* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download FACTORS AFFECT
Bond valence method wikipedia , lookup
Cluster chemistry wikipedia , lookup
Hydroformylation wikipedia , lookup
Metal carbonyl wikipedia , lookup
Jahn–Teller effect wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Metalloprotein wikipedia , lookup
Stability constants of complexes wikipedia , lookup
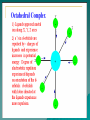
METAL LIGAND BONDING IN TRANSITION METAL COMPLEXES VALENCE BOND THEORY This theory was developed by Linus Pauling. According to this theory the bonding in metal complexes arises when a filled ligand orbital containing a pair of electrons overlaps with a vacant hybrid orbital on the metal atom. The VBT assumes the bonding between the metal atom and ligands to be purely covalent. BASIS OF VALENCE BOND THEORY The central metal atom in the complexes makes available a number of empty orbitals for the formation of coordinate bonds with suitable ligands. The appropriate atomic orbitals of the metal hybridise to give a set of new orbitals of equivalent energy, called hybrid orbitals. Each ligand has at least one orbital containing a lone pair of electrons. The empty hybrid orbitals of metal ion overlap with the filled orbitals of the ligand to form a covalent sigma bond. the bond is called coordinate bond. LIMITATIONS OF VALENCE BOND THEORY IT PROVIDES ONLY QUALITATIVE EXPLANATIONS FOR COMPLEXES. IT DOES NOT EXPLAIN THE VARIATIONS OF MAGNETIC MOMENT WITH TEMPERATURE. IT DOES NOT TAKE INTO ACCOUNT THE SPLITTING OF d –ENERGY LEVELS. IT DOES NOT PREDICT THE RELATIVE STABILITIES OF DIFFERENT STRUCTURES. CRYSTAL FIELD THEORY THE C.F.T THEORY WAS DEVELOPED BY H.BETHE AND V. BLECK.THIS THEORY CONSIDERS THE BOND BETWEEN METAL ION AND THE LIGAND AS PURELY ELECTROSTATIC. BASIS OF CRYSTAL FIELD THEORY THE TRANSITION METAL ION IS SURROUNDED BY THE LIGAND WITH LONE PAIR OF ELECTRONS. ALL TYPES OF LIGANDS ARE REGARDED AS POINT CHARGES. THE INTERACTIONS BETWEEN METAL ION AND THE NEGATIVE END OF ANION IS PURELY ELECTROSTATIC. THE LIGANDS SURROUNDING THE METAL ION PRODUCE ELECTRICAL FIELD WHICH INFLUENCES THE ENERGY OF d ORBITALS OF CENTRAL METAL ION. SHAPES of d- ORBITAL LIGANDS APPROACHING THE BARE METAL ION SPLITTING OF d-ORBITALS IN OCTAHEDRAL COMPLEXES HIGH SPIN COMPLEXES LOW SPIN COMPLEXES EXAMPLES OF HIGH AND LOW SPIN COMPLEXES VARIATION OF CFSE IN OCTAHEDRAL WITH DIFFERENT LIGANDS CFSE IN HIGH SPIN COMPLEXES CFSE IN LOW SPIN COMPLEXES TETRAHEDRAL COMPLEXES SPLITTING OF d-ORBITALS IN TETRAHEDRAL COMPLEXES SQUARE PLANAR COMPLEXES SPLITTINGIN SQUARE PLANAR COMPLEXES FACTORS AFFECTING MAGNITUDE OF CRYSTAL FIELD SPLITTING NATURE OF THE LIGAND- SMALL LIGANDS APPROACH THE LIGANDS EASILY, SO THEY CAN CAUSE GREAT CRYSTAL FIELD SPLITTING. LIGANDS CONTAINING EASILY POLARISABLE ELECTRON PAIR WILL BE DRAWN MORE EASILY TO THE METAL ION. THE METAL ION WITH HIGHER OXIDATION STATE CAUSE LARGE CRYSTAL FIELD SPLITTINGTHAN IS DONE BY THE ION WITH LOWER OXIDATION STATE. THE EXTENT OF C.F.S FOR SIMILAR COMPLEXES OF METAL IN THE SAME OXIDATION STATE INCREASED BY ABOUT THIRTY TO FIFTY PERCENT ON GOING FROM FIRST TRANSITION SERIES TO THIRD TRANSITION SERIES. THE INCREASE IS ALMOST SAME AMOUNT AS GOING FROM SECOND TRANSITION SERIES TO THIRD TRANSITION SERIES. THIS MAY BE EXPLAINED ON THE BASIS THAT 4d ORBITAL IN COMPARISON TO 3d ORBITAL ARE BIGGER IN SIZE.AS A RESULT 4d ORBITAL INTERACT STRONGLY WITH LIGAND. CRYSTAL FIELD SPLITTING ENERGY OF TETRAHEDRAL COMPLEXES IS NEARLY HALF THE VALUE FOR OCTAHEDRAL COMPLEXES. IN OTHER WORDS, THE VALUE OF C.F.S.E FOR TETRAHEDRAL COMPLEXES IS SMALL AS COMPARED TO PAIRING ENERGY P.THE TETRAHEDRAL COMPLEXES ARE THEREFORE MOSTLY HIGH SPIN COMPLEXES. LIMITATIONS OF CRYSTAL FIELD THEORY It does not take into account the partial covalent character of metal-ligand bond. It does not consider multiple bonding between metal ion and ligand. It does not explain the relatives strengths of ligands. It does not explain the charge transfer bands. It considers only d-orbitals of metal ions.