* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download D. To increase stability of the drug
Survey
Document related concepts
Discovery and development of ACE inhibitors wikipedia , lookup
Discovery and development of cephalosporins wikipedia , lookup
Drug design wikipedia , lookup
Pharmacognosy wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Neuropharmacology wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Drug interaction wikipedia , lookup
Discovery and development of integrase inhibitors wikipedia , lookup
Theralizumab wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Drug discovery wikipedia , lookup
Discovery and development of neuraminidase inhibitors wikipedia , lookup
Transcript
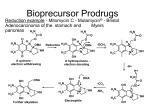
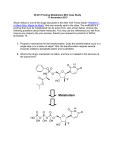
Pharmaceutics IV/PH 406 Chemical Targeting Dr. Hamm-Alvarez/PSC 406A 2-1445/[email protected] What is a prodrug? Prodrugs are derivatives of drugs that can be converted enzymatically or chemically to the biologically active drug, through the action of hydrolases, transferases, lipases, oxidoreductases, etc. Prodrugs can be designed to enhance the delivery characteristics and therapeutic value of the parent drug Since the prodrug approach is based on chemical modification of drugs, this is considered a chemical approach to drug targeting. As with any drug targeting strategies, the prodrug strategy requires constant balancing between toxicity and specificity Criteria for characterization as a successful prodrug: 1. ABSORPTION. The prodrug must be readily transported to the target site and uptake must be reasonably rapid. 2. ACTIVATION. Once at the site, the prodrug must be selectively cleaved to the active drug relative to its conversion at other sites in the body. 3. RETENTION. The active drug, once selectively generated at the site, must be retained by the tissue. Paths to the discovery of prodrugs: 1. Analysis of metabolites of pharmacologically active drugs a. Codein (prodrug) is converted to morphine (active drug) b. Acetylsalicylic acid (prodrug) is converted to salicylic acid 2. Design of prodrugs by chemical modification of drugs with some in vivo activity. This is a RATIONAL approach to improve the usefulness of an already useful drug 3. Design of an active prodrug starting from a drug that has no in vivo activity, but promising in vitro activity. I. What is the role of hydrophilicity and hydrophobicity in chemical targeting? 2 II. Why use chemical targeting? A. To alter membrane permeability Ampicillin: The antibiotic ampicillin is very hydrophilic and does not easily diffuse across the gastrointestinal epithelium following oral administration. If its free carboxyl group is esterified, its hydrophilicity is drastically reduced. Several prodrug derivatives of ampicillin have been generated by esterification, including Bacampicillin and Pivampicillin, which are readily converted into ampicillin by esterases primarily in the gastrointestinal mucosa, and then the active species, Ampicillin, is released into the bloodstream. H C O H C N S +NH3 N C OR O R = H Ampicillin R = CH(CH3)OCOOC2H5 R = CH2OCOC(CH3)3 Bacampicillin Pivampicillin B. To choose dosage form Chloramphenicol is an antibiotic that can be given orally but its use is limited in pediatric applications by bitter taste. To interact with taste receptors, the drug needs to be solubilized in saliva. When solubility is reduced by complexing the drug with palmitate, the bitter taste is eliminated. The prodrug is converted in the gut by pancreatic lipase to chloramphenicol and palmitate; luckily pancreatic lipase is active even on the insoluble particles. Chloramphenicol palmitate ester Pancreatic lipase 3 Chloramphenicol + Palmitate C. To alter aqueous solubility Fosphenytoin sodium (Cerebryx) is a phosphate ester prodrug of phenytoin. It is a replacement for parenteral therapy with the antiepileptic drug, phenytoin. Fosphenytoin is supplied as a ready-mixed solution in water buffered to physiological pH. In contrast, phenytoin has to be administered in 40% propylene glycol and 10% ethanol, adjusted to pH of 12. The high pH causes adverse effects at the injection site, so the desired properties of the prodrug include improved aqueous solubility under the necessary dosage form to