* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download PowerPoint - Balancing Equations
Chemical potential wikipedia , lookup
Electron configuration wikipedia , lookup
Isotopic labeling wikipedia , lookup
Bose–Einstein condensate wikipedia , lookup
Transition state theory wikipedia , lookup
Electrochemistry wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Detailed balance wikipedia , lookup
State of matter wikipedia , lookup
Chemical bond wikipedia , lookup
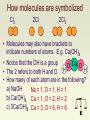
How molecules are symbolized Cl2 2Cl 2Cl2 • Molecules may also have brackets to indicate numbers of atoms. E.g. Ca(OH)2 • Notice that the OH is a group O Ca O H • The 2 refers to both H and O H • How many of each atom are in the following? a) NaOH Na = 1, O = 1, H = 1 b) Ca(OH)2 Ca = 1, O = 2, H = 2 c) 3Ca(OH)2 Ca = 3, O = 6, H = 6 Balancing equations: MgO • The law of conservation of mass states that matter can neither be created or destroyed • Thus, atoms are neither created or destroyed, only rearranged in a chemical reaction • Thus, the number of a particular atom is the same on both sides of a chemical equation • Example: Magnesium + Oxygen • Mg + O2 MgO Mg + O O Mg O • However, this is not balanced • Left: Mg = 1, O = 2 • Right: Mg = 1, O = 1 Balance equations by “inspection” Mg + O2 MgO 2Mg + O2 2MgO is correct Mg + ½O2 MgO is incorrect Mg2 + O2 2MgO is incorrect 4Mgwith + 2elements O2 4MgO is incorrect Hints: start that occur in one compound on each side. Treat polyatomic ions that repeat as if they were a single entity. a) P4 + 5 O2 P4O10 b) 2 Li + 2 H2O H2 + 2 LiOH c) 2 Bi(NO3)3 + 3 K2S Bi2S3 + 6 KNO3 d) 2 C2H6 + 7 O2 4 CO2 + 6 H2O From Balance the Following Equations 1. __ KNO3 __ KNO2 + __ O2 2. __ Pb(NO3)2 __ PbO + __ NO2 + __ O2 3. __ P4 + __ I2 __ PI3 4. __ MgO + __ H3PO4 __ Mg3(PO4)2 + __ H2O 5. __ Br2 + __ KI __ I2 + __ KBr 6. __ Ca(OH)2 + __ HNO3 __ Ca(NO3)2 + __ H2O 7. __ Bi2O3 + __ H2 __ Bi + __ H2O 8. __ Fe + __ O2 __ Fe3O4 9. __ CaO + __ C __ CaC2 + __ CO2