* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
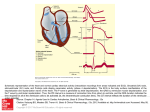
Download Early repolarization in the inferolateral leads of the
Survey
Document related concepts
Heart failure wikipedia , lookup
Saturated fat and cardiovascular disease wikipedia , lookup
Cardiovascular disease wikipedia , lookup
Remote ischemic conditioning wikipedia , lookup
Jatene procedure wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Cardiac surgery wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Coronary artery disease wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Heart arrhythmia wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Transcript
D 1212 OULU 2013 UNIV ER S IT Y OF OULU P. O. B R[ 00 FI-90014 UNIVERSITY OF OULU FINLAND U N I V E R S I TAT I S S E R I E S SCIENTIAE RERUM NATURALIUM Professor Esa Hohtola HUMANIORA University Lecturer Santeri Palviainen TECHNICA Postdoctoral research fellow Sanna Taskila MEDICA Professor Olli Vuolteenaho ACTA EARLY REPOLARIZATION IN THE INFEROLATERAL LEADS OF THE ELECTROCARDIOGRAM PREVALENCE, PROGNOSIS AND CHARACTERISTICS SCIENTIAE RERUM SOCIALIUM University Lecturer Hannu Heikkinen SCRIPTA ACADEMICA Director Sinikka Eskelinen OECONOMICA Professor Jari Juga EDITOR IN CHIEF Professor Olli Vuolteenaho PUBLICATIONS EDITOR Publications Editor Kirsti Nurkkala ISBN 978-952-62-0230-3 (Paperback) ISBN 978-952-62-0231-0 (PDF) ISSN 0355-3221 (Print) ISSN 1796-2234 (Online) U N I V E R S I T AT I S O U L U E N S I S Jani Tikkanen E D I T O R S Jani Tikkanen A B C D E F G O U L U E N S I S ACTA A C TA D 1212 UNIVERSITY OF OULU GRADUATE SCHOOL; UNIVERSITY OF OULU, FACULTY OF MEDICINE, INSTITUTE OF CLINICAL MEDICINE, DEPARTMENT OF INTERNAL MEDICINE; OULU UNIVERSITY HOSPITAL D MEDICA ACTA UNIVERSITATIS OULUENSIS D Medica 1212 JANI TIKKANEN EARLY REPOLARIZATION IN THE INFEROLATERAL LEADS OF THE ELECTROCARDIOGRAM Prevalence, prognosis and characteristics Academic dissertation to be presented with the assent of the Doctoral Training Committee of Health and Biosciences of the University of Oulu for public defence in Auditorium 10 of Oulu University Hospital, on 19 October 2013, at 12 noon U N I VE R S I T Y O F O U L U , O U L U 2 0 1 3 Copyright © 2013 Acta Univ. Oul. D 1212, 2013 Supervised by Professor Heikki Huikuri Docent Juhani Junttila Docent Olli Anttonen Reviewed by Docent Vesa Virtanen Docent Antti Ylitalo Opponent Doctor Sami Viskin ISBN 978-952-62-0230-3 (Paperback) ISBN 978-952-62-0231-0 (PDF) ISSN 0355-3221 (Printed) ISSN 1796-2234 (Online) Cover Design Raimo Ahonen JUVENES PRINT TAMPERE 2013 Tikkanen, Jani, Early repolarization in the inferolateral leads of the electrocardiogram : Prevalence, prognosis and characteristics University of Oulu Graduate School; University of Oulu, Faculty of Medicine, Institute of Clinical Medicine, Department of Internal Medicine; Oulu University Hospital Acta Univ. Oul. D 1212, 2013 University of Oulu, P.O. Box 8000, FI-90014 University of Oulu, Finland Abstract “Early repolarization” (ER), consisting of J waves and ST-segment elevation in the standard 12lead electrocardiogram (ECG), has been considered a benign finding for over 60 years until 2008 an over-presentation of ER ECG pattern in infero-lateral leads was described in patients with idiopathic ventricular fibrillation. The purpose of this thesis was to assess the prevalence and prognostic significance, and to characterize the clinical features of this ECG pattern primarily in the general population. This thesis investigated the electrocardiographic features of a total of 20,308 general population individuals, 565 athletes, 432 victims of sudden cardiac death and 532 survivors of an acute coronary event. The overall prevalence of infero-lateral ER ECG pattern in the general population samples was in range between 3.3 and 6.1 percent. ER seemed to have a heritable basis as siblings of individuals displaying the ER pattern had significantly increased odds for presenting this ECG pattern. Several strong associations between ER pattern and clinical factors were observed in all populations. The ER patterns with horizontal or descending ST-segments were associated with significantly increased arrhythmic mortality during a follow-up of 30±11 years, and high amplitude (>0.2mV) J waves in the inferior leads were associated with a high risk of arrhythmic death (multivariate adjusted hazard ratio 3.13 (95% confidence interval 1.55 to 6.32). The ER ECG pattern was also independently associated with sudden cardiac death during an acute coronary event, with an odds ratio of 2.02 (95% CI 1.04 to 3.61) for suffering sudden death. The ER patterns with rapidly ascending ST-segments, the dominant type of ER ECG in young, healthy individuals, were not associated with any adverse outcomes. A multicentre meta-analysis of genome wide association study of ER ECG pattern performed in a total of 14,633 individuals pointed to an associated locus in KCND3 (Kv4.3) gene, which encodes a subunit of the Ito channel, but the association did not reach genome-wide significance and could not be replicated in all study populations. In conclusion, the results of these studies demonstrated that the ER ECG pattern with horizontal or descending ST-segments, especially in the inferior leads, is associated with an increased risk of sudden arrhythmic death in the general population, and that individuals carrying this ECG pattern are at increased risk of suffering fatal arrhythmia during an acute coronary event. The ER pattern with rapidly ascending ST-segments is common in young healthy athletes and is not associated with any increased risk of fatal events. Although the ER ECG pattern increases the relative risk of arrhythmic events, the absolute risk in an individual is low and therefore this ECG pattern should not be interpreted as a high-risk marker of mortality. The genetic background of this ECG pattern remains to be clarified. Keywords: arrhythmia, early repolarization, electrocardiography, mortality, sudden cardiac death Tikkanen, Jani, Varhainen repolarisaatio sydänfilmissä. Oulun yliopiston tutkijakoulu; Oulun yliopisto, Lääketieteellinen tiedekunta, Kliinisen lääketieteen laitos, Sisätaudit; Oulun yliopistollinen sairaala Acta Univ. Oul. D 1212, 2013 Oulun yliopisto, PL 8000, 90014 Oulun yliopisto Tiivistelmä Normaalissa 12-kytkentäisessä sydänfilmissä (EKG) J-aalloista ja ST-segmentin noususta koostuvaa varhaista repolarisaatiota (ER) pidettiin yli 60 vuotta hyvänlaatuisena muutoksena, kunnes vuonna 2008 ER esiintyminen alaseinä-lateraalikytkennöissä liitettiin idiopaattiseen kammiovärinään. Tämän väitöstutkimuksen tarkoitus oli selvittää EKG-muutoksen esiintyvyys ja ennustearvo normaaliväestössä, sekä selvittää sen kliinisiä erityispiirteitä. Väitöstutkimusta varten tutkittiin 20,308 normaaliväestön henkilöä, 565 urheilijaa, 432 äkillisesti (sydänperäisesti) menehtynyttä henkilöä ja 532 akuutista sepelvaltimotautikohtauksesta selviytynyttä potilasta. ER-EKG:n kokonaisesiintyvyys alaseinä-lateraalikytkennöissä normaaliväestön otoksissa vaihteli 3.3 % ja 6.1 % välillä. ER-EKG:n esiintyessä merkittävästi muita useammin sisaruksilla ER-EKG vaikutti periytyvältä muutokselta, ja lisäksi kaikissa otoksissa EREKG liittyi useisiin muihin tunnettuihin kliinisiin muuttujiin. ER-EKG, ts. J-aallot, joita seurasivat horisontaalinen tai laskeva ST-segmentti, liittyivät merkittävästi lisääntyneeseen rytmihäiriökuolleisuuteen pitkän seuranta-ajan (30±11 vuotta) aikana, ja korkea-amplitudiset (>0.2mV) Jaallot alaseinäkytkennöissä liittyivät korkeaan riskiin kuolla äkillisesti rytmihäiriöön (monimuuttuja-vakioitu riskisuhde 3.13, 95 % luottamusväli 1.55–6.32). ER-EKG lisäsi lisäksi itsenäisesti sydänperäisen äkkikuoleman riskiä akuutin sepelvaltimotautikohtauksen aikana (vetosuhde 2.02, 95 % luottamusväli 1.04–3.61). Etenkin nuorilla ja terveillä urheilijoilla esiintynyt EKG tyyppi, J-aallot ST-segmentin nousujen yhteydessä, ei liittynyt huonoon ennusteeseen. Monikeskusmeta-analyysi perimänlaajuisista assosiaatiotutkimuksista (GWAS) 14,633 henkilöllä antoi viitteitä geenimuutoksesta KCND3, joka koodittaa osaa Ito kanavasta, mutta löydöstä ei kyetty toistamaan kaikissa tutkituissa aineistoissa. Väitöstutkimuksen osatöiden perusteella ER-EKG muutos ilman ST-segmentin nousua, erityisesti alaseinäkytkennöissä esiintyessään, liittyy normaaliväestössä lisääntyneeseen riskiin sydänperäiselle rytmihäiriökuolemalle, ja että henkilöillä, joilla muutos esiintyy, on lisääntynyt riski menehtyä äkillisesti sepelvaltimotautikohtauksen aikana. ER-EKG ST-segmentin nousuihin liittyen on yleinen löydös nuorilla terveillä urheilijoilla ja se ei näytä liittyvän lisääntyneeseen riskiin rytmihöiriöille. Vaikka ER-EKG lisääkin suhteellista riskiä rytmihäiriöille ja ennenaikaiselle kuolemalle väestötasolla, absoluuttinen riski on pieni, eikä muutosta tulisi luokitella suureksi riskitekijäksi. Löydöksen geneettinen tausta on vielä avoin kysymys. Asiasanat: EKG, kuolleisuus, rytmihäiriö, sydänperäinen äkkikuolema, varhainen repolarisaatio To my family 8 Acknowledgements This thesis was carried out in the Department of Internal Medicine, University of Oulu, Finland during the years 2008-2012. In part, the studies were carried out in the following collaborative institutions: National Institute for Health and Welfare, Helsinki, Finland; Broad Institute of Harvard and MIT, Cambridge, Massachusetts, USA; Massachusetts General Hospital, Boston, Massachusetts USA. My deepest and most sincere thanks go to my supervisor and mentor Professor Heikki Huikuri, whose outstanding clinical knowledge and vision in cardiac electrophysiology and sudden cardiac death research are internationally highly recognized. Heikki has delivered an exceptionally inspiring athmosphere for his research group, and without his optimism and innovativeness but also criticism throughout this project, the outcome of this thesis could not have been as good as it was. Even as a novise medical student I always felt as a member of a larger family, and never had any issues in expressing my ideas and questions. I was privileged to have a supervisor who was actively engaged in this work and gave me excellent support throughout the project. I am also very grateful to my co-supervisors and dear colleagues Docent Juhani Junttila and Docent Olli Anttonen, whose never-ending enthusiasm and optimism towards our research projects encouraged me to pursue even higher and more descriptive results, not to ignore all their support and understanding of personal life in general. I’m very thankful for my friendship with Juhani and his family. I want to thank all the members of my follow-up group, Professor Timo Mäkikallio, Docent Juha Perkiömäki and Mirja Peltola. Particularly I’d like to thank Timo for his warm personal appearance and his guidance in both science and life in