl Tonicity- NaCl Baricity- Glucose & H2O Preservatives – methyhydroxy benzoate ( fungicide) Reducing agents – Sodium Metabisulphate - to prevent oxidation of adrenaline ADDITIVES & MIXTURES vasoconstrictors Slows the rate of absorption Prolongs duration Reduces toxicity Enhnances the intensity of block Contraindicated: End arteries- ring blocks, penile blocks IVRA Adrenaline : Most commonly used & potent Conc: 1 in 80,000(12.5µg/ml) to 1 in 300,000(3.3µg/ml) Max dose should not exceed 0.5 mg. Noradrenaline : 1 in 80,000 were used but now rarely used due to its pressor effects Felypressin : Non catecholamine vasoconstrictor- vasopressin analouge, causes only peripheral vosoconstriction,no action on heart Safer for IHD patients but can cause pallor constrict coronary circulation. Dental use – 0.03 i.u./ml Phenyehrine Not used now , less effective than adrenaline CO2 (cabonated solutions) It is supposed to speed the onset of action CO2 rapidly diffuses across neurilemma & decreases intracellular pH ,so enhances the conversion of tertairy base(B) to the active form(BH+) But in clinical practice ,doubtful. CO2 is rapidly buffered by intracellular proteins and these solutions are unstable & can precipitate Dextrans : Prolong the duration because of its high molecular weight & are effective with combination with adreanline “Macromolecules” are formed between dextrans and LA & they are held in tissues for long periods. Hyaluronidase : It aids in spreading LA by breaking down tissue bariers Used mostly in Opthal practice. Mixtures-compounding Lidocaine + Bupivacaine To cause faster onset with long duration Will decrease there individual doses and thus reduce toxicity However ‘ toxicity is additve’ if it occurs. If ester is combined with amide, toxicity may increase because amide slows hydrolysis of ester by inhibiting plasma cholinesterase Eutectic mixture The combined melting point of the eutectic mixture is less than the either of the drugs EMLA- 5% cream Mixture of unionised(base) 2.5% prilocaine & 2.5% lidocaine It takes 1-2 hours to act- pediatrics Infants –contraindicated- methaemoglobinemia TAC solution The combination of tetracaine, adrenaline, and cocaine (TAC) has commonly been used for repair of lacerations in the face and scalp of children Other topicals PRAMOXINE : Minor burns ,pruritis, sigmidoscopy, laryngoscopy DYCLONINE HEXYLCAINE PIPERCOLINE All can be used before direct laryngoscopy and they are not amides or esters ,so useful for patients allergic to them. CLINICAL USES Regional anaesthesia Topical or surface anaesthesia Local infiltration Peripheral nerve blocks Bier block Epidural Spinal Anlagesia (propofol) To reduce intubation response To decrease ICT Ventricular dysrhytmias Suppression of grand mal seizures