* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 15.2 COMPLEX FORMATION And THE SHAPE OF COMPLEX IONS
Bond valence method wikipedia , lookup
Cluster chemistry wikipedia , lookup
Jahn–Teller effect wikipedia , lookup
Metal carbonyl wikipedia , lookup
Hydroformylation wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Spin crossover wikipedia , lookup
Metalloprotein wikipedia , lookup
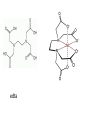
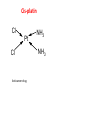
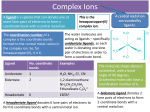
COMPLEX FORMATION And THE SHAPE OF COMPLEX IONS L.O.: Recall definition of ‘ligand’, ‘co-ordinative bond’, ‘co-ordination number’. Identify unidentate, bidentate and multidentate ligands. Explain the effect of the size of ligand on the shape of the complex ion. 2+ H2.O. H2O: Fe H2O: :OH2 :OH2 .. H2O A complex ion is formed when a transition metal ion is surrounded by ions or other molecules (called ligands), which are bonded to it by co-ordinative bonds. The number of co-ordinative bond that surround the metal ions called co-ordination number. COMPLEX IONS - LIGANDS Ligands atoms, or ions, which possess lone pairs of electrons form co-ordinate bonds to the central ion donate a lone pair into vacant orbitals on the central species Ligand Formula chloride Cl¯ cyanide NC¯ hydroxide HO¯ oxide O2water H2O ammonia NH3 Name of ligand chloro cyano hydroxo oxo aqua ammine CO-ORDINATION NUMBER & SHAPE the shape of a complex is governed by the number of ligands around the central ion the co-ordination number gives the number of ligands around the central ion a change of ligand can affect the co-ordination number Co-ordination No. Shape Example(s) 6 Octahedral 4 Tetrahedral 2 [Cu(H2O)6]2+ [CuCl4]2- Square planar Pt(NH3)2Cl2 Linear [Ag(NH3)2]+ Multidentate ligands: have more than one lone pair which can bond to a transition metal. Ligands which form only one co-ordinative bond per ligand and are said to be unidentate. Ligands which donate 2 lone pairs per ligand and are said to be bidentate. Multidentate ligands have more than one lone pair which they can bond to a transition metal. edta COMPLEX IONS - LIGANDS some ligands attach themselves using two or more lone pairs classified by the number of lone pairs they use multidentate and bidentate ligands lead to more stable complexes Multidentate form several co-ordinate bonds HAEM A complex containing iron(II) which is responsible for the red colour in blood and for the transport of oxygen by red blood cells. Co-ordination of CO molecules interferes with the process Cis-platin Cl Pt Cl Anticancer drug. NH3 NH3 5.4 EXERCISE 2 – Complex ions