reduce side effects. Fosphenytoin is rapidly converted to phenytoin by phosphatases present in the liver, red blood cells and other tissues, with a conversion half life of 8-15 min. H H N O N O N CH2 O O P O O N O CH2O PO -3 4 H O Fosphenytoin Phenytoin D. To increase stability of the drug Levodopa or L-dopa is a prodrug of dopamine that is used for the treatment of Parkinson’s disease. Dopamine itself is presystemically activated by sulfate and glucuronide conjugation, requiring continuous infusion. L-dopa is transported across the blood brain barrier by a neutral amino acid transporter and then converted to dopamine by L-dopa decarboxylase. LDopa can be absorbed and activated in non-CNS tissues due to the presence of the neutral amino acid transporter and the enzyme, decarboxylase, in nontarget tissues. This non-specific absorption and activation is responsible for most side effects in L-Dopa therapy. An improvement of CNS targeting is achieved if the blood-brain barrier impermeable decarboxylase inhibitor (carbidopa) is co-administered. This prevents the activation of L-dopa in peripheral tissues but not in the brain (Sinemet, Merck , Sharp & Dohme) 4 E. To eliminate toxicity Aspirin (acetylsalicylic acid, a prodrug) is less corrosive than salicylic acid. Dipivefrin is a pivalic ester prodrug of adrenaline that is given in treatment of glaucoma by local administration. This ester is 100 times more active in lowering intraocular pressure than adrenaline, due to more efficient transport through the cornea which is followed by cleavage of the ester when it passes from the cornea into the aqueous humor. Because lower doses of the ester are given, the risks of cardiac side effects due to systemic adrenaline absorption from tear duct overflow are lowered. OR RO CH R = H Adrenaline R = COC(CH3)3 Dipivefrin 5 CH2NH2Cl III. How are prodrugs activated in a tissue-specific manner? A. Presence of tissue-specific enzymes Acyclovir is used in treatment of herpes simplex viruses types 1 and 2, and also for varicella zoster virus (“shingles”). This is an example of a drug that is converted into the active form only in the virally infected cells due to the presence of a specific viral enzyme. The basis for its use is that viruses are less selective in the phosphorylation of possible nucleotide precursors. Administration of acyclovir to cells expressing viral enzymes results in phosphorylation of acyclovir by viral thymidine kinase and production of a phosphorylated product which is trapped in the cell. This product can be phosphorylated again by cellular enzymes to yield the active triphosphate compound, which can be incorporated into viral DNA and disrupt the replication cycle by inhibiting viral DNA polymerase. Administration of acyclovir to cells not expressing viral enzymes causes little effect, since the host enzymes do not modify the prodrug. The selectivity here comes from the activation process, prodrug accessibility to phosphorylating enzyme and retention. O O N HN H 2N N N N HN HSV-TK H 2N N N P O HO O O GMP Kinase O O N HN N HN CELLULAR enzymes H2N P P N N H2N P P O P N N O O O Valacyclovir: valine ester of acyclovir with better oral bioavailability 6 Even if a selective viral converting enzyme is absent, generation of nucleotide analog triphosphates is a general strategy for treatment of viruses, particularly if there is a differential selectivity for incorporation of the nucleotide analog triphosphate by viral rather than cellular polymerases. The tissue specific enzymes in the case of many second and third generation antivirals are the polymerases themselves. Problem: Nucleotide is too polar to enter but sometimes the conversion step of nucleoside to nucleotide is inefficient. Figures from Zemlicka, BBA 1587:286, 2002. Approach: Modification of different pronucleotides with lipophilic phosphoramidates, which can be intracellularly cleaved by esterases—PPA pronucleotides 7 8 Modification of methylenecyclopropane 9 B. Use of unique environment within target tissue Hypoxia/Reducing Environment or altered pH Hypoxic cells are enriched in tumors as a result of poor vascularization. Hypoxic cells are also targets in cardiovascular disease (ischemia), neurodegenerative diseases, immune diseases and others. These cells have low