Ni recovery using KOH, NaOH, and NH4OH in the presence of
... first. A significant amount of iron is also co-leached, which in turn is removed by precipitation as ammonium jarosite. Substantial amounts of nickel have been found entrained in this precipitate, which is discarded. An attempt was made to optimally recover the entrained nickel, using acidic dissolu ...
... first. A significant amount of iron is also co-leached, which in turn is removed by precipitation as ammonium jarosite. Substantial amounts of nickel have been found entrained in this precipitate, which is discarded. An attempt was made to optimally recover the entrained nickel, using acidic dissolu ...
ION-SELECTIVE ELECTRODES - Clayton State University
... - The study of the interconversion of chemical energy and electrical energy - The study of redox reactions - Electrochemical processes involve the transfer of electrons from one substance to another ...
... - The study of the interconversion of chemical energy and electrical energy - The study of redox reactions - Electrochemical processes involve the transfer of electrons from one substance to another ...
industry: applying chemical reactions
... empty Riverwood Corportion building into a chemical manufacturing plant in Riverwood. Because of a lack of employment opportunities in Riverwood, many of your friends and neighbors have expressed relief at this news from EKS Nitrogen Products Company and WYE Battery Technology Corporation.
... empty Riverwood Corportion building into a chemical manufacturing plant in Riverwood. Because of a lack of employment opportunities in Riverwood, many of your friends and neighbors have expressed relief at this news from EKS Nitrogen Products Company and WYE Battery Technology Corporation.
CHAPtER 4 Electrolysis
... The anode in an electrolytic cell is positive since the DC source withdraws electrons from it. Electrons are supplied to the positive anode by the oxidation of the copper electrode itself. In an electrolytic cell, the polarity of the electrodes is determined by the external DC source. In contrast, t ...
... The anode in an electrolytic cell is positive since the DC source withdraws electrons from it. Electrons are supplied to the positive anode by the oxidation of the copper electrode itself. In an electrolytic cell, the polarity of the electrodes is determined by the external DC source. In contrast, t ...
Mnemonic Devices - Free WonderKids-e
... (2) Reactions of Chromium (VI) When chromium is in the +6 oxidation state, it forms different compounds depending on the pH of the solution. (a) In neutral or basic solutions the chromate ion, CrO42 − , predominates. (b) In acidic solutions the dichromate ion predominates: 2CrO 42− + 2 H + → Cr2 O72 ...
... (2) Reactions of Chromium (VI) When chromium is in the +6 oxidation state, it forms different compounds depending on the pH of the solution. (a) In neutral or basic solutions the chromate ion, CrO42 − , predominates. (b) In acidic solutions the dichromate ion predominates: 2CrO 42− + 2 H + → Cr2 O72 ...
electrochemistry
... exothermic reaction is normally lost as heat. However, it can be trapped and converted into electrical energy if the reactants involved are not in direct contact with each other. In galvanic cells redox reactions is split into two halfreactions, each occurring in two separate compartments, called ha ...
... exothermic reaction is normally lost as heat. However, it can be trapped and converted into electrical energy if the reactants involved are not in direct contact with each other. In galvanic cells redox reactions is split into two halfreactions, each occurring in two separate compartments, called ha ...
Chemistry-Unit-2-Battery-Technology-Cells-and-Battery
... battery’s design and conditions. The sealed leadacid batteries can withstand upto 2000 – rechargings. Generally the most costly, largest, heaviest cells are the longest–lived. The battery’s own internal self – discharging is low. The length of time that is generally required for recharging proce ...
... battery’s design and conditions. The sealed leadacid batteries can withstand upto 2000 – rechargings. Generally the most costly, largest, heaviest cells are the longest–lived. The battery’s own internal self – discharging is low. The length of time that is generally required for recharging proce ...
Electrochemistry
... more difficult (require more work) to introduce additional Zn2+ ions into the positivelycharged electrolyte or for electrons to flow into right compartment where they are needed to reduce the Cu2+ ions, thus effectively stopping the reaction after only a minute amount has taken place. In order to su ...
... more difficult (require more work) to introduce additional Zn2+ ions into the positivelycharged electrolyte or for electrons to flow into right compartment where they are needed to reduce the Cu2+ ions, thus effectively stopping the reaction after only a minute amount has taken place. In order to su ...
Sec 5.8 - 5.11 notes
... 3) Electrons are ________ at the anode. 4) _________ is the electrode where reduction occurs. 5) In the half-rx at the cathode, e-‘s are on the ______ side of the equation. 6) Electrons flow from the ______ toward the ______ in the __________. 7) The higher half-rx on the table is the one for the __ ...
