* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Categorization
Survey
Document related concepts
Transcript
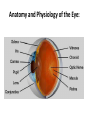
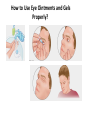
Ophthalmic Preparations • Definition: They are specialized dosage forms designed to be instilled onto the external surface of the eye (topical), administered inside (intraocular) or adjacent (periocular) to the eye or used in conjunction with an ophthalmic device Categorization • Solution/suspension into conjunctival sac • Lotions for irrigating & cleansing eye surface • Ointments/ creams/ gels for lid margin/ conjunctival sac • Contact lenses sol facilitate wearing & care of lenses • Parenterals (intracorneal,intraviterous, retrobulbar inj • Solids in conjunctival sac release over long period Drugs used in the eye: • • • • • • • • • • • Miotics e.g. pilocarpine Hcl Mydriatics e.g. atropine Cycloplegics e.g. atropine Anti-inflammatories e.g. Corticosteroids, antihistamines Anti-infectives (antibiotics, antivirals and antibacterials) Anti-glucoma drugs e.g. pilocarpine Hcl, beta-blockers Surgical adjuncts e.g. irrigating solutions Diagnostic drugs e.g. sodiumfluorescein Anesthetics e.g. tetracaine surgical procedurs Astringent e.g. zinc sulphate Diagnostic agents e.g fluorescein Anatomy and Physiology of the Eye: Formulation • • • • A.I/s therapeutic effect Vehicle ,usu aq./ can be oil (tetracycline HCl) Antimicrobial preservative , maintain sterility Adjuvants, adjust tonicity, viscosity, pH to ↑comfort in use & stability of A.I • Suitable container for storage & use • Most important sterility Preservatives: • Preservatives are included in multiple-dose eye solutions for maintaining the product sterility during use. • Preservatives not included in unit-dose package. • The use of preservatives is prohibited in ophthalmic products that are used at the of eye surgery because, if sufficient concentration of the preservative is contacted with the corneal endothelium, the cells can become damaged causing clouding of the cornea and possible loss of vision. So these products should be packaged in sterile, unit-of-use containers. • The most common organism is Pseudomonas aeruginosa that grow in the cornea and cause loss of vision. Benzalkonium chloride • Preservative of choice if not C/I for incompatibility or sensitivity • It is generally used in combination with 0.010.1% disodium edetate (EDTA). The chelating, EDTA has the ability to render the resistant strains of P.aeruginosa more sensitive to benzalkonium chloride. • Well-tolerated on the eye up to conc of 0.02%w/v, usu used at 0.01%w/v • Stable to sterilization by autoclave • Rapid bactericidal action G+ve/G-ve bacteria working on cell envelop • Active in the controlled aq. Environment & pH values of ophthalmic products • Activity is reduced in the presence of multivalent cations as they compete w/ the antibacterial for negatively charged sites on bacterial cell surface • Reduced activity if heated w/ methylcellulose or formulated with anionic & certain conc of nonionic surfactants • Incompatible w/ fluorescein ( large anion), & nitrates • It is sorbed from solutions through contact w/ rubber • Its antibacterial activity is enhanced by aromatic alcohols ( benzyl alcohol) • Its surface activity may be used to enhance the transcorneal passage of non-lipid-soluble drugs such as carbacol • It should not be used with local anesthetics as anesthetics will abolish blink reflex & preservative will result in drying & irritation of the cornea. Chlorhexidine acetate / gluconate • Cationic biguanide bactericide • Antibacterial activity in aq soln similar to benzalkonium Cl • Activity reduced in presence of other formulation ingredents • Activity against G-ve bacteria is enhanced by presence of aromatic alcohols + di-Na edetate • Activity antagonized by multivalent cations • Stability is greatest at pH 5-6, less stable to autoclaving than benzalkonium Cl • Well tolerated by the eye but allergic rxns may occur Chlorbutol • Chlorinated alcohol used at 0.5% w/v • Compatible with most ophthalmic preparations • Disadvantages: volatility, absorption by plastic containers, lack of stability at high temp Phenylmercuric salts • They are slowly active over a wide pH range against bacteria & fungi • Activity increased by phenylethanol • Activity decreased by di-Na-edetate • Marked absorption by rubber • Not used if there is a suitable alternative • The organic mercurial's should not be used in eye drops which require prolonged usage as it can lead to IO deposition of mercury Thiomersal • Organic mercurial • Bacteriostatic, fungistatic action • allergy Tonicity • Eye drops are to be made isotonic with the lacrimal fluid (equivalent to 0.9% NaCl) • Eye can tolerate small volumes of eye drops with tonicity range equivalent to 0.7-1.5% w/v NaCl. • Common tonicity adjusting agents: NaCl, KCl, buffer salts, glycerin, propylene glycol, mannitol Isotonicity Lacrimal fluid is isotonic with blood having an isotonicity value Corresponding to that of 0.9% Nacl solution • How many grams of NaCl should be used in compounding the following prescription? Pilocarpine nitrate 0.3 g Sodium chloride q.s Purified water ad 30 ml Make isotonic sol. For the eye Note : Pilocarpine nitrate NaCl equivalent 0.23 Viscosity enhancers: (to retard the rate of setting of particles) Disadvantages: 1- produce blurring vision as when dry form a dry film on the eye lids 2- make filteration more difficult pH Adjustment and Buffers: • pH adjustment is very important as pH affects: 1- to render the formulation more stable 2- The comfort, safety and activity of the product. Eye irritation increase in tear fluid secretion Rapid loss of medication. 