oxygen tension, low nutrient levels and often low pH. Targeting of hypoxic tumor cells has been most studied because these cells are often resistent to radiation and chemotherapy. The bioreductive approach involves the selective release of the active species from the prodrug in the presence of a reducing environment. Examples of bioreductive enzymes in hypoxic cells: DT-diaphorase, NADPH reductase, Cytochrome b5 reductase, xanthine dehydrogenase Jaffar et al., Adv Drug Del Rev 53, 217, 2001 pH can be similarly utilized, by the triggering of pH sensitive cyclization reactions in specific prodrugs, and by the use of lowered pH to active and/or retain prodrugs in specific intracellular compartments (lysosomes, for instance) 10 C. Bring the converting enzyme to the target tissue (*Note, these strategies blend the chemical targeting and biological targeting approaches!) ADEPT: Some of the best examples of the use of highly toxic drugs in a targeted manner as prodrugs use the “ADEPT” strategy (Antibody-directed enzyme prodrug therapy). This approach targets an antibody-enzyme conjugate to the target site (usually a tumor, which has some characterized antigens). A prodrug is then added, which can be locally cleaved at the target site, such that toxicity to the surrounding tissues can be minimized. Important points for prodrug selection: 1. the prodrug should have a large differential in cytotoxicity between drug and prodrug 2. the prodrug should be a good substrate for the enzyme under physiological conditions 3. mammalian homologues should not be able to cleave the prodrug 4. the biologically active agent should have a short half life Important points for enzyme selection: 1. Activity under physiological conditions 2. Low immunogenicity 3. No endogenous activity in humans 4. High specific activity (because the amount of enzyme that can be delivered is low) 5. Examples: Carboxypeptidase G2—active with nitrogen mustard derivatives, which can normally not be utilized due to extreme toxicity. Carboxypeptidase A—can cleave methotrexate--alanine, endogenous CPA is a problem Alkaline phosphatase—can cleave a wide variety of very potent prodrugs including precursors to etoposide, mitomycin, doxorubicin, phenol mustard phosphate; however, large pool of endogenous alkaline phosphatase is highly problematic, leading to non-specific cleavage of prodrug 11 Other issues: Technical difficulties in generation of an antibody-enzyme conjugate that retains antigen-binding and enzymatic activities. Typically, conjugation chemistry based on thioether linkages is most commonly utilized. Fusion proteins combining antibody-enzyme activities, e. g. “Catalytic Antibodies” may be possible (see below) Immunogenicity of bacterial enzymes (used to avoid problems with endogenous activity) and non-human antibodies, necessity to use immunosuppressive drugs Example of catalytic antibody approach: release of etoposide from novel prodrug at tumor site by injected catalytic antibody (Shabat et al. PNAS 98:7528, 2001) 12 Remaining Issues in this study: Targeting of catalytic antibody (injection here, not based on use of tumor antigen), solution would be further modifying to produce a hybrid or bifunctional antibody GDEPT: Because of many of the technical difficulties above, another approach that has been taken is to introduce the activating enzyme into the target tissue with a gene therapy approach, and then to introduce the prodrug. This is also called suicide gene therapy. Major Issues: 1. Efficient, selective and non-toxic delivery of the suicide gene to the target tissue 2. Factors involved in prodrug choice remain similar to those for ADEPT 3. Because of limitations in efficiency of foreign gene expression, need a bystander effect (i.e., active drug activity on adjacent cells); for this reason some investigations have explored the possibility of external tethering of the expressed enzyme to better facilitate the bystander effect 13 Kirn et al. Trends Molec Med 8:S68, 2002. GDEPT systems tested in clinical trials: Herpes simplex virus thymidine kinase-canciclovir combination Cytosine deaminase-5-fluorocytosine combination 14 Virotherapy (Chemical/Biological Targeting) Viruses have evolved to infect cells, replicate, induce cell death, release additional particles