general. I want to express my warmest thanks to all my co-authors, who all cannot be listed here, but who certainly all are acknowledged. I’ve been privilidged to have such wonderful people to work with. Special thanks go to Tuomas Kenttä for both all his help in technology but also friendship outside work. I’d also like to thank Harri Rissanen for never rejecting my continuous suggestions for new statistical analyses and for his supportive thoughts on the results. I owe my thanks also to Aapo Aro for his input in data gathering and our fluent collaboration. I warmly thank Kimmo Porthan not only for his efforts towards this thesis but also for our friendship and fluent cooperation throughout the past few years. I also wish to acknowledge my debt to my colleagues in Boston, especially Peter Noseworthy and Christopher NewtonCheh, for their expertise and sincerity. I also highly acknowledge and appreciate the 9 input of Antti Reunanen and Veikko Salomaa from Institute of Health and Welfare in gathering and organizing our unique population cohorts. I appreciate and thank the official pre-examiners of this dissertation, Antti Ylitalo and Vesa Kivinen, for detailed commentary and constructive criticism. I greatly acknowledge the financial support of this thesis provided by the Finnish Foundation for Cardiovascular Research, the Finnish Medical Foundation, the Aarne Koskelo Foundation, the Emil Aaltonen Foundation, the Finnish Culural Foundation, the Paavo Nurmi Foundation, the Orion-Farmos Foundation, the Ida Montin Foundation, the Aarne and Aili Turunen Foundation, and the Maud Kuistila Memorial Foundation. Without these grants, the research work this magnitude and all the congresses around the globe would never have been possible. I wish to thank all my colleagues and also friends outside medicine for all the great memorable moments and support. I also want to express my gratitude to my family in law for all the support during the past few years. They've always had the patience to listen to the latest turns in my work and encouraged to keep on going, not to mention all their essential help in everyday life. I wish to express my deepest appreciation to my parents Pertti and Mirja, and my sister Tiia, for their enormous care and support throughout my life. My dad, a nuclear physicist and scientist himself, and my mom, a teacher of natural science, have always been exceptional role models for me, and with my sister we’ve experienced a lot of happiness in our childhood. You all have provided me with a solid foundation with your trust and unconditional love. I owe you everything I've ever achieved in my life. Above all, my warmest thoughts and gratitude go to my lovely wife Johanna and our wonderful sons Väinö and Veikko. I know I've spent too much time struggling with scientific work or practising medicine during late hours. Johanna, you have taken care of our children and home so incredibly well, and words cannot describe how grateful I am for your patience and all the emotional support. I would be nothing without you. I love you. Oulu, May 2013 10 Jani Tikkanen Abbrevations ARVD BMI BrS CAD CVD CI CPVT ECG ER ERP ERS EU fQRS GWA(S) HR ICD IVF LBBB LQTS LVEF N/A QRSd QTc OR RBBB RR SAECG SCD SNP(s) SQTS STEMI VF arrhythmogenic right ventricular dysplasia body mass index Brugada syndrome coronary artery disease cardiovascular disease confidence interval catecholaminergic polymorphic ventricular tachycardia electrocardiogra/m, -hic –phy early repolarization early repolarization ECG pattern early repolarization syndrome European Union fragmented QRS complex genome wide association (stud/y, -ies) hazard ratio implatable cardioverter defibrillator idiopathic ventricular fibrillation left bundle branch block long QT syndrome left ventricular ejection fraction not applicable Nonspecific QRS complex prolongation QT interval corrected for heart rate odds ratio right bundle branch block relative risk signal averaged electrocardiogram sudden cardiac death single nucleotide polymorphism(s) short QT syndrome ST-elevation myocardial infarction ventricular fibrillation 11 12 List of original articles This thesis is based on the following original articles, which are referred to in the text by their Roman numerals. I Tikkanen JT, Anttonen O, Junttila MJ, Aro AL, Kerola T, Rissanen HA, Reunanen A & Huikuri HV (2009) Long-term outcome associated with early repolarization on electrocardiography. N Engl J Med 361: 2529–2537 II Noseworthy PA*, Tikkanen JT*, Porthan K*, Oikarinen L, Pietilä A, Harald K, Peloso GM, Merchant FM, Jula A, Väänänen H, Hwang SJ, O’Donnel CJ, Salomaa V, Newton-Cheh C & Huikuri HV (2011) The early repolarization pattern in the general population: clinical correlates and heritability. J Am Coll Cardiol 57: 2284–2289 III Tikkanen JT, Junttila MJ, Anttonen O, Aro AL, Luttinen S, Kerola T, Sager SJ, Rissanen HA, Myerburg RJ, Reunanen A & Huikuri HV (2011) Early repolarization: electrocardiographic phenotypes associated with favorable long-term outcome. Circulation 123: 2666–2673 IV Tikkanen JT, Wichmann V, Junttila MJ, Rainio M, Hookana E, Lappi O, Kortelainen ML, Anttonen O & Huikuri HV (2012) Association of early repolarization and sudden cardiac death during an acute coronary event. Circ Arrh Electrophysiol 5: 714–718. V Sinner MF*, Porthan K*, Noseworthy PA*, Havulinna AS*, Tikkanen JT*, MüllerNurasyid M, Peloso G, Ulivi S, Beckmann BM, Brockhaus AC, Cooper RR, Gasparini P, Hengstenberg C, Hwang SJ, Iorio A, Junttila MJ, Klopp N, Kähönen M, Laaksonen MA, Lehtimäki T, Lichtner P, Lyytikäinen LP, Martens E, Meisinger C, Meitinger T, Merchant FM, Nieminen MS, Peters A, Pietilä A, Perz S, Oikarinen L, Raitakari O, Reinhard W, Silander K, Thorand B, Wichmann HE, Sinagra G, Viikari J, O'Donnell CJ, Ellinor PT, Huikuri HV, Kääb S, Newton-Cheh C & Salomaa V (2012) A metaanalysis of genome-wide association studies of the electrocardiographic early repolarization pattern. Heart Rhythm 9: 1627–1634. *Equal authorship 13 14 Contents Acknowledgements 9 Abbrevations 11 List of original articles 13 Contents 15 1 Introduction 19 2 Review of the literature 21 2.1 Basic electrophysiology and electrocardiography................................... 21 2.1.1 Cardiac action potential ................................................................ 21 2.1.2 Normal electric cycle in the ECG ................................................. 22 2.1.3 Cellular basis of electrocardiographic J waves ............................. 23 2.1.4 Electrocardiography as an evaluation tool.................................... 27 2.2 Cardiovascular morbidity and mortality ................................................. 27 2.2.1 Sudden unexplained death in the community ............................... 28 2.3 Cardiomyopathies ................................................................................... 31 2.3.1 Hypertrophic cardiomyopathy ...................................................... 31 2.3.2 Dilated cardiomyopathy ............................................................... 32 2.3.3 Arrhythmogenic right ventricular dysplasia ................................. 32 2.3.4 Myocardial fibrosis....................................................................... 33 2.4 Channelopathies ...................................................................................... 34 2.4.1 Long QT syndrome....................................................................... 35 2.4.2 Short QT syndrome ...................................................................... 36 2.4.3 Catecholaminergic polymorphic ventricular tachycardia ............. 37 2.5 J wave associated syndromes .................................................................. 37 2.5.1 Brugada syndrome – anterior J waves .......................................... 37 2.5.2 Early repolarization – inferolateral J waves ................................. 42 2.6 QRS complex abnormalities ................................................................... 48 2.6.1 Prolonged QRS duration and bundle branch blocks ..................... 48 2.6.2 Peri-infarction block ..................................................................... 49 2.6.3 Fragmented QRS complex ........................................................... 49 2.7 Summary of the literature........................................................................ 50 3 Aims of the study 53 4 Study populations 55 4.1 Study population A .................................................................................. 55 4.2 Study populations B and C ...................................................................... 55 4.3 Study population D ................................................................................. 56 15 4.4 Study population E .................................................................................. 57 5 Study methods 59 5.1 Study I. Prevalence and long-term prognosis of early repolarization pattern in general population ............................................ 59 5.2 Study II. Clinical features and heritability of early repolarization pattern in the general population ............................................................. 60 5.3 Study III. Long-term prognosis of various early repolarization patterns and the significance of ST-segment in general population................................................................................................ 61 5.4 Study IV. The significance of early repolarization pattern in acute coronary events ....................................................................................... 62 5.5 Study V. Identification of common genetic variants influencing early repolarization pattern...................................................................... 62 5.6 Statistical analysis ................................................................................... 63 6 Results 65 6.1 Prevalence and prognostic significance of early repolarization pattern in a large Finnish general population with a long followup (I) ........................................................................................................ 65 6.2 The clinical correlates and heritability assessment of early repolarization pattern in general population (II) ..................................... 70 6.3 The long-term significance of ER ECG pattern subtypes and STsegment morphology (III) ....................................................................... 75 6.4 Significance of early repolarization pattern during acute coronary events (IV)............................................................................................... 81 6.5 Meta-analysis of genome wide association studies of early repolarization pattern (V) ........................................................................ 