... 3) Electrons are ________ at the anode. 4) _________ is the electrode where reduction occurs. 5) In the half-rx at the cathode, e-‘s are on the ______ side of the equation. 6) Electrons flow from the ______ toward the ______ in the __________. 7) The higher half-rx on the table is the one for the __ ...
Oxidation-Reduction and Electrochemistry
... Faraday also defined a number of terms: The anode is therefore that surface at which the electric current, according to our present expression, enters: it is the negative extremity of the decomposing body; is where oxygen, chlorine, acids, etc., are evolved; and is against or opposite the positiv ...
... Faraday also defined a number of terms: The anode is therefore that surface at which the electric current, according to our present expression, enters: it is the negative extremity of the decomposing body; is where oxygen, chlorine, acids, etc., are evolved; and is against or opposite the positiv ...
Electrode Potentials hw - A
... Large blocks of magnesium are bolted onto the hulls of iron ships in an attempt to prevent the iron being converted into iron(II), one of the steps in the rusting process. Use the data below, where appropriate, to answer the questions which follow. ...
... Large blocks of magnesium are bolted onto the hulls of iron ships in an attempt to prevent the iron being converted into iron(II), one of the steps in the rusting process. Use the data below, where appropriate, to answer the questions which follow. ...
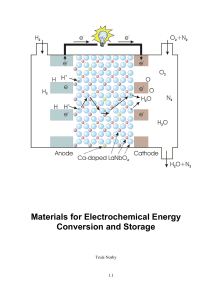
Materials for Electrochemical Energy Conversion and Storage
... alkaline cell, Zn(s) + 2MnO2 + 2H2O = Zn2+ + 2MnOOH + 2 OH- ...
... alkaline cell, Zn(s) + 2MnO2 + 2H2O = Zn2+ + 2MnOOH + 2 OH- ...
Hard water:
... The electrolyte which ionize to a small extent on dilution are called weak electrolyte. They have a low value of equivalent conductance even at a higher concentration and are not completely ionized even at very great dilution. Weak Acids: All organics acids like acetic acids, propionic acid and H2SO ...
... The electrolyte which ionize to a small extent on dilution are called weak electrolyte. They have a low value of equivalent conductance even at a higher concentration and are not completely ionized even at very great dilution. Weak Acids: All organics acids like acetic acids, propionic acid and H2SO ...
OXIDATION NUMBERS
... An electrode is half a cell. If two different electrodes are connected together, an electrochemical cell is formed. Usually, two electrode compartments are joined by a salt bridge. A salt bridge allows ions to flow, thus completing the electrical circuit, but prevents the ions in the two electrode c ...
... An electrode is half a cell. If two different electrodes are connected together, an electrochemical cell is formed. Usually, two electrode compartments are joined by a salt bridge. A salt bridge allows ions to flow, thus completing the electrical circuit, but prevents the ions in the two electrode c ...
Electrochemical Fundamentals
... The amount of energy stored per unit weight is considerably lower than that of an electrochemical battery (3-5 W·h/kg for an ultracapacitor compared to 30-40 W·h/kg for a lead acid battery). It is also only about 1/10,000th the volumetric energy density of gasoline. ...
... The amount of energy stored per unit weight is considerably lower than that of an electrochemical battery (3-5 W·h/kg for an ultracapacitor compared to 30-40 W·h/kg for a lead acid battery). It is also only about 1/10,000th the volumetric energy density of gasoline. ...
Chapter 20: Electrochemistry
... Stoichiometry & Electrolysis Lithium is a very active metal, and can be formed through the electrolysis of LiCl. What mass of Lithium and chlorine can be formed through the electrolysis of molten LiCl if 4300.0 amps passes through a cell operating for 24.0 hours? ...
... Stoichiometry & Electrolysis Lithium is a very active metal, and can be formed through the electrolysis of LiCl. What mass of Lithium and chlorine can be formed through the electrolysis of molten LiCl if 4300.0 amps passes through a cell operating for 24.0 hours? ...
ch19 MSJ jlm
... The Ag|Ag+ half-cell has a higher reduction potential than the Cu|Cu2+ half-cell. This means that the Ag/Ag+ half-cell will more readily undergo reduction when compared to the Cu/Cu2+ halfcell, and the Cu|Cu2+ half-cell will undergo oxidation. The Ag|Ag+ standard reduction potential is Eored = +0.8 ...