and to spread. Replication in tumor tissue leads to amplification of the input dose, while a lack of replication in normal tissues is associated with efficient clearance. Since we now understand many factors required for viruses to appropriately replicate, as well as the factors promoting carcinogenesis, we can envision the design of novel agents such as replication-selective oncolytic viruses (virotherapy) as targeted anticancer therapy. Virotherapy can consist of modified viruses with infectivity in many cells (adenovirus) or viruses with inherent tumor sensitivity (Newcastle disease virus). Engineered viruses: most work has focused on adenovirus. Adenovirus replication and cell lysis requires expression of early genes including E1A and B. One example is the insertion of the prostate-specific antigen (PSA) promoter/enhancer element upstream of the E1A region of the viral genome. Expression of PSA is high in prostate tumor cells, enabling activation of E1A selectively in these cells. Another case is based on the p53 inactivation observed in most tumors. A compensatory mutation in adenovirus (deletion of a p53 binding protein) renders this virus selective for replication in cells lacking p53. The mechanism of this is not completely understood but the efficacy of this virus has been demonstrated in phase II trials of p53-minus tumors in head and neck cancer patients. The other major signaling modification identified in cancer cells includes mutations in the RB pathway. Selective adenovirus deletions in regions which interface with this signaling pathway have enabled the selective expression of these variants in tumors. 15 16 Combination Virotherapy/GDEPT The Armed Therapeutic Virus™ program (ONYX Pharmaceuticals) consists of using selectively replicating adenoviruses to deliver anticancer genes, such as the genes that make enzymes capable of converting inactive prodrug molecules into toxic chemotherapeutic agents. Press Release: Onyx Presents Data on Tumor-Specific Intravenous Gene Delivery at 11th International Conference on Gene Therapy of Cancer RICHMOND, CALIF. (December 13, 2002) – Onyx Pharmaceuticals, Inc. (Nasdaq: ONXX) today announced the presentation of preclinical data showing the ability of ONYX-411, a second-generation therapeutic virus, to specifically deliver anticancer gene products to tumors following systemic (intravenous) administration. The presentation was given today by Yuqiao Shen, Ph.D., senior scientist of Onyx Pharmaceuticals at the 11th International Conference on Gene Therapy of Cancer in San Diego, California. “We believe these results provide compelling in vivo evidence that ONYX411, an adenovirus engineered to selectively replicate in tumors with mutations in the retinoblastoma (RB) pathway, can be used as a safe and efficient vector for the delivery of gene products to the tumor,” said Leonard Post, Ph.D., Onyx senior vice president, research and development. “Onyx’ proprietary technology for inserting the therapeutic gene into the vector is designed to ensure that the gene is expressed only when the virus replicates. Since the virus is designed to replicate only in tumors, the gene expression is restricted to the tumors. Using this technology, we believe we can increase the cancer-killing properties of our viruses while minimizing the effect on healthy cells, even after intravenous administration.” In his presentation, Dr. Shen described experiments in which ONYX-443 was administered intravenously to immunodeficient mice carrying human tumor xenografts. ONYX-443 is an Armed Therapeutic Virus product constructed by modifying ONYX-411 to express a gene that makes the enzyme cytosine deaminase (CD). CD converts a non-toxic prodrug (5fluorocytosine, or 5-FC) into a highly toxic chemotherapeutic agent (5fluorouracil, or 5-FU). Dr. Shen reported that strong expression of CD was 17 detected only in tumors, not in liver or other normal tissues, following the systemic administration of ONYX-443. Moreover, during time course studies (five days post-virus injection until 24 days post-injection in some experiments), CD expression in tumors increased with time, presumably due to the continuous replication and spread of the virus. Expression of CD appeared to correlate with virus replication in vivo. 18