84 7 Discussion 87 7.1 Key findings ............................................................................................ 87 7.2 Prevalence of early repolarization patterns (I-IV) ................................... 88 7.3 Demographic features, clinical correlates and heritability of early repolarization pattern (I-III) .................................................................... 90 7.4 Prognosis of early repolarization pattern and the significance of ST-segment morphology (I, III) .............................................................. 92 7.5 Significance of early repolarization pattern during acute coronary events (IV)............................................................................................... 94 7.6 Genetics of early repolarization pattern (V) ............................................ 95 16 7.7 Early repolarization – overall evidence ................................................... 97 7.7.1 The overall evidence of ERP among general population in the literature.................................................................................. 97 7.7.2 Analogies with QT syndromes ..................................................... 99 7.7.3 Association with Brugada syndrome ............................................ 99 7.7.4 Early repolarization or delayed depolarization? ......................... 100 7.7.5 Markers of higher risk of adverse events.................................... 102 7.8 Screening for early repolarization and patient management ................. 107 7.9 Remaining questions and future directions ........................................... 108 8 Strengths and limitations of the thesis 109 9 Conclusions and clinical implications 113 References 115 Original publications 143 17 18 1 Introduction Sudden cardiac death (SCD) is a major cause of mortality in Western societies. It mainly comprises of coronary artery disease and its complications, followed by other cardiomyopathies (Huikuri et al. 2001, Josephson & Wellens 2004, Rubart & Zipes 2005). However, in up to 20% of SCDs, there are no structural abnormalities evident (Puranik et al. 2005). In those situations, the underlying cause is often an electrical disorder, such as Brugada syndrome or long-and-shortQT syndromes. When no specific etiology is known, a term idiopathic ventricular fibrillation is usually used. All of these cardiac states leading to unexpected death are a major issue in affected families, the general community and medical professionals. The standard 12-lead electrocardiogram (ECG), invented over 100 years ago by Willem Einthoven (Moukabary 2007), has been an important assistant in gathering diagnostic and prognostic evidence of electrophysiological disorders of the heart. Even in the current era of molecular biology and genetics, ECG is, if not the most important, at least a significant tool in recognizing the arrhythmia syndromes. Its ease of usage has lead to its spread to all medical professionals and thus it has had a major impact on health care. In the absence of a conduction defect or chest pain, ST-segment elevation often accompanied by J waves, an ECG variant called early repolarization (ER), has been considered a normal state for over 60 years, ever since its first description in 1936 (Myers & Klein 1947a, Shipley & Hallaran 1936). After some case-reports pointing to the arrhythmogenic potential of early repolarization, a series of case-control studies in 2008 unexpectedly described an apparent overpresentation of J waves in patients with idiopathic ventricular fibrillation (IVF) (Haissaguerre et al. 2008, Nam et al. 2008, Rosso et al. 2008). These findings challenged physicians to think again about the belief that this pattern was simply a normal variant, leading to some confusion among both professionals and patients. Therefore a thorough investigation of this phenomenon was urgently warranted. Since early repolarization is known to affect a large section of the population, the focus of this thesis was to assess the prevalence and prognostic significance, and to characterize the clinical features of this ECG pattern primarily in the general population. 19 20 2 Review of the literature 2.1 Basic electrophysiology and electrocardiography The report of the first human electrocardiogram was published in 1887 (Waller 1887), and subsequently it has been used in examination of the heart. The father of the modern ECG is William Einthoven, whose terminology is still being used and whose original research remains fundamental in understanding the ECG. Even though the ECG itself is an ancient entity, it still is a routine part of the patient’s examination. The equipment is widely available, very cost effective and non-invasive, and thus ECG represents an excellent tool in clinical practice. The interpretation of changes in surface ECG can be used in diagnosing otherwise unsuspected cardiac conditions and is indicated in all patients who are being evaluated for suspected ventricular arrhythmias (European Heart Rhythm Association et al. 2006). Knowledge of how to evaluate the standard 12-lead ECG should be familiar to every physician. 2.1.1 Cardiac action potential The action potential of a cardiac myocyte is of interest of electrophysiologists since it not only makes possible the muscle contractions, but also has the potential to induce cardiac arrhythmias. The action potential is a complex event generated by the transient flow of potassium, sodium, and calcium ions across the cell membrane thourgh various ion channels, pumps and exchangers. The cardiac action potential consists of five phases. Phase 0 is the rapid depolarization, phase 1 the early rapid repolarization, phase 2 is the plateau or dome, phase 3 is the final rapid repolarization, and phase 4 is the resting membrane potential. Once the cell has been electrically stimulated, phases from 4 to 0 are generated by the influx or efflux of ions as a consequence of a sequence of events. The consecutive activation of adjacent cells is the basis of cardiac depolarization and repolarization. The ventricular myocite action potential with the currents involved is presented in Figure 1. In the resting state, interior voltage of the cardiac cell is more negative than exterior. The inward sodium and the L-type calcium are the main depolarizing currents. These currents trigger the rapid up-stroke of the action potential, making the membrane potential more positive, with calcium being also involved in the 21 plateau phase. There are several ionic currents involved in the repolarization phase, but the two mainly responsible are the rapid and slow components of the delayed rectifier potassium currents. The repolarizing currents restore the cell’s resting membrane potential by gradually reducing the potential and making it more negative. Fig. 1. Cardiac myocite action potential. The properties and distribution of the ion channels can differ in various cardiac tissues, they are regulated by several genes and they can be modulated by several exogenous factors, such as drugs and electrolyte imbalances. 2.1.2 Normal electric cycle in the ECG On the basis of biophysical principles, every deflection in the ECG represents a coincident voltage gradient generated by cellular electrical activity. The normal surface ECG is the result of temporal changes in the depolarization and repolarization processes; Action potentials between two random regions of the heart are similar in duration and the small variations in timing of local 22 depolarization and repolarization produce the electrocardiographic patterns of the QRS complex and the T-wave. The QRS complex reflects the rapid depolarization of the cardiac ventricles, whereas the T-wave describes ventricular repolarization. Often also J and U waves are claimed to represent ventricular repolarization. The origin of J waves will be discussed in depth in the next chapter. The ST-segment between the QRS complex and T-wave represents the time period, in which all cells in the ventricles are normally depolarized. The leads of an ECG collate the electrical activity from different anatomical areas, for examplethey are important in the recognition of regional ischemia. Usually four lead categories are used: inferior, lateral, anterior and septal leads. Often septal and anterior leads are merged into one category. The inferior lead category includes leads II, III and aVF of the ECG; the lateral category includes leads I, aVL, and precordial leads V(3-)4 through V6; and the anterior category includes precordial leads V1 through V3(-4). 2.1.3 Cellular basis of electrocardiographic J waves The J-point is the point at which the QRS complex finishes and the ST-segment starts, i.e. the onset of the ST-segment. J waves are deflections in the terminal portion of the QRS complex or immediately following it. They are usually described as an elevation of the J-point (normally isoelectric), appearing as a late delta wave following the QRS complex, or as a secondary R wave. Several investigators described prominent J waves during hypothermia in the early 1940s, but over the years, the hypothermic J wave has generally came to be named after Joseph Osborn, who examined the physiological effects of hypothermia and defined the typical electrocardiographic associations in 1953 (Osborn 1953). This ECG deflection has also been called late delta wave, J-point wave, and epsilon wave. The typical electrocardiographic patterns and their variations are illustrated in Figure 2. Three distinct cell types have been demonstrated to be present in the ventricular myocardium; endocardial, epicardial and mid-myocardial (M) cells. The morphology of phase 1 in the action potential in the epicardial and M cells differs from that in the endocardial cells, due to the presence of prominent transient outward current (Ito). Differences in the electrophysiological properties within these cells, leading to transmural differences in the early phases of the action potential, are the basis for the various deflections, such as presentation of J waves, in the ECG. Ventricular epicardial and M cell action potentials, unlike 23 endocardial action potentials, display a prominent phase 1 due to the large Ito current, giving rise to the spike-and-dome configuration of the action potential. Furthermore, the distribution of Ito and thus also the degree of the action potential notch differ regionally, especially between right and left ventricles; the highest density of Ito is present in the right ventricle. The presence of a prominent Ito mediated action potential notch in ventricular epicardium but not in endocardium, produces a transmural voltage gradient, and this is believed to lead to the manifestation of a J wave in the ECG. This transmural voltage gradient is established as the ventricular activation moves from endocardium to epicardium, with the epicardium becoming activated last. Increases in