... The Ag|Ag+ half-cell has a higher reduction potential than the Cu|Cu2+ half-cell. This means that the Ag/Ag+ half-cell will more readily undergo reduction when compared to the Cu/Cu2+ halfcell, and the Cu|Cu2+ half-cell will undergo oxidation. The Ag|Ag+ standard reduction potential is Eored = +0.8 ...
CHAPTER 8: ENERGY FROM ELECTRON TRANSFER
... The efficiency is reduced because some of the radiant energy is reflected by the cell or absorbed to produce heat instead of an electric current. Concentrating on Concepts 29. Explain the significance of the title of this chapter, “Energy from Electron Transfer.” Answer: In every electrochemical pro ...
... The efficiency is reduced because some of the radiant energy is reflected by the cell or absorbed to produce heat instead of an electric current. Concentrating on Concepts 29. Explain the significance of the title of this chapter, “Energy from Electron Transfer.” Answer: In every electrochemical pro ...
Document
... That's one way of thinking of the electrical circuit part of the electrochemical cell. The electrons pushed around the external circuit can do work (run a motor, illuminate a light bulb, etc). The amount of work possible is a function of both the voltage (potential) and of the current (number of ele ...
... That's one way of thinking of the electrical circuit part of the electrochemical cell. The electrons pushed around the external circuit can do work (run a motor, illuminate a light bulb, etc). The amount of work possible is a function of both the voltage (potential) and of the current (number of ele ...
Redox Reactions and Electrochemistry
... • But the voltage for producing O2(g) from solution is considerably higher than the standard potential, because of the high activation energy needed to form O2(g). • The voltage for this half cell seems to be closer to –1.5 V in reality. • The result then is the production of Cl2(g) and Cu(s). an ...
... • But the voltage for producing O2(g) from solution is considerably higher than the standard potential, because of the high activation energy needed to form O2(g). • The voltage for this half cell seems to be closer to –1.5 V in reality. • The result then is the production of Cl2(g) and Cu(s). an ...
LATENT HEAT AND ELECTRODE POTENTIAL
... come greater than X. Such results are contrary both to experience and to the principles of thermodynamics. Theory (7) and experiment indicate that at the mp of M, the liquid and solid forms of M must have the same potential. Although this potential, or the emf, E, of a cell of which M forms one elec ...
... come greater than X. Such results are contrary both to experience and to the principles of thermodynamics. Theory (7) and experiment indicate that at the mp of M, the liquid and solid forms of M must have the same potential. Although this potential, or the emf, E, of a cell of which M forms one elec ...
Jahn-Teller assisted Na diffusion for high performance Na ion batteries
... interslab distance can shrink from 5.7 Å at the beginning of the high-voltage O3 phase to as low as 4.8 Å at the end of charge. This process is largely reversible, and the interslab distance expands back upon discharge, indicating good Na diffusivity even at very low Na slab thicknesses. The reversib ...
... interslab distance can shrink from 5.7 Å at the beginning of the high-voltage O3 phase to as low as 4.8 Å at the end of charge. This process is largely reversible, and the interslab distance expands back upon discharge, indicating good Na diffusivity even at very low Na slab thicknesses. The reversib ...
Batteries are all over the place -- in our cars, our
... Batteries are all over the place -- in our cars, our PCs, laptops, portable MP3 players and cell phones. A battery is essentially a can full of chemicals that produce electrons. Chemical reactions that produce electrons are called electrochemical reactions. In this article, you'll learn all about ba ...
... Batteries are all over the place -- in our cars, our PCs, laptops, portable MP3 players and cell phones. A battery is essentially a can full of chemicals that produce electrons. Chemical reactions that produce electrons are called electrochemical reactions. In this article, you'll learn all about ba ...
AP* Chemistry ELECTROCHEMISTRY Terms to Know
... in salt water acts as the anode and is oxidized instead of the steel hull extending the life of the vessel. Additionally, Titanium is also immune to microbiologically influenced corrosion. ...
... in salt water acts as the anode and is oxidized instead of the steel hull extending the life of the vessel. Additionally, Titanium is also immune to microbiologically influenced corrosion. ...
Nickel–metal hydride battery

A nickel–metal hydride battery, abbreviated NiMH or Ni–MH, is a type of rechargeable battery. The chemical reaction at the positive electrode is similar to that of the nickel–cadmium cell (NiCd), with both using nickel oxyhydroxide (NiOOH). However, the negative electrodes use a hydrogen-absorbing alloy instead of cadmium. A NiMH battery can have two to three times the capacity of an equivalent size NiCd, and its energy density can approach that of a lithium-ion battery.