* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Amiodarone Infusion Guideline
Heart failure wikipedia , lookup
Electrocardiography wikipedia , lookup
Antihypertensive drug wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Ventricular fibrillation wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
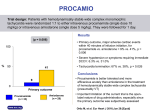
Amiodarone Infusion Guideline AMIODARONE INFUSION 1 ampoule = 150mg Amiodarone hydrochloride in 3mL, fine white crystalline powder Amiodarone infusion should only be initiated under hospital or specialist supervision. Indications: • All types of tachyarrhythmia’s including: o paroxysmal supraventricular, o nodal and ventricular tachycardias; o atrial fibrillation and flutter; o ventricular fibrillation • Tachyarrhythmia’s associated with Wolff-Parkinson-White syndrome • Arrhythmia’s when all other agents cannot be used. Dose: Initial Bolus Dose Then 23 hour infusion Tachyarrhythmia’s • Add 300mg • Add 450mg amiodarone to amidodarone to 250mL 250mL 5% Dextrose infuse over 11.5 hours 5% Dextrose. • Infuse over 1 hour • then add a further 450mg amiodarone to 250mL 5% Dextrose infuse over 11.5 hours Max. Dose 1200mg in 24 hours Infuse both at 22mL/hour Note: use non-PVC (excel) 250mL 5% Dextrose Injection USP bags Note: Oral therapy should be initiated as soon as possible after adequate response is obtained. Overlap of oral and intravenous medication is recommended for two days to prevent plasma levels falling and efficacy being lost. Administration: • Solution must be diluted before administration • Compatible with 5% Dextrose ONLY. • Use non-PVC (excel) 250mL 5% Dextrose Injection USP bags and low-sorbing PE/PVC tubing. • Add required dose to 250mL of 5% Dextrose Injection USP non-PVC (excel) bag. • Administration via a large bore vein or by a central venous catheter is recommended. Amiodarone Infusion Guideline Document Owner: Medication Safety Committee WCDHB-MSC Version 1, Issued 19/12/13 Page 1 of 3 Master Copy is Electronic Amiodarone Infusion Guideline Observation and Monitoring: • Continuous cardiac monitoring is required with daily ECG and QT interval checks. • BP and heart rate hourly for the first 4 hours, then 4 hourly until confusion complete. If hypotension occurs, slowing or stopping the infusion temporarily is usually adequate. • Doctors must chart acceptable clinical parameters on the prescription sheet as severe hypotension and/or bradycardia may occur. Mechanism of action: Amiodarone is a Class III antiarrythmic agent which inhibits adrenergic stimulation (alpha-and betablocking properties). It prolongs the action potentional and refractory period in myocardial tissue through its effect on sodium, potassium and calcium channels; and, it further decreases AV conduction and sinus node function. Contraindications: • Known hypersensitivity to iodine or amiodarone. • Sinus bradycardia, sino-atrial heart block • Unless pacemaker fitted avoid in severe conduction disturbances or sinus disease node • Severe respiratory failure, circulatory collapse, or severe arterial hypotension • Thyroid dysfunction • Avoid bolus injection in congestive failure or cardiomyopathy • Lactation • Combined therapy with medicines that may induce ‘Torsades de Pontes’ Precautions: • In patients with hypotension, uncompensated or severe heart failure. • Patients with pacemakers/implantable defibrillators • In patients with cardiac disorders – may occasionally worse existing heart failure Discontinue in patients with 2nd or 3rd degree AV block, sinoatrial block, bifascicular or trifascicular block in patients with previous history of atypical ventricular tachycardia • Deranged LFTs • Pregnancy –use only if no alternative • Patients with history of thyroid dysfunction • Concomitant use with medications that prolong QT interval • Chronic renal failure; electrolyte imbalance (e.g. Hypokalaemia, hypomagnesaemia); may lead to reduced effectiveness or arrhythmia Possible adverse effects: • Common decrease in blood pressure (moderate and transient), hot flushes sweating or nausea, taste disturbances, • Less common onset or worsening of arrhythmia, conduction disturbances, peripheral neuropathy, myopathy • Very rare acute liver disorders with elevated serum transaminases and/or jaundice; interstitial pneumonitis, headache, ataxia, thrombocytopenia, rash, haemolytic or aplastic anaemia Amiodarone Infusion Guideline Document Owner: Medication Safety Committee WCDHB-MSC Version 1, Issued 19/12/13 Page 2 of 3 Master Copy is Electronic Amiodarone Infusion Guideline Drug interactions: Medications Antipsychotics Citalopram Dabigatran Statins Digoxin Anti-coagulants Interaction Can lead to dangerous QT prolongation if used together; potentially fatal torsade de pointes arrhythmia. Risk of QT prolongation if used together Plasma concentration of Dabigatran may be raised Risk of muscular toxicity is increased when given in combination with statins metabolised by CYP 3A4. Amiodarone can double digoxin levels Metabolism inhibited by Amiodarone and INR levels may increase Storage: Store below 30oC. Protect from light. Do not refrigerate. Discard any unused solution after 12 hours. List of excipients: Polysorbate 80, benzyl alcohol and water for injections, as well as hydrochloric acid and sodium hydroxide for pH adjustment. Note: Amiodarone contains 2 atoms of iodine and bears a structural resemblance to the molecule of thyroxine. Each 3ml ampoule contains approximately 56mg iodine References: References 1 2 Title/Description Drug information handbook 21st Ed. 2012-1013 Notes on Injectable Drugs 6th Ed. 2010 3 4 5 6 British National Formulary 64th Ed. Published September 2012. Amiodarone Hydrochloride injection Datasheet. Available from www.medsafe.govt.nz Drugs in Pregnancy and Lactation 8th Ed. Briggs, G. Freeman, R. Yaffe S Stockleys Drug Interactions 7th Ed. Pharmaceutical Press Amiodarone Infusion Guideline Document Owner: Medication Safety Committee WCDHB-MSC Version 1, Issued 19/12/13 Page 3 of 3 Master Copy is Electronic