repolarizing currents (Ito) or decreases in depolarizing currents (INa, ICa) accelerate repolarization and accentuate the action potential notch, thus leading to augmentation of the J wave or the appearance of ST-segment elevation. A further increase in net repolarizing current can lead to a partial or even complete loss of the action potential dome, which increases transmural voltage gradient and this evokes an even greater accentuation of J waves and ST-elevation. (Antzelevitch et al. 1991, Hlaing et al. 2005, Yan & Antzelevitch 1996a) In clinical situations, the appearance of prominent J waves is typically associated with hypothermia or hypercalcemia. (Hlaing et al. 2005, Krantz & Lowery 2005, Sridharan & Horan 1984) The augmentation of J waves during hypothermia may be the result of slowing of the kinetics of activation of Ito, as well as the slowing of conduction, leading to an epicardial notch manifesting itself as a distinct deflection in the ECG. Prominent J waves can also be observed during other metabolic alterations, such as acidosis or hypokalemia, and they can be found in several other clinical conditions and even during normal states, as described later. Under all of these conditions, an outward shift in the currents active in phase 1 can lead to augmentation of the action potential notch and thus unmask or augment the J wave. Furthermore, similarities between early phase ischemia and J waves in terms of ECG appearance have been reported in laboratory models. Myocardial ischemia is known to reduce inward currents and to increase the outward potassium currents particularly by intensifying Ito especially in the epicardium, thus possibly unmasking J waves (Yan & Antzelevitch 1999). In classical early repolarization, with concave upward ST-segment elevations, a large voltage gradient is present between two regions and the current flow towards the region still at plateau phase creates a positive ST-segment elevation 24 in the electrocardiographic leads, reflecting the electrical activity in that particular region. Simply put, the heterogeneity in repolarization evokes a current flow from the normal myocardium towards the area with a shorter action potential. These two regions could be two different cardiac regions or different cardiac layers with regional or transmural voltage gradients. To create J waves with only mild or no ST-elevation, the voltage gradient needs to be present early in the repolarization phase, but not later in the action potential, which would create an ST-elevation. Figure 1 illustrates a theoretical mechanism for the origin of a J wave in which the two regions are epi- and endocardium within the same electrophysiological area. Fig. 2. Theoretical origin of a J wave 25 Fig. 3. Possible consequences of a prominent Ito mediated action potential notch in epicardium but not endocardium If one is trying to distinguish a J wave from altered ventricular activation, manifested as various terminal QRS changes, the heart rate can deliver useful diagnostics. In a normal ventricular activation from endocardium to epicardium, acceleration in the heart rate reduces Ito due to its slow recovery from inactivation, thus resulting in a decrease in the J wave amplitude (Yan et al. 2003). In contrast, during a variety of alterations of normal activation and conduction, the increase in heart rate tends to amplify the magnitude of the terminal QRS changes. Similarly drugs that block sodium channel and inhibit Ito, such as quinidine, reduce the J wave amplitude, but they have no effect on the conduction abnormalities. Mainly as a sequence of the known slow recovery of Ito, factors that influence Ito kinetics or ventricular activation sequence can modify the presence of J waves in an ECG. The regional control of repolarization is poorly understood and thus also the basis of regional J waves, i.e. inferior and lateral manifestations, remains also unknown. Arrhythmogenic potential of J waves There are several theories pointing to an arrhythmogenic potential of J waves. If larger Ito currents underlie J waves than any further outward shift of the currents during the early phase of the action potential can lead to a loss of the epicardial action potential dome, and then to a dispersion of repolarization between the region where the dome is lost and regions at which the plateau phase and action potential duration are at least relatively maintained. An all-or-none repolarization due to accelerated Ito could cause a marked shortening of action potential at some sites but not others, which could initiate arrhythmogenesis, either by initiating 26 extrasystoles (by phase 2 re-entry) or via boundary currents analogous to the injury currents observed in acute myocardial ischemia. Re-entry is the propagation of an electrical impulse around two interconnected pathways. The phase 2 re-entrant beat is capable of initiating polymorphic VT or VF via R-on-T extrasystoles. (Gussak & Antzelevitch 2000, Yan & Antzelevitch 1996b, Yan & Antzelevitch 1999) Hypothermia-related giant J waves without ST segment elevation reflect an increased dispersion of repolarization, eventually leading to spontaneous ventricular fibrillation (Fish & Antzelevitch 2004). Hypokalemia has also been reported to increase the amplitude of J waves and lead to multiple episodes of ventricular fibrillation (Myojo et al. 2012). 2.1.4 Electrocardiography as an evaluation tool Over the years, ECG has been used in the diagnostics and risk assessment in the community, in patients with different heart conditions as well as in competitive athletes. Resting and exercise ECGs are now widely used for risk stratification as a supplement to the conventional risk factors. It is believed that the information gathered from an ECG would lead to better risk-reduction therapies in asymptomatic patients without any apparent heart disease (O'Malley & Redberg 2010). 2.2 Cardiovascular morbidity and mortality Cardiovascular diseases (CVDs) are the main cause of death in Europe and other Western societies. Although the death rates from CVD have declined, CVD still accounts for 12% to 25% of potential years of life lost due to premature deaths. The main forms of CVD are coronary artery disease (CAD) and stroke, these account for 40% of all deaths in the European Union (EU). Coronary artery disease itself is the single most common cause of death also before the age of 65 in the EU. In addition to being the largest contributor to mortality in Europe, CVD and CAD are responsible for significant morbidity. The annual rates of discharges for CAD in Europe are around 600 /100,000. CVD is estimated to cost the EU economy almost 200 billion euros a year in health care costs, productivity losses and costs attributable to the informal care of patients with CVD. (European Cardiovascular Disease Statistics, 2012 edition) There is no doubt about the magnitude of the problem. 27 The main risk factors for CVD and CAD are smoking, raised cholesterol levels and blood pressure, and thus control of these conventional risk factors remains essential in the prevention of the disease and its complications. One of the most feared consequences of coronary artery disease is sudden cardiac death (SCD), which is unfortunately also often the first and only manifestation of the disease. Furthermore, most of these deaths occur in the general population, in individuals who have been considered to be at a low risk for such an event (Huikuri et al. 2001, Myerburg 2002). SCD accounts for about 50% of all CAD related deaths and up to 20% of all deaths, and thus represents an enormous public health problem. Today, one of the major challenges faced in current cardiology is to predict who will die suddenly from ventricular arrhythmias and a large amount of effort is being put into solving this problem. 2.2.1 Sudden unexplained death in the community To begin with, it is important to distinguish sudden cardiac death from acute myocardial infarction (AMI) or heart attack. SCD is the manifestation of a fatal heart rhythm disorder such as ventricular tachycardia, fibrillation or severe bradycardia, leading to hemodynamic collapse, whereas myocardial infarction evolves due to an occlusion of a coronary artery leading to an insufficient blood supply to the myocardium. Acute myocardial infarction might lead to SCD, but these conditions are not to be considered as uniform events, as SCD can occur also in the absence of myocardial ischemia. (Zipes & Wellens 1998) The European Society of Cardiology has proposed the following definition for sudden cardiac death: death from an unexpected circulatory arrest, usually due to a cardiac arrhythmia occurring within an hour of the onset of acute symptoms (European Heart Rhythm Association et al. 2006). Prior cardiac disease may have been known to be present, but the time and mode of death is unexpected (Priori et al. 2001). In Europe and North America, the incidence of sudden cardiac death ranges from 50 to 100 /100 000/year among general population (Byrne et al. 2008, Chugh et al. 2004, de Vreede-Swagemakers et al. 1997, Vaillancourt et al. 2004). In a recent report from Finland, the annual incidence of SCD was 57 /100 000 (Hookana et al. 2011a). In younger populations, the incidence of SCD is lower, approximately 1-2 /100,000/year (Papadakis et al. 2009). The tradgedy is that in two thirds of all SCDs in adulthood, the death occurs among previously 28 asymptomatic victims, i.e. it is the first clinical manifestation of the disease or unexpectedly in individuals in whom the disease had been diagnosed, but who had been assessed to be at low risk (Huikuri et al. 2001, Myerburg 2001, Myerburg & Vetter 2007, Wellens et al. 1995). The proportion of death in asymptomatic individuals is even greater among adolescents and young adults. Mechanisms of sudden cardiac death Cardiac arrhythmias represent a major risk factor for cardiac arrest and sudden death. The accurate identification of the electrophysiological mechanisms responsible for SCD is difficult, since the pathophysiology is complex and the underlying mechanisms are likely to be multifactorial. The rhythm most often recorded at the time of cardiac arrest is ventricular fibrillation, which is believed to account for approximately three quarters of SCDs, followed by bradyarrhythmias and electromechanical dissociation (Bayes de Luna et al. 1989, Luu et al. 1989). It is still unclear, how a previously asymptomatic, apparently healthy individual suddenly develops SCD at a certain time. It is believed, that several dynamic factors interact with a fixed substrate to stimulate arrhythmias, ultimately leading to hemodynamic collapse. The factors possibly modulating cardiac conduction have been postulated to include transient ischemia, inflammation, pharmacologic effects, electrolyte changes and other systemic metabolic