3- to enhance aqueous solubility of the drug. 4- to enhance the drug bioavailability 5- to maximize preservative efficacy Examples: boric acid/borax, phosphate buffer, citrate buffer pH Adjustment and Buffers: pH & buffer Antioxidants • To protect A.I from oxidation (adrenaline, proxymetacaine, sulphacetamide, phenylephrine) • Na metabisulphite (preferred at acid pH & has antimicrobial activity & enhances activity of phenylmercuric nitrate) & Na sulphite (preferred at basic pH) are used at 0.1% w/v . Stable at soln protected from light Chelating agents • Such as di-Na-edetate are added to chelate heavy metal traces so enhance stability • It is also useful to enhance antibacterial activity & chemical stability at conc up to 0.1% Bioavailability • Most A.I are salts of weak bases • To penetrate the cornea the bases need to be at alkaline pH (unionized) • At tear pH they are able to penetrate the outer lipid layer of the lipid-water-lipid structure of the cornea • Inside the epithelium it will partially dissociate • So the water soluble moiety will traverse the middle aq stromal layer of the cornea • When it reaches the junction of the stroma & the endothelium it will again partially associate forming the lipid soluble moiety so it crosses the endothelium • Finally the drug dissociates into its water soluble form & enter aq humor • Then it can diffuse to the iris & the cillary body (site of action • Most effective penetration of lipophilichydrophilic-lipophilic corneal membrane is by A.I having both hydrophilic & lipophilic forms • Ex. Highly water soluble steroid phosphate ester have poor corneal penetration, but the less water soluble , more lipophilic steroid has much better corneal penetration Storage conditions • To minimize degradation of eye drops storage temp & conditions must be considered at time of formulation. • The stability of several eye drops is improved by refrigerated storage (2-8C) ex. Chloramphenicol eye drops Containers for eye drops • They should protect the eye drop from microbial contamination, moisture & air • Container material should not be shed or leached into solution, neither should any of the eye drop formulation be sorbed by the container • It should withstand sterilization method used if the product is to be sterilized in final container • Containers may be glass, plastic & may be single or multiple dose Single dose containers • The minims is single dose eye drop container • Container sealed at its base & has a nozzle with a screw cap • Sterilized by autoclaving in an outer heatsealed pouch with peel-off paper backing Plastic bottle • Most commercially prepared eye drops are supplied in plastic dropper bottles • The bottles are made from polyethylene or polypropylene & are sterilized by ionizing radiation prior to filling with sterile formulation under aseptic conditions Glass bottles • Most extemporaneously prepared eye drops are supplied in amber partially ribbed glass bottle bottle • Made from neutral /soda glass which had the inetrnal surface treated during manufacture to reduce release of alkali when in contact with sol • Neutral can be autoclaved more than once • Soda autoclaved once • Teat made from synthetic or natural rubber • Natural withstand autoclaving at 115⁰C for 30 min but not the high temp of dry heat sterilization • Silicone rubber will withstand dry heat sterilization & are suitable for use with oily eye drops . They are permeable to water vapor (susp become cake) • So aq eye drops having rubber teat have shelf life of 3 months whichcan be lengthened by supplying the sterile eye drop in bottle with an ordinary screw cap together with a separately wrapped & sterilized silicone rubber dropper • Teats & caps are used once only\ • All components are thoroughly washed with filtered distilled or deionized water dried & stored in a clean area until required • Rubber teats sorb preservatives & antioxidants during autoclaving & storage • Studies are to be done individually to counteract this loss Preparation of eye drops • • • • Preparation involves Preparation of the solution Clarification Filling & sterilization Preparation of the solution • The aq eye drop vehicle containing any necessary preservatives, antioxidant, stabilizer, tonicity modifier, viscosity enhancer or buffer should be prepared first • Then A.I is added & vehicle made up to vol. Clarification • They should be free from particulate matter & BP has stringent requirement for their absence • Sintered glass filter or membrane filter of 0.451.2 µm pore size are suitable • The clarified sol is either filled directly into the final container which are sealed prior to heat sterilization or filled into a suitable container to filtration sterilization • Clarified vehicle is used to prepare eye drops susp. Which are filled into final container & sealed prior to sterilization Sterilization • Autoclaving at 115⁰C for 30 min or 121⁰C for 15 min • Heating at 98-100⁰C for 30 min together with either benzalkonium Cl or phenylmercuric acetate or nitrate or thiomersal. This method is described in BP but is no longer a pharmacopeial recommendation method • Filtration through a membrane filter having 0.22 µm pore size into sterile container using strict aseptic technique • Dry heat sterilization at 160⁰C for 2 hours employed for non-aqueous preparation, Silicone rubber teat must be used • Immediately following sterilization the eye containers must be covered with a readily breakable seal to distinguish btw opened & unopened containers How to Use Eye Ointments and Gels Properly? Formulation of eye lotions • eye lotions used to assist in the cleaning of the external surface of the eye. • Remove non-impacted foreign body or clean away conjunctival discharge • For surgical use in surgical or 1st aid procedure should not contain antimicrobial preservative & should be in single-use container • Preparation is very simple, most common consist of NS • • • • • • Requirements Sterile & usu containing no preservative Isotonic w/ lacrimal fluid Neutral pH Large vol. but not greater than 200ml Non-irritant to occular tissue Labels include • Title identifying the product & conc of contents • Sterile until open • Not to be taken • Use once & discard the remaining solution • Expiray date • Preserved eye lotions would need additional label • Avoid contamination of contents during use • Discard remaining solutions not more than 4 weeks after 1st opening • Lotions should be supplied in coloured fluted bottles & sealed to exclude MO