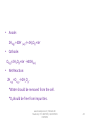* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chemistry-Unit-2-Battery-Technology-Cells-and-Battery
Survey
Document related concepts
Transcript
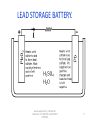
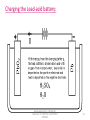
BATTERY TECHNOLOGY www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 1 Commercial Cells. Galvanic cells used as source of electric energy for various consumer, industrial and military applications. Classification: Primary Cells. Eg.: Dry cell Secondary Cells. Eg.: Lead acid cell. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 2 Objectives: Describe the major features of commercial cells. Know the two major types of batteries. Distinguish between primary & secondary battery types. Know the various applications of Dry cell, Nicad cell, Lead acid cell, H2-O2 fuel cell and CH3OH – O2 fuel cell. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 3 Basic Requirements Of Primary Cell. Compactness and lightweight. Fabricated from easily available raw materials. Economically priced. High energy density and constant voltage. Benign environmental properties Longer shelf life and discharge period. Leak proof containers and variety of design options. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 4 Basic Requirements Of Secondary Cell. Long shelf-life and cycle life. High power to weight ratio Short time for recharging Tolerance to service condition. High voltage & high energy density. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 5 Primary Cells. Produce electricity from chemicals that are sealed into it. Cannot be recharged as the cell reaction cannot be reversed efficiently by recharging. The cell must be discarded after discharging. e.g. Zinc - manganese dioxide cell (Dry cell) Mercuric oxide – Zinc cell. Silver oxide – zinc cell. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 6 Secondary Cells Generation of electric energy, that can be restored to its original charged condition after its discharge by passing current flowing in the opposite direction. These cells have a large number of cycles of discharging and charging. They are known as rechargeable cells, storage cells, or accumulators. e.g. Lead storage cell. Nickel- cadmium cell. Lithium- ion batteries. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 7 Differences Primary Batteries Cell reaction is irreversible Secondary Batteries Cell reaction is reversible. Must be discarded after use. May be recharged Have relatively short shelf life Have long shelf life. Function only as galvanic Functions both galvanic cells . Cell & as electrolytic cell. They cannot be used as They can be used as energy storage devices storage devices (e.g. solar/ thermal energy converted to electrical energy) They cannot be recharged They can be recharged. e.g. Dry cell. Li-MnO2battery.Lead acid, Ni-Cd battery. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 8 DRY CELL(LECLANCHE CELL) www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 9 • Anode: Zinc metal container. • Cathode: MnO2 + Carbon (powdered graphite) • Electrolyte: Aqueous paste of NH4Cl and ZnCl2 • Cell Scheme: Zn(s)/ ZnCl2(aq),NH4Cl(aq),MnO2(s)/C • O.C.V. = 1.5 V www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 10 Working. Primary Electrode Reactions: Anode: Zn(s)→Zn2+ (aq)+ 2eCathode: 2MnO2(s)+H2O(l) + 2e- → Mn2O3(s) + 2OH-(aq) Net Reaction: Zn(s)+2MnO2(s)+ H2O(l) → Zn2+(aq)+Mn2O3(s)+2OH-(aq) www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 11 Secondary Reactions: NH4+(aq)+OH-(aq) → NH3(g)+H2O(l) Zn2+(aq)+2NH3(s)+2Cl - → [Zn(NH3)2 Cl2] Zn + 2MnO2 + 2NH4Cl →[Zn(NH3)2Cl2]+ H2O+ Mn2O3 www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 12 Applications: In small portable appliances where small amount of current is needed. In consumer electronic devices- quartz wall clocks, walkman etc. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 13 Advantages. Dry cell is cheap. Normally works without leaking (leak proof cells). Has a high energy density. It is not toxic It contains no liquid electrolytes. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 14 Disadvantages. Voltage drops due to build up of reaction products around the electrodes when current is drawn rapidly from it . It has limited shelf life because the zinc is corroded by the faintly acid,ammonium chloride. The shelf life of dry cell is 6-8 months. They cannot be used once they get discharged. Its emf decreases during use as the material is consumed. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 15 Lead-acid battery: www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 16 LEAD STORAGE BATTERY. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 17 • Anode: Spongy lead on lead grid. • Cathode: Porous PbO2. • Electrolyte: H2SO4(aq)( 20 %) (density 1.21-1.30g/ml) • Cell Scheme: Pb/PbSO4;H2SO4(aq);PbSO4;PbO2/Pb O.C.V. = 2V (Pair of plates) www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 18 Reactions during discharging. • Anode: Pb (s) → Pb2+ (aq) + 2ePb2+(aq) + SO42-(aq) → PbSO4(s) Pb(s)+ SO42-(aq) → PbSO4(aq) + 2e• Cathode:PbO2(s)+ 4H+(aq)+2e- →Pb2+(aq)+ 2H2O(l) Pb2+(aq)+SO42-(aq)→PbSO4(s) PbO2(s)+4H+(aq)+SO42-(aq)+2e- → PbSO4(s)+ 2H2O(l) • Overall: Pb (s)+PbO2 (s)+4H+(aq)+ 2SO42-(aq) → 2PbSO4 (s)+2H2O(l) www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 19 Charging the Lead-acid battery: www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 20 Charging reactions • Cathode: PbSO4(s)+2H2O(l)→PbO2(s)+ SO42-(aq)+4H+(aq) +2e• Anode : PbSO4(s) + 2e- → Pb(s)+ SO42- (aq) • Net:2PbSO4 (s)+ 2H2O(aq) → Pb(s)+ PbO2(s) +2H2SO4 www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 21 Limitations. • Self discharge: They are subject to self discharge with H2 evolution at negative plates and O2 evolution at positive plates. Pb +H2SO4 PbSO4 + H2 PbO2 + H2SO4 PbSO4 +H2O +1/2 O2 SO42- +2 H+ (From dissociation of water) H2O H+ +OH- H2SO4 • Loss of Water: Due to evaporation, self discharge and electrolysis of water while charging. Hence water content must be regularly checked and distilled water must be added. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 22 • Sulfation: If left in uncharged state, for a prolonged period, or operated at too high temperatures or at too high acid concentrations, transformation of porous PbSO4 into dense and coarse grained form by re crystallization. * This results in passivation of negative plates inhibiting their charge acceptance. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 23 • Corrosion of Grid: Can occur due to overcharging when grid metal gets exposed to the electrolyte. This weakens the grid and increases the internal resistance of the battery. • Effectiveness of battery is reduced at low temperature due to increase in the viscosity of electrolyte. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 24 • Recent years have seen the introduction of “maintenance – free batteries” without a gas – release vent. Here the gassing is controlled by careful choice of the composition of the lead alloys used i.e. by using a Pb-Ca (0.1 % ) as the anode which inhibits the electrolysis of water • Alternatively, some modern batteries contain a catalyst (e.g. a mixture of 98% ceria (cerium oxide) & 2% platinum, heated to 1000o C) that combines the hydrogen and oxygen produced during discharge back into water. Thus the battery retains its potency and requires no maintenance. Such batteries are sealed as there is no need to add water and this sealing prevents leakage of cell materials. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 25 Applications. *Automative: For starting, lighting and ignition of IC engine driven vehicles. *Consumer Applications: Emergency lighting, security alarm system. *Heavy duty Application: Trains, lift trucks, mining machines etc. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 26 Advantages: A lead storage battery is highly efficient. The voltage efficiency of the cell is defined as follows. Voltage efficiency = average voltage during discharge average voltage during charge The voltage efficiency of the lead – acid cell is about 80 %. The near reversibility is a consequence of the faster rate of the chemical reactions in the cell i.e. anode oxidizes easily and cathode reduces easily leading to an overall reaction with a high negative free energy change. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 27 A lead – acid battery provides a good service for several years. Its larger versions can last 20 to 30 years, if carefully attended (i.e. longer design life) It can be recharged. The number of recharges possible range from 300 to 1500, depending on the battery’s design and conditions. The sealed leadacid batteries can withstand upto 2000 – rechargings. Generally the most costly, largest, heaviest cells are the longest–lived. The battery’s own internal self – discharging is low. The length of time that is generally required for recharging process is less i.e. recharge time is 2-8 hours depending on the status of battery. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 28 Low environmental impact of constituent materials is an added advantage It has sensitivity to rough handling and good safety characteristics. Ease of servicing as indicated by several local battery service points. It is a low- cost battery with facilities for manufacture throughout the world using cheap materials. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 29 NICKEL- CADMIUM CELL www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 30 Anode: Porous cadmium powder compressed to cylindrical pellets. Cathode: Ni(OH)3 or NiO(OH) mixed with 20% graphite powder Electrolyte: 20-28% Aq. KOH jelled with a jelling agent. Cell Scheme: Cd/Cd(OH)2,KOH,Ni(OH)2, Ni(OH)3/Ni O.C.V. = 1.25V www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 31 Reactions during discharging. Anode: Cd(s)+2OH-(aq)→Cd(OH)2(s)+ 2eCathode: 2Ni(OH)3(s)+2e- → 2Ni(OH)2(s)+2OH-(aq) • Net Reaction: Cd(s)+2Ni(OH)3(s)→ 2Ni(OH)2(s)+ Cd(OH)2(s) www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 32 Charging reactions: Anode: Cd(OH)2(s)+2e-→ Cd(s) +2OH-(aq) Cathode: 2Ni(OH)2(s) +2OH-(aq)→2Ni(OH)3(s)+2eNet: 2Ni(OH)2(s)+Cd(OH)2(s)→2Ni(OH)3(s)+ Cd(s) www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 33 Discharging reaction: Anode: Cd(s)+2OH-(aq) → Cd(OH)2(s) + 2e- Cathode: 2NiO (OH) (s) + 2 H2O + 2 e- → 2Ni (OH)2(s) + 2OH-(aq) Net Reaction: Cd(s) + 2NiO (OH) (s) + 2H2O → 2 Ni(OH)2 (s) + Cd(OH)2(s) www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 34 Charging reactions: -ve pole: Cd(OH)2 (s) + 2e-→ Cd(s) + 2OH-(aq) +ve pole: 2 Ni(OH)2(s) + 2OH-(aq) → 2 NiO(OH) (s) + 2H2O+2eOverall reaction: 2 Ni(OH)2 (s) + Cd(OH)2(s) → 2 NiO(OH) (s) + Cd(s) +2H2O(l) www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 35 Applications. In flash lights, photoflash units and portable electronic equipments. In emergency lighting systems, alarm systems. In air crafts and space satellite power systems. For starting large diesel engines and gas turbines etc., www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 36 Advantages. Can be recharged many times. They maintain nearly constant voltage level throught their discharge. There is no change in the electrolyte composition during the operation. It can be left unused for long periods of time at any state of charge without any appreciable damage (i.e. long shelf life). It can be encased as a sealed unit like the dry cell because gassing will not occur during nominal discharging or recharging. They exhibit good performance ability at low temperatures. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 37 They can be used to produce large instantaneous currents as high as 1000-8000 A for one second. It is a compact rechargeable cell available in three basic configurations – button, cylindrical and rectangular. They have low internal resistance. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 38 Disadvantages. It poses an environmental pollution hazard due to higher toxicity of metallic cadmium than lead. Cadmium is a heavy metal and its use increases the weight of batteries, particularly in larger versions. Cost of cadmium metal and hence the cost of construction of NiCad batteries is high. The KOH electrolyte used is a corrosive hazardous chemical. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 39 Fuel Cells. A fuel cell is a galvanic cell in which chemical energy of a fuel – oxidant system is converted directly into electrical energy in a continuous electrochemical process. • Cell Schematic Representation: Fuel;electrode/electrolyte/electrode/oxidant. e.g. H2-O2; CH3OH-O2 www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 40 • The reactants (i.e. fuel + oxidant) are constantly supplied from outside and the products are removed at the same rate as they are formed. • Anode: Fuel+ oxygen → Oxidation products+ ne• Cathode: Oxidant + ne- → Reduction products. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 41 Requirements Of Fuel Cell. • Electrodes: Must be stable, porous and good conductor. • Catalyst: Porous electrode must be impregnated with catalyst like Pt, Pd, Ag or Ni, to enhance otherwise slow electrochemical reactions. • Optimum Temperature: Optimum. • Electrolyte: Fairly concentrated. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 42 Hydrogen – Oxygen Fuel Cell www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 43 • Anode: Porous graphite electrodes impregnated with finely divided Pt/Pd. • Cathode: Porous graphite electrodes impregnated with finely divided Pt/Pd. • Electrolyte: 35-50% KOH held in asbestos matrix. • Operating Temperature: 90oC. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 44 • Anode : 2H2(g) +40H- (aq)→ 4H2O(l)+4e• Cathode: O2(g)+2H2O(l)+4e- →4OH(aq) • Net Reaction: 2H2(g)+O2(g)→2H2O(l). *Water should be removed from the cell. *O2should be free from impurities. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 45 Applications. • Used as energy source in space shuttles e.g. Apollo spacecraft. • Used in small- scale applications in submarines and other military vehicles. • Suitable in places where, environmental pollution and noise are objectionable. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 46 CH3OH-O2 Fuel Cell • Both electrodes: Made of porous nickel plates impregnated with finely- divided Platinum. • Fuel: Methyl alcohol. • Oxidant: Pure oxygen / air. • Electrolyte: Conc.Phosphoric acid/Aq.KOH • Operating Temperature: 150-200oC. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 47 • The emf of the cell is 1.20 V at 25oC. • MeOH is one of the most electro active organic fuels in the low temperature range as *It has a low carbon content *It posseses a readily oxidizable OH group *It is miscible in all proportions in aqueous electrolytes. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 48 • At anode: CH3OH + 6OH- →CO2 + 5H2O + 6e• At cathode: 3/2 O2 +3H2O + 6e- →6OHNet Reaction: CH3OH +3/2O2 →CO2 + 2H2O. It is used in millitary applications and in large scale power production. It has been used to power television relay stations. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 49 Advantages Of Fuel Cells. • High efficiency of the energy conversion process. • Silent operation. • No moving parts and so elimination of wear and tear. • Absence of harmful waste products. • No need of charging. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 50 Limitations Of Fuel Cells. • Cost of power is high as a result of the cost of electrodes. • Fuels in the form of gases and O2 need to be stored in tanks under high pressure. • Power output is moderate. • They are sensitive to fuel contaminants such as CO,H2S, NH3 & halides, depending on the type of fuel cell. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 51 Differences. • Fuel Cell Galvanic Cell *Do not store chemical Stores chemical energy energy *Reactants are fed from The reactants form an outside continuously. integral part of it. *Need expensive noble These conditions are metal catalysts. not required *No need of charging Get-discharged when stored – up energy is exhausted. *Never become dead Limited life span in use *Useful for long-term Useful as portable power services electricity generation. www.bookspar.com | Website for Students | VTU NOTES | QUESTION PAPERS 52