alterations perhaps involving several other dynamic, often transient events. Of these, the most common factor triggering fatal arrhythmias is severe myocardial ischemia, which accounts for about 80% of all cases. Genetics of SCD SCD is believed to have a strong genetic background. However, coronary artery disease, the premier contributor to SCD, is known to develop as a result of complex interactions between genetics, lifestyle habits and the environment. A familial component to SCD have been demonstrated in several independent studies (Dekker et al. 2006, Jouven et al. 1999, Kaikkonen et al. 2006), and a history of sudden deaths within the family can be found in up to 30% of victims of SCD (Behr et al. 2003, Behr et al. 2008). Therefore the presence of a family history of unexplained seizures and early sudden death should be systematically obtained. Nowadays genetic testing is performed more often to confirm the 29 diagnosis in a patient with clinical manifestations of a certain condition. However, it should be noted that although genetic and heritable factors are evident in several inherited syndromes, family members with the same genetic mutation might present with different clinical signs and symptoms ranging from being completely asymptomatic to suffering sudden death. In other words, the phenotype does not always match the genotype, i.e. the penetrance of the syndrome is usually incomplete. A large number of the arrhythmia syndromes, some of which will be described later in this literature review, are influenced by not only genetic, but also individual and environmental factors (Priori 2004). Nevertheless genotyping is increasingly useful in risk stratification as the cost effectiveness and sequencing technologies are developing. The ultimate goal is to be able to provide gene-specific therapies. The genome-wide association (GWA) study is a method of genetic epidemiology in which many common genetic variants in different individuals are examined to identify associations between single-nucleotide polymorphisms (SNPs) and traits such sas SCD. After the first published GWA study in 2005, the numbers of GWA studies have skyrocketed. GWA studies have the ability to identify genetic variants with effects on the investigated disease, but they require large sample sizes in order to reach statistically significant associations. Recently, a large-scale GWA meta-analysis identified a locus associated with SCD susceptibility in individuals of European ancestry (Arking et al. 2011). However, much work is still required to define the functional basis for the observed associations before any medical interventions can be developed with clinical significance. Idiopathic ventricular fibrillation Idiopathic ventricular fibrillation is a disorder especially affecting young adults, which presents as syncope or cardiac arrest due to polymorphic ventricular tachycardia (Belhassen & Viskin 1993, Viskin & Belhassen 1990). The term idiopathic refers to the absence of any identifiable etiology and therefore idiopathic VF is a diagnosis made after the exclusion of other known etiologies. There is no demonstrable structural heart disease or identifiable channelopathy to account for the arrhythmias. In some contexts, this condition is also referred to as sudden unexplained death syndrome or sudden arrhythmic death syndrome. 30 A typical patient with idiopathic VF is aged 20 to 40 years, tends to develop arrhythmic storms unrelated to stress with numerous episodes of ventricular fibrillation and fails to respond to conventional antiarrhythmic therapy (Haissaguerre et al. 2008, Haissaguerre et al. 2009b, Martini et al. 1989). In fact, the initial report hinting at the malignancy of early repolarization and also the impotus for this thesis, was the observation that one out of three patients with idiopathic VF had exhibited early repolarization in the inferolateral leads (Haissaguerre et al. 2008). 2.3 Cardiomyopathies Cardiomyopathies cause heart failure, ventricular arrhythmias and sudden cardiac death because of structural changes in the myocardium. They are defined as disorders in which the myocardium is structurally and functionally abnormal in the absence of CAD, valvular disease or congenital heart disease (Elliott et al. 2008a). Cardiomyopathies are the most common cause of death in non-ischemic SCDs, which account for approximately 20% of all sudden cardiac deaths (Hookana et al. 2011b, Huikuri et al. 2001). 2.3.1 Hypertrophic cardiomyopathy Hypertrophic cardiomyopathy (HCM) involves myocardial hypertrophy in the absence of any obvious hemodynamic load that could cause extensive hypertrophy (Elliott et al. 2008b, Maron & Maron 2012, Richardson et al. 1996). It is a rare cause of sudden death among middle-aged adults, but in young adults, particularly in competitive athletes, it is a common cause of SCD (Huikuri et al. 2001, Maron et al. 1998). The prevalence of HCM is estimated to be 0.2%. In Finnish children, the prevalence has been reported to be 0.03% (Arola et al. 1997). The annual incidence of SCD is <1%, although the prognosis varies widely from asymptomatic to high risk of SCD (Adabag et al. 2005, Elliott et al. 2006, Maron et al. 2000). The electrocardiographic features of HCM primarily reflect voltage patterns associated with left ventricular hypertrophy and they are often difficult to distinguish from the secondary hypertrophy seen in other heart illnesses (Fananapazir et al. 1989, Savage et al. 1978). Typically, there is evidence of left atrial and ventricular enlargement and marked strain patterns characterized by prominent ST-T wave changes in the absence of hypertension. However, in a 31 recent study the pre-participation screening ECG was normal in 10% of patients with hypertrophic cardiomyopathy (Rowin et al. 2012). In the obstructive form of HCM with asymmetrical septal hypertrophy, prominent inferior Q waves and/or tall R waves in V1 may be present. In apical HCM, there is a possibility for the absence of increased voltages, but prominent T wave inversions in the antero-lateral leads are usually present. Similar ECG changes have been observed among young healthy male athletes, with so-called athlete’s hearts, but in that situation usually repolarization changes are less prominent and there are no atrial enlargement patterns (Corrado et al. 2010). 2.3.2 Dilated cardiomyopathy Dilated cardiomyopathy (DCM) is a condition in which the left or both ventricles dilate leading to dysfunction of the heart and the symptoms of heart failure. The prevalence of DCM is estimated to be under 0.04% (Dec & Fuster 1994, Manolio et al. 1992). Ventricular arrhythmias are common in the advanced stage of DCM (Cleland et al. 2002), but in rare cases, SCD can be the first manifestation of the disease (Adabag et al. 2010). The median survival of patients with DCM has been reported to be 2 years, and half of the deaths occur suddenly and unexpectedly (Fuster et al. 1981, Johnson & Palacios 1982a, Johnson & Palacios 1982b). The electrocardiographic features of DCM are non-specific, but concomitant with the structural changes related to the condition (atrial enlargement and ventricular dilatation with thin walls due to loss of myocardium). ST-T wave abnormalities particularly in the lateral leads and abnormal intraventricular conduction are common. A rare ECG combination of left bundle branch block and right axis deviation has been reported to be suggestive for dilated or congestive cardiomyopathy (Nikolic & Marriott 1985). The presence of symptoms of heart failure and ultimately echocardiography can distinguish these changes from other cardiac illnesses. 2.3.3 Arrhythmogenic right ventricular dysplasia Arrhythmogenic right ventricular dysplasia or cardiomyopathy (ARVD or ARVC) is an inherited heart disease, which involves cardiomyocyte death and replacement of the myocardium with fibro-fatty tissue in the right ventricle. 32 (Corrado et al. 1997, Dalal et al. 2005, Marcus et al. 1982) In advanced cases also the left ventricle might be affected, causing dilated cardiomyopathy. This disorder is a common cause of SCD in young athletes (Thiene et al. 1988). The fibro-fatty replacement has been proposed to lead to delayed conduction and abnormal depolarization in the affected areas, predisposing to re-entry-mediated ventricular tachycardia. The most common presenting symptoms are palpitations, syncope and death (Dalal et al. 2005, Fontaine et al. 1999, Marcus & Fontaine 1995). The prevalence of ARVD is unknown, but has been estimated to be 0.02% (Cox et al. 2009, McKenna et al. 1994, Norman & McKenna 1999). However, the condition is likely to be under-diagnosed due to the challenging diagnosis. The clinical presentation varies from an asymptomatic state to SCD. ARVD is a progressive condition and therefore the risk of SCD might change over time. The annual incidence of mortality has been estimated to be 1% to 3%, depending on the treatment provided (Corrado et al. 2000). The golden standard for diagnosis is the demonstration of the fibro-fatty infiltration, which can only be observed during cardiac surgery or autopsy, but fortunately ECG changes are present in up to 90% of patients (Dalal et al. 2005, McKenna et al. 1994). The most recent major electrocardiographic diagnostic criteria for ARVD have included epsilon waves, QRS >110 ms, inverted T-waves in precordial leads V1-V3, and left bundle branch block morphology VT with superior axis (Jain et al. 2009, Marcus et al. 2010). Minor ECG criteria are Twave inversions in 2-3 consecutive precordial leads, LBBB morphology VT without superior axis, terminal activation delay, and right bundle branch block pattern with T-wave inversions in precordial leads. T-wave inversions in left precordial leads are considered as characteristic although non-specific for ARVD, but are still mostly ignored in clinical situations. The prevalence of these T-wave changes has been estimated to be between 1% and 3% (Marcus 2005). In a recent Finnish study, the prevalence of T-wave inversions in the right precordial leads was 0.5% among middle-aged general population subjects (Aro et al. 2012). In this large sample, T-wave changes in V1-V3 did not predict any increased mortality or sudden death. 