* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Slide 1
Survey
Document related concepts
Transcript
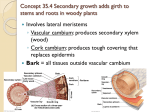
Substitution reactions at octahedral complexes: the search for mechanism Begin by determining whether the intimate mechanism is a or d. Table 1 Aquation refers to the reaction [Co(NH3)5X]n+ + H2O [Co(NH3)5(H2O)]3+ + X Rate constants vary by 6 orders of manitude Strongly dependent on the nature of the leaving group Anation refers to the reaction [Co(NH3)5(H2O)]3+ + Y [Co(NH3)5(H2O)]n+ + H2O Rate constants vary by a factor of 10 Weakly dependent on the nature of the entering group d activation Table 2 - Data for Ru3+ Rate more sensitive to the nature of the entering group than the leaving group •anation reactions vary by 3 orders of magnitude •aquation reactions vary by at most 2 orders of magnitude probably under a activation Steric effects If one crowds the metal ion: • speed up reactions under d activation • retard reactions under a activation Table 3 - Data for Co3+ complexes of the type Cl N N Co R N N As bulk of the equatorial ligand R increases, so does the rate of the reaction Cl d activation Electronic effects If the inert ligands stabilise a 5 coordinate intermediate, and the reaction proceeds faster, then we conclude the reaction is under d activation Table 4 •The saturated complex (cyclam) reacts slowly •bis(dmg) complex reacts faster -unsaturated, with electron-withdrawing substituents •trans[14]diene reacts fastest - unsaturated; electron donating group (CH2) on N So, increasing the donation of electron density to the metal ion stabilises the loss of the chloride axial ligand d activation Table 5 The reactivity of cis versus trans complexes N Cl Cl N N Co X N Co N N N N X displacement of Clby H2O H 2O NH3 donors only ClOH- low down in the spectrochemical series donors cis complexes where these are present are quite reactive This accords with a mechanism under d activation Cl- departing p orbital of a donor like Clof OH- in the cis position donates electron density into emerging vacant metal orbital Cl- departing donor in the trans position orthogonal orbitals (no net overlap) rearrange (slow) donation Consider an aqua complex. We saw in Chapter 3 that... D saturating rate constant = k1 rate of dissociation of departing X interchange rate constant of X and Y I =k A = only saturates at the diffusion limit Hence, for a D mechanism, ksat = k1 and the limit is set by the rate of water exchange For an Id mechanism, ksat = k, the rate constant for the exchange of departing H2O and entering Y But [H2O] = 55 M in aqueous solution since [H2O]outer sphere >> [Y]outer sphere, the rate is also limited by the rate of water exchange For an Ia mechanism, ksat = k, the rate constant for the exchange of departing H2O and entering Y. But this is dominated by bond forming between entering Y and the metal rate could be greater than the rate of H2O exchange Hence: for d actication, rate cannot be > rate of H2O exchange for a activation, the rate may be greater than the rate of H2O exchange Table 6 Rh3+ and Ir3+ complexes under associative activation Effects of charge See Table 7 For d activation: [Cr(H2O)5X]n+ [Cr(H2O)5]m+ + X (X = H2O, OH-) (This is a D process; Id would have Y involved as X departs.) As the charge on the metal complex increases, the stronger the MX bond rate decreases Rate is faster when X = H2O (n+ = 3+) than when X = OH- (n+ = 2+) Cr3+ data is in line with a d intimate mechanism Electrostriction Ordering or disordering of solvent molecules around the metal centre during a chemical reaction Effect is predominantly seen in values of S‡ _ + _ [+ _ [ + _ [ [ + = = Charge density has been increased in the transition state S‡ < 0, as the solvent becomes more ordered around the system o [ o o [ o = The ordering of the solvent is largely unaffected and the contribution to S‡ will be close to zero. [+ _ [ + _ = There is charge neutralisation in the transition state; the solvent will be less ordered and the electrostriction contribution to S‡ > 0 Corrections for electrostriction effects should be made before any definitive statements concerning mechanism based on values of S‡. After correction for electrostriction effects: S‡ > 0 S‡ < 0 S‡ 0 d a no conclusions can be reached