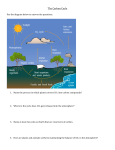
* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 1 • Venus – Thick, hot, CO 2 – dominated weakly oxidizing
Survey
Document related concepts
Transcript
• Venus
– Thick, hot, CO2 – dominated weakly oxidizing
atmosphere
• Mars
– Thin, cold CO2 – dominated weakly oxidizing
atmosphere
• Jupiter, Saturn, Uranus, Neptune
– Massive H2 & hydrocarbon – based, reducing
atmospheres
– Large escape velocity & little outgassing:
atmosphere probably similar to that with which
they were originally formed
• Titan (moon of Saturn)
– N2-dominated, weakly reducing, complex
hydrocarbon – based chemistry
1
2
• Main species identified: CO2, CO, SO2, O2, HCl
• Catalytic cycles involving CO, ClO and SO destroy O2
(analogous to ozone chemistry in Earth's
stratosphere!).
• CO2 photolysis one of the main processes (due to
large concentration of CO2).
– Note: recombination reaction CO + O + M CO2
+ M is spin-forbidden, hence needs to occur
catalytically!
• HCl photolysis liberates hydrogen, determines
escape rate of H from Venusian atmosphere.
• H radicals may also form H2O.
• Summary: photochemistry converts HCl and CO2 to
H2O, CO and Cl22 in the stratosphere. Cl2 transported
downward, and cycle is closed by thermochemical
reactions in the lower atmosphere.
• Very strong greenhouse effect due (mainly) to
extremely high absolute concentration of CO2.
• Thick cloud layers with complex structure; probably
consist mainly (though not solely) of sulfuri acid
• Clouds may also contain some water, metal chlorides
(e.g. Fe2Cl6 and Al2Cl6), phosphorus compounds
such as H3PO4 and possibly S8, possiblly NOHSO4.
• Lightning may generate some NOX from N2 (3.5% of
atmosphere), this may be responsible for formation of
H2SO4 (remember: very little O2 around!) by the net
reaction (several steps)
NO2 + SO2 + H2O => H2SO4 + NO
3
• Chemistry dominated by thermal reactions of sulfur
and carbon species (little UV due to shielding by
clouds, but lots of thermal energy due to high T).
• Main compounds: CO2, CO, COS, SO, SO2, SO3, S2,
S3, Sx>3, H2S
• Oxidized species SO2, SO3 more abundant than
reduced species COS, H2S: indirect evidence for
oxidizing conditions.
• Strong winds (up to 60x the planetary rotation speed,
compare to 10-20% on Earth)
• Atmosphere physically stable: little evidence of
seasonal changes or variable meteorology.
4
• Large seasonal variability, in contrast to Venus; even
the total pressure varies by ca 25%.
• Lots of weather: dust storms, cloud layer of
(probably) water ice forms during winter, CO2
condensation probably occurs in polar regions during
winter (Tmin = 125 K)
• CO2 photolysis important as on Venus, recombination
of CO and O likely catalyzed by HOX species (more
abundant on Mars than on Venus due to greater
relative abundance of H2O).
• H2O photolysis produces hydrogen atoms, part of
which are lost to space ("drying" of Mars).
• Atmosphere of Mars is simple enough that chemistry
models incorporating about 100 reactions work very
well.
• .
• Escape velocity only about 5 km/s due to low gravity.
• Hydrogen easily lost; also larger atoms and
fragments can be lost if they are produced with
enough energy in high-altitude photochemistry.
• The total amount of hydrogen lost over the life of the
solar system (4.6 billion years) corresponds to a
2.5...500 m thick layer of water (large uncertainty e.g.
due to unknown UV fluxes in early solar system).
• The early Martian atmosphere may have been quite
thick!
5
•
Wayne pg. 584
• Very massive planets: essentially no escape to
space, and more gravitational sedimentation of gases
by mass than on inner planets.
• Composition: mainly H2, some percent He
• Important trace gases: CH4, some C2H6, C2H4 &
heavier hydrocarbons; NH3, PH3, H2O
• Reducing environment: very few oxidants (and no
free oxygen)
• Photochemistry converts CH4 into heavier
hydrocarbons and NH3 into N2H4
• General cycle: heavier & more complex molecules
formed by photochemistry, transported downward,
decomposed thermally & react with H2 to regenerate
CH4, NH3.
– (Partially contradicts general theme of "less
complicated molecules at higher altitudes")
6
•
Wayne pg 590
• Smaller but denser than Jupiter or Saturn
• Roughly similar composition, but e.g. higher He/H2
ratio (closer to that of the sun, at about 15% He). • Also, much more CH4 (1-2% vs 0.3-0.5% for Jupiter &
Saturn)
• General chemical cycle similar as for Jupiter &
Saturn: photolysis & production of more complex CH, N-H & P-H species higher up, thermal reactions
close the cycle lower down.
• Quantitative differences due to less UV radiation, and
colder temperatures: hydrocarbon condensation
occurs on Neptune, unlike the other gas giants.
• Some interesting but poorly understood chemistry:
Neptune's atmosphere has HCN, unlike any other
planet's except Earth (Titan also has HCN). 7
• The only satellite in the solar system that possesses a massive
atmosphere (1.5 times Earth's atmospheric pressure).
• Main constituents: N2 (> 97%), Ar, CH4, H2
• Oxidation state is reducing, but less so than that of the gas
giants: intermediate between inner planets & gas giants.
• Complex mix of hydrocarbons form due to photochemistry.
Somewhat analogous to Jupiter & Saturn's chemistry, but the
relative lack of H2 leads to higher yields of more complex and
also unsaturated hydrocarbons (with lower H:C ratio).
• Photochemistry of N2 (e.g. photodissociation, and
photoionization of N to N+) also plays a role in forming species
such as HCN, C2N2, HCCN
• Some very complex hydrocarbons (e.g. tholins, polycyclic
aromatics) also found: possible link to life?
• Hydrocarbons condense into aerosols, form coloured
(usually orange) clouds: photochemical smog!
• Liquid hydrocarbon lakes found near poles.
• Wikipedia: "The atmosphere is so thick and the
gravity so low that humans could fly through it by
flapping "wings" attached to their arms"
• Don't forget to wear breathing gear & warm clothes
though (T < 100 K)
8
• CO2 present in the atmosphere. This requires a
source of oxygen, as the reducing atmosphere would
otherwise convert CO2 into CO.
• Oxygen probably enters the atmosphere as water
from outside, e.g. meteorites or sputtering from
Saturn's rings or other satellites.
• "Chemistry of H2O, CO and CO2 may ultimately prove
to be as significant to reducing atmospheres as that
of the "trace constituent" reduced compounds (e.g.
CH4, H2) is to oxidizing ones.
•
Wayne pg 598
9
• Pluto, Triton, Callisto, Europa Io and some other of
the larger moons also have (small) atmospheres
• Io has a SO2 – dominated atmosphere, very strong
ionization due to proximity to Jupiter (strong magnetic
field). Io actually has a "torus" of gas encircling
Jupiter along it's orbit!
• Europa has more water than Earth, but frozen. May
have a small water-oxygen atmosphere.
• Comets also have a chemically rich and diverse
atmosphere, containing at least H2O, CO, nitrogen
compounds and hydrocarbons.
• Over 500 planets outside our solar system have been
discovered; likely at least some of these have
atmospheres.
10
11