2.3.4 Myocardial fibrosis Usually the formation of fibrosis in the myocardium is triggered by tissue stresses such as myocardial ischemia, inflammation or hypertensive overload. Infrequent cases of SCD as the first manifestation of underlying disease have been associated 33 with idiopathic myocardial fibrosis, the presence of fibrosis in the absence of other cardiac diseases. Recently, fibrotic cardiomyopathy, especially in victims under 40 years, was associated with non-ischemic SCD in a large Finnish population (Hookana et al. 2011b). Earlier myocardial fibrosis had been identified in only 1% to 3% of cases among autopsy series of victims of sudden cardiac death (Chugh et al. 2000a, Drory et al. 1991, Priori 1997). This condition involves transmural, interstitial fibrosis and other structural abnormalities, which can cause electrical heterogeneity and that are recognized as determinants of ventricular arrhythmogenesis (John et al. 2004). The electrocardiographic features of the condition are thought to include T wave inversions throughout the precordial leads, although fibrosis could cause a wide range of non-specific ECG changes as a result to modification in impulse conduction. Thus far there is no reliable data available of the variety of ECG changes present in a fibrotic heart. In the future, improvements in imaging techniques and biomarkers will probably allow more precise recognition of the condition. 2.4 Channelopathies A significant portion of unexplained sudden cardiac deaths are caused by a spectrum of different arrhythmia syndromes with a structurally normal heart and no other identifiable aetiology leading to sudden death. These conditions are rare, but often serious. These so called sudden arrhythmic death syndromes or channelopathies have been estimated to account for approximately 5% of SCDs (Chugh et al. 2000b). Most cases occur in young men, who die during sleep or at rest, usually without any prior cardiac symptoms such as syncope. Although the absolute number of unexpected SCD among previously healthy persons in the community is small, the presence of this kind of risk is disstressing and every death causes unnecessary sorrow within the families of the victims. The so-called channelopathies are caused by mutations in genes leading to electrical instability of the heart, and thus predisposing the individual to malignant arrhythmias. The heterogeneous group of channelopathies include short and long QT syndromes, Brugada syndrome and catecholaminergic polymorphic ventricular tachycardia (CPVT). These cardiac abnormalities are believed to have a structurally normal heart, a genetic basis, and they are known to predispose to life-threatening arrhythmias. 34 2.4.1 Long QT syndrome The long QT syndrome (LQTS) is an inherited syndrome characterized by prolongation of the heart rate corrected QT interval in a standard electrocardiogram with vulnerability for ventricular tachyarrhythmias potentially leading to sudden cardiac death (Moss et al. 1991). The QT interval might also be prolonged due to so-called acquired factors, such as drugs that modify cardiac repolarization. Both forms, congenital and acquired QT prolongation, have the potential to develop ventricular arrhythmias (Algra et al. 1991, Chugh et al. 2009, Moss et al. 1991, Straus et al. 2006). A total of twelve genes have been identified as being involved in inherited LQTS, but three subtypes account for the vast majority of cases (Hedley et al. 2009). It is noteworthy that nearly a quarter of affected patients may not have any identifiable gene mutations (Lehnart et al. 2007, Tester et al. 2006). The prevalence of long QT syndrome has been estimated to be 4/10,000 (Schwartz et al. 2009), but the disorder remains underdiagnosed as some of the LQTS gene carriers have a normal QTc duration. Recently, a very high risk of adverse events was observed in patients with the LQTS gene carries although presenting with normal resting ECGs (Goldenberg et al. 2011). There is a considerable overlap between patients with LQTS associated mutations and individuals in the general population with otherwise prolonged QT interval, as the prevalence of prolonged QTc has been reported to be as high as 11% (Schwartz et al. 2009, Straus et al. 2006). Idiopathic abnormal prolongation of the QTc has been associated with a five-fold risk of SCD among patients with CAD (Chugh et al. 2009). In the general population, the mortality risk has been reported to be moderate when QTc is less than 500ms (Priori et al. 2003, Priori et al. 2004). The electrocardiographic features include QT interval prolongation and morphological T-wave abnormalities. Heart rate correction is required in order to obtain reliable values, and Bazett’s formula is the most commonly used correction method. Leads II and V5 are usually recommended for interval measurements (Schwartz et al. 1993). The current limits for prolonged QTc (heart rate corrected QT interval) are 440 or 450 ms for men and 460 ms for women (Rautaharju et al. 2009a), but clinically significant prolongation might be greater. Several suggestive ECG patterns have been correlated to different genotypes (Schwartz et al. 2001), but they are not in general clinical usage. 35 2.4.2 Short QT syndrome The short QT syndrome (SQTS) is a rare arrhythmogenic repolarization abnormality characterized by a very short QT interval. The possible clinical consequences related to the condition include atrial fibrillation, syncope, cardiac arrest and sudden cardiac death. (Gaita et al. 2003a, Gussak et al. 2000, Gussak & Bjerregaard 2005, Viskin et al. 2004) Similarly to the situation of prolongation, the QT interval may be shortened due to either congenital or acquired causes (Maury et al. 2008). SQTS is also genetically similarly heterogeneous as LQTS. Thus far, five mutations have been identified in SQTS (Antzelevitch et al. 2007a, Bellocq et al. 2004, Brugada et al. 2004, Priori et al. 2005). The electrocardiographic features are a QT interval significantly below the lower limit of normal, and tall and sharp T waves (Gaita et al. 2003b). The lower limit of normal corrected QT interval in the general population is believed to be 340-360ms according to Gaussian normal distributions of QTc. In SQTS, the QTC is <320ms, but the exact cutoff values for QTc in SQTS are still a matter of debate. Currently, diagnostic scores for inherited SQTS include the QTc <370330ms and J-point to T-peak interval <120ms, clinical symptoms, family history and genotype (Gollob et al. 2011). A short QT interval in standard resting ECGs in the general population is not uniformly associated with fatal cardiac events. The prevalence of a short QT interval (<340 ms) was 0.4% in a Finnish general population sample, and none of those individuals had died during a very long follow-up of nearly 40 years (Anttonen et al. 2007). There is a report of a prevalence of 1% in another European sample with a similar QTc cutoff value (Kobza et al. 2009). Furthermore, the presence of a short QTc was rare and it was not associated with any increased risk of SCD in Italian and Japanese populations (Funada et al. 2008, Gallagher et al. 2006, Moriya et al. 2007). Recently it has been demonstrated that nearly two thirds of SQTS subjects also present J waves in the inferolateral leads (Miyamoto et al. 2012, Watanabe et al. 2010), which is consistent with prior case reports (Efremidis et al. 2009). J waves may also increase the likelihood of arrhythmic events in those with SQTS, but further studies are required. 36 2.4.3 Catecholaminergic polymorphic ventricular tachycardia Catecholaminergic polymorphic ventricular tachycardia (CPVT) is one of the most malign forms of ventricular arrhythmia in the absence of structural abnormalities of the heart. It manifests often during early childhood, adolescence, or young adulthood causing syncope during exercise. Sudden death is commonly the first manifestation of this disease. (Leenhardt et al. 1995) CPVT causes symptoms in up to 80% of patients under the age of 40 carrying the condition which means that the cumulative mortality may be as high as 40% (Sumitomo et al. 2003). CPVT is not recognizable in the resting ECG, since it does not display any typical ECG abnormalities. Polymorphic ventricular extrasystoles are typical findings during exercise, but syncope during exercise or during sympathetic activation are the keys to identifying this disease. 2.5 J wave associated syndromes The cellular basis and arrhythmogenic potential of J waves were presented previously in this literature review. It is known that the magnitude and location of J waves can vary and influence the prognostic significance. In this chapter, the basics of Brugada syndrome and early repolarization, both in which J waves are present, are briefly described. Due to their similarity in ECG characteristics, clinical outcomes and shared arrhythmic platform, it has been proposed that Brugada syndrome and early repolarization syndrome should be collectively referred to as “J wave syndromes” as a continuous spectrum of phenotypic expression (Anzelevitch & Yan 2010). 2.5.1 Brugada syndrome – anterior J waves Historical background The Brugada syndrome (BrS) is an inherited arrhythmic disorder predisposing to sudden cardiac death. It was first described in 1992 as a new clinical entity of right bundle branch block, ST-segment elevation followed by a negative T-wave and susceptibility to SCD in patients without any obvious structural heart disease (Brugada & Brugada 1992). Thereafter the syndrome has been defined in a series of independent studies, and today it is recognized as an important cause of SCD 37 in young men, especially in Southeast Asians. (Antzelevitch et al. 2005a, Nademanee et al. 1997). Fig. 4. Theoretical examples of Brugada ECG types in leads V1 to V3. Electrocardiographic characteristics and diagnostics The diagnosis of Brugada syndrome can be made by recognizing its distinct ECG pattern from baseline or drug induced ECG. The electrocardiographic features of Brugada syndrome include ST-elevation in right precordial leads in combination with an atypical right bundle branch block pattern and T-wave abnormalities (Brugada & Brugada 1992). The diagnosis of Brugada syndrome is definite if there is a positive family history, arrhythmia related symptoms or if documented arrhythmias are observed in a patient presenting with type I ECG pattern, as explained below. Three types of Brugada ECG patterns can be recognized, of which the type 1 is required for the diagnosis of Brugada syndrome. This is described as a coved ST-elevation over 2 mm 40ms after the J-point followed by a negative T-wave in at least two right precordial leads at baseline or after drug challenge (class I antiarrhythmic drugs) (Antzelevitch et al. 2005b, Wilde et al. 2002). Types 2 and 3 are considered as suggestive, but not diagnostic for Brugada syndrome in the absence of documented arrhythmias, unexplained syncope or druginduced/spontaneous conversion to a typical type 1 pattern. The type 2 ECG 38 pattern differs from type 1 since it exhibits upright T waves in right precordial leads, and the type 3 pattern otherwise resembles type 2, but there is only minimal ST-segment elevation. The three different Brugada ECG types are presented in Figure 3. An interesting article was recently published with results suggesting that patients carrying the type 1 Brugada pattern in only one precordial lead presented with similar clinical profile and had a similar arrhythmic risk as those with the diagnostic two positive precordial leads, and therefore a revision of the concensus criteria of Brugada syndrome has been proposed (Richter et al. 2010). Furthermore, one notable observation is the fluctuation between normal ECG and different types of Brugada ECG patterns, which is common in patients with Brugada syndrome (Tatsumi et al. 2006, Veltmann et al. 2006). Thus, it is important to acknowledge that all of the mentioned three Brugada patterns may coexist in the same patient at different times, and that the patterns might sometimes be completely absent and present only during some transient circumstance. Similar to the situation with the QT-prolongation in LQTS, pharmacological agents can also provoke the typical Brugada ECG pattern, and therefore drug challenge with class I antiarrhythmics (flecainide, ajmaline, procainamide, etc.) is sometimes required to unmask the characteristical ECG changes. Epidemiology and prognosis The prevalence of Brugada type 1 ECG pattern among the general population in European region ranges from 0% to 0.2% (Gallagher et al. 2008, Hermida et al. 2000, Junttila et al. 2004, Sinner et al. 2009), but in individuals of SoutheastAsian origin, it is more common (Matsuo et al. 2001, Miyasaka et al. 2001). The syndrome predominates in males (approximately 80% of patients are men). The mean age for diagnosis, and the average age of the first event are around 30-40 years. (Antzelevitch et al. 2005a, Brugada et al. 2002, Brugada et al. 2003, Priori et al. 2002, Probst et al. 2010a) Brugada syndrome patients usually remain asymptomatic, but between 17% to 42% of them have been reported to suffer from syncope or SCD at some time during their lives (Brugada et al. 2002, Eckardt et al. 2005, Priori et al. 2002), although this is probably an overestimation of the risk as most of the asymptomatic patients are never diagnosed with the syndrome. The annual incidence of malignant arrhythmias among previously asymptomatic subjects has been estimated to be 0.5%, among patients with prior syncope it is 1.9%, and 39 among patients with aborted cardiac arrest it is as high as 7.7% (Probst et al. 2010b). The long-term risk of malignant arrhythmias has been reported to range from 0.5% up to 8% among previously asymptomatic patients, and from 6% up to 62% among patients with a previous episode of cardiac arrest or syncope (Antzelevitch et al. 2005a, Brugada et al. 2002, Brugada et al. 2003, Giustetto et al. 2009, Priori et al. 2002, Probst et al. 2010a). The prevalence of types 2 and 3 has been claimed to be as high as 0.5% in Western populations, but fortunately they do not seem to predispose to sudden cardiac death in a similar fashion to type 1 (Gallagher et al. 2008, Junttila et al. 2004). In individuals with the type 1 Brugada ECG pattern, prolonged QRS (>100ms) and a markedly elevated J-point in precordial leads, are signs of an elevated risk of SCD. Furthermore, medication, anesthesia, or the fever-induced type 1 pattern, have been claimed to possess a high risk of ventricular arrhythmias. (Junttila et al. 2008a, Junttila et al. 2008b) A family history of SCD does not seem to reliably predict a high arrhythmia risk in Brugada syndrome (Brugada et al. 2003, Probst et al. 2010a). Genetics and pathophysiology Brugada syndrome is typically an autosomal-dominantly transmitted inherited condition with a heterogenous genetic basis, but the disease can be sporadic in a proportion of patients. During past years, several mutations have been linked with the syndrome. The most commonly found genotype in Brugada syndrome is a mutation in SCN5A, which leads to a loss of function of the cardiac sodium channel, but these mutations account for only between 11 to 28% of BrS cases. (Antzelevitch et al. 2005a) Sudden death in Brugada syndrome typically occurs at rest, often at night, when vagal tone is augmented (Matsuo et al. 1999). SCD is caused by polymorphic ventricular tachycardia originating in the right ventricular outflow tract (Morita et al. 2003). The pathophysiologic mechanisms of Brugada syndrome are still somewhat indefinite. The syndrome has been considered as a channelopathy since its recognition, but no single causal factor appears to apply for all patients. The ECG pattern has been demonstrated to be modulated by genetic mutations and the kinds of pharmacological agents which can disrupt the function of ion channels, 40 but often also signs of right ventricular structural abnormalities are found. Basically, there are two theories of depolarization and repolarization to explain the pathophysiologic mechanisms responsible for the syndrome, and there is evidence supporting both theories (Hoogendijk et al. 2010). The repolarization disorder hypothesis is supported by several pieces of clinical and experimental evidence. First, sodium channel blockers provoke STelevation in BrS (Brugada et al. 2000). Second, there is an association with the SCN5A mutations, which result in reduced INa current (Chen et al. 1998). Third, quinidine, an antiarrhythmic drug with Ito blocking properties, normalizes the ECG pattern and prevents arrhythmias (Belhassen et al. 2004, Hermida et al. 2004). Furthermore, the ECG pattern is dynamic and more prevalent among males, and ST-elevations are usually facilitated by slow heart rates (Antzelevitch et al. 2005a, Atarashi et al. 1996, Matsuo et al. 1998). There is good evidence to support depolarization disorder hypothesis as well. First, structural changes, such as fibrosis, have been found in BrS patients (Corrado et al. 1996, Corrado et al. 2001). Second, there is a high prevalence of late potentials, which also indicate an increased risk of suffering arrhythmic events (Hisamatsu et al. 2004, Ikeda et al. 2001). There are also other signs of general conduction abnormalities, such as axis deviation and QRS prolongation (Atarashi et al. 2001, Brugada & Brugada 1992). Additionally, fragmented QRS complexes in BrS also are indicative of an increased risk of recurrent VF (Morita et al. 2008a). Finally, flecainide, a conduction slowing drug, induces a greater QRS widening in BrS patients than controls (Shimizu et al. 2000). Based on these observations, it seems reasonable to conclude, that BrS is likely to be a multifactorial disease and thus further studies will be needed to clarify this issue. Patient management in Brugada syndrome Family screening in first-degree relatives is recommended due to the known heritability of Brugada syndrome. All patients diagnosed with Brugada syndrome should be followed-up on a regular basis to identify possible development of symptoms. If syncope or seizures appear, the patient should be promptly clinically evaluated. Since fever may trigger arrhythmias in Brugada patients, close monitoring is recommended during fever episodes. Patients are also advised to avoid all drugs that are known to have the possibility to trigger ventricular 41 arrhythmias, and to avoid unnecessary usage of other medication. (Antzelevitch et al. 2005a) Ultimately the implantation of an ICD is the only proven and effective treatment for prevention of SCD in Brugada syndrome. An ICD is recommended in all patients with symptoms and for asymptomatic patients in whom arrhythmias are inducible. (Antzelevitch et al. 2005a) The rate of inappropriate shocks has been reported to be considerable (Sarkozy et al. 2007), and therefore efforts have been made to search for possible drug options for the treatment. Quinidine has been proposed as an alternative to ICD (Belhassen et al. 2009), but at present, ICD is the standard treatment. 2.5.2 Early repolarization – inferolateral J waves Historical background and terminology Early repolarization, ST-segment elevation in the absence of conduction abnormalities or chest pain, occurring particularly in young and physically active, bradycardic individuals, has been considered as a normal state for over half a century (Fenichel 1962, Gibbons et al. 1977, Goldman 1953a, Grant et al. 1951, Kambara & Phillips 1976a, Klatsky et al. 2003a, Myers & Klein 1947b, Wasserburger & Alt 1961). It was first described in 1936 as J deflections manifested as slurring or notching of the terminal part of QRS complex in healthy young men and women (Shipley & Hallaran 1936). In fact, since this ECG pattern predominates among young and fit individuals with slow heart rates, it has been generally viewed as a marker of good health. Fig. 5. Theoretical example of classical early repolarization in precordial leads V2 to V5. During the years, ER was further defined as an elevated take-off of the STsegment at J-point (Wasserburger & Alt 1961). Historically, the hallmark of early 42 repolarization has indeed been an elevation of the ST-segment in the absence of conduction abnormalities or chest pain, since it could be confused with myocardial injury, ischemia or pericarditis. Furthermore, most recommendations for standardization and interpretation of the electrocardiogram include a statement that the term early repolarization is used to describe a normal QRS-T variant with ST segment elevation (Rautaharju et al. 2009b). Figure 4 illustrates some theoretical examples of precordial leads in classical early repolarization. Since the high impact publication by Haissaguerre and his colleagues in 2008 (Haissaguerre et al. 2008), the term early repolarization pattern has been used to describe an elevation of the QRS-ST junction (J-point), manifested as terminal QRS notching or slurring (Figure 5), and also referred to as J waves, with or without ST segment elevation in the infero-lateral leads. Leads V1 to V3 were excluded to avoid confusion with BrS and ARVD. This definition is somewhat inconsistent with the classical viewpoint, in which J waves are nearly inevitably considered to be accompanied by ST-segment elevation, and the antero-septal leads are also included. In fact, traditionally early repolarization has been used to describe ST-elevation in antero-lateral leads. Subsequently inevitably the question arouse of whether this described abnormality at the end of the QRS complex reflecting early repolarization or alternatively delayed activation of the inferolateral wall (Wellens 2008); this will be discussed in more detail in the Discussion. Throughout this thesis, the term early repolarization (ER) ECG pattern is used to describe terminal QRS changes (J waves) irrespective of the following presence or absence of ST segment elevation. Fig. 6. J waves, manifested as notching and slurring in theoretical limb leads (lead II). Electrocardiographic characteristics As stated in the previous section, the classical definition of early repolarization is more or less equivalent to ST-segment elevation. However, in recent years, the usage of the term has evolved. Today, early repolarization can be considered as an elevation at J-point or a distinct J wave manifested as slurring or notching of the 43 terminal part of the QRS complex, and often, but not necessarily, accompanied by ST-segment elevation (Haissaguerre et al. 2008). These QRS changes are often described as at least 0.1 mV “hump-like” deflections immediately after a positive QRS complex; either a well-defined positive deflection inscribed on the S wave (notching) or a smooth transition from QRS to ST segment (slurring). By definition, at least two leads positive for these terminal QRS changes within the same cardiac location are required for the diagnosis of an early repolarization pattern. The grading of ERP is not always straightforward due to electrophysiological issues. Since QRS complex onset and offset usually varies according to which lead is being inspected, the grading of ERP and measurement of the J-point is occasionally problematic. In fact, there might be up to 20 ms difference in the end of the QRS complex between two leads (Lepeschkin & Surawicz 1952). Therefore, the gradual shifting of QRS into ST-segment as “slurring” is more or less subjective, and similarly notching of the terminal QRS in one lead might be considered to be either due to a conduction delay or to a fragmented QRS (Bastiaenen & Behr 2012). Defining the point for amplitude measurement is not always accurate due to these issues. In most cases, an early repolarization pattern is recognizable from resting baseline ECG, but the knowledge of its instability does exist. Heart rate and autonomic tone have a major impact on the variability of ERP (Gross 2010). Adrenergic stimulation suppresses the pattern, and conversely vagal tone increases the occurrence of J waves and their amplitude (Abe et al. 2010, Kalla et al. 2000, Shinohara et al. 2006a, Takagi et al. 2000). Early repolarization pattern and the risk of adverse events Before 1980, most of the literature concerning early repolarization viewed the pattern as a sign of good prognosis. However, these reports were small in sample size, poorly matched with controls, and mainly consisted of early repolarization in the precordial leads. (Fenichel 1962, Goldman 1953b, Goldman 1960, Grusin 1954, Kambara & Phillips 1976b, Wasserburger & Alt 1961) More recently, in the 1990s and early 2000, numerous cases have been reported describing J waves in otherwise typical idiopathic ventricular fibrillation (Kalla et al. 2000, Komiya et al. 2006, Riera et al. 2008a, Sahara et al. 2002, Shinohara et al. 2006a, Tsunoda et al. 2004). These cases commonly exhibited J waves in the inferior leads, and J 44 waves augmented during slower heart rates and immediately prior to the onset of VF. Other than the obvious distribution difference in the presence of ER patterns, the proarrhythmic potential of J waves with cellular mechanistic explanations in late 1990s probably is the reason why more emphasis was placed on J waves rather than on plain ST-elevation. However, as late as 2003, Klatsky and colleagues published a study on ER, highlighting the benign nature of the pattern in a follow-up of over 12 years (Klatsky et al. 2003b). They defined the ERP as ST-elevation, but did not describe in detail the number of leads needed for diagnosis of early repolarization, and actually only 29% of their ERP positives had J waves present. A landmark case-control study on ERP was published in 2008 by Haissaguerre and colleagues (Haissaguerre et al. 2008). They found that the early repolarization ECG pattern was over six times more frequent in patients with IVF than controls, and ICD monitoring exhibited higher incidence of recurrent VF in case patients with ERP than those cases without the pattern. These results were soon confirmed in two independent case-control studies (Nam et al. 2008, Rosso et al. 2008). The publication of these high impact studies stimulated the ongoing revisitation of early repolarization. Table 1 summarizes the most widely cited publications on the prognosis of ERP prior to the onset of this thesis in 2008. More recent evidence will be presented in detail in the Discussion of the thesis. The syndrome of early repolarization Since the groundbreaking publications indicating an over-presentation of ERP in patients with idiopathic VF, the term early repolarization syndrome (ERS) has been commonly used to describe patients with an early repolarization ECG pattern and documented idiopathic VT/VF. This term is to be considered only in those patients with documented malignant arrhythmias, whereas the term early repolarization pattern refers to the ECG pattern itself, and can be applied also to asymptomatic individuals. Patients with the early repolarization syndrome are likely to be young males with a history of cardiac arrest during sleep (Abe et al. 2010, Haissaguerre et al. 2008, Rosso et al. 2008). The characteristic ECG includes ERP with high amplitude J waves in the infero-lateral leads, which are augmented prior to an arrhythmic event (Nam et al. 2008, Tereshchenko et al. 2012). 45 Genetic studies have thus far revealed mutations mainly in the candidate genes that might influence the phase 2 of the action potential. A gain of function mutation in KCNJ8, which encodes the KATP-channel, has been found in patients with ERS in several independent investigations (Barajas-Martinez et al. 2012, Delaney et al. 2012, Haissaguerre et al. 2009a, Medeiros-Domingo et al. 2010). Another study found mutations in CACNA1, CACNB2 and CACNA2D1 in 16% of probands with ERS (Burashnikov et al. 2010). These mutations are postulated to cause loss of function affecting the L-type calcium current. Loss of function SCN5A mutations have also been found in 6% of Japanese ERS patients (Watanabe et al. 2011). The function of SCN5A was briefly discussed in the BrS chapter above. However, a recent study demonstrating the autosomal dominant heritance of familial ERS detected no mutations in the above genes. The phenotype is likely influenced by many independent loci and interactions. A further discussion on the genetics and heritability of early repolarization will be presented later in this thesis. 46 47 emphasis on J waves 124 controls, 132 athletes Inferolateral leads, emphasis on J waves 1395 controls 45 IVF patients, N/A N/A N/A N/A 12 years 5-26 years 1-20 years None 1-3 years <1 year Follow-up <2 years N/A denotes not applicable (no follow-up in case reports or case-control series) Rosso (2008) Inferolateral leads, emphasis on J waves 19 IVF patients, 412 controls Nam (2008) Inferolateral leads, 206 IVF patients, Haissaguerre (2008) Inferolateral leads Mainly precordial leads 8 2081 Klatsky (2003) Mainly precordial leads One or more leads Mainly precordial leads Precordial leads Precordial leads Distribution of ERP Precordial leads Various case reports 75 65 48 Wesserburger (1961) Kambara (1976) 5 Fenichel (1962) 54 Goldman (1960) No. of subjects 25 Grusin (1954) Authors (year) Goldman (1953) controls Increased prevalence of ERP among patients with IVF than leads ERP more common in IVF patients, especially ERP in multiple controls Increased prevalence of ERP among patients with IVF than bradycardia and immediately prior to the onset of VF ERP in patients with IVF. Augmentation of J waves during ERP not associated with hospitalization or arrhythmias Vast majority with ERP remained free of heart disease Vast majority with ERP remained free of heart disease ERP is a normal variant disease ERP persisted and none of the subjects developed heart ERP persisted in majority of subjects in the follow-up Main findings 2-4mm J point elevation in mid precordial leads a normal variant Table 1. Summary of the publications of prognosis of ERP prior to this thesis. 2.6 QRS complex abnormalities Abnormal ventricular conduction, which is manifested as variations in the QRS complex, is known to have the potential to cause life-threatening brady- and tachyarrhythmias, such as heart block and ventricular fibrillation. Nonspecific QRS prolongation, bundle branch blocks, peri-infarction block and fragmentation of the QRS complex are briefly presented within this literature review, as both of the proposed J wave syndromes also possess theories of depolarization as the underlying pathophysiologic mechanism. Figure 7 illustrates theoretical examples of various QRS complex abnormalities. 2.6.1 Prolonged QRS duration and bundle branch blocks Nonspecific prolongation of the QRS complex (QRSd) have been shown to predict adverse outcomes in patients with various cardiac conditions, including coronary artery disease and congestive heart failure (Bode-Schnurbus et al. 2003, Bongioanni et al. 2007, Desai et al. 2006, Elhendy et al. 2005, Iuliano et al. 2002, Kashani & Barold 2005, Oikarinen et al. 2004, Teodorescu et al. 2011a, Wang et al. 2008, Zimetbaum et al. 2004). Recently, a QRS duration ≥ 110ms, in the absence of complete or incomplete bundle-branch block was claimed to be a strong predictor of cardiac and arrhythmic death in a sample of middle-aged general population subjects (Aro et al. 2011), in which the prevalence of this nonspecific intraventricular conduction delay was 0.6%. Another recent study reported similar findings, i.e. QRS >100ms including incomplete branch blocks, was a strong predictor of SCD among middle aged men, and each 10ms increment in QRS duration was associated with a 27% higher SCD risk (Kurl et al. 2012). These observations indicate that QRSd has significant importance in the prognosis of individuals in the general population. The pathophysiology of QRSd could be anticipated to include various forms of cardiomyopathies, which have remained clinically absent but often cause conduction abnormalities. A genetic predisposition is also possible, since several loci of the genome have been associated with QRS duration and ventricular conduction (Sotoodehnia et al. 2010). For some time, right bundle branch block (RBBB) has been considered as a normal variant or at maximum as a marker of marginal risk if it occurred in the absence of cardiac disease (Fahy et al. 1996, Fleg et al. 1983, Miller et al. 2008, 48 Rotman & Triebwasser 1975). It presents as a prolonged QRS >120ms and a typical RSR’ pattern in V1 due to slowed conduction in the right ventricle. In a recent Finnish study, RBBB was not associated with increased mortality rates among a general population sample with a long follow-up (Aro et al. 2011). Conflicting results have been reported on the prognostic significance of left bundle branch block (LBBB). LBBB manifests itself as prolonged QRS >120ms and broad monomorphic S wave in V1. The pattern in an asymptomatic state has been associated with higher mortality rates in several studies (Eriksson et al. 2005, Miller et al. 2005), including the Finnish study previously mentioned (Aro et al. 2011), while others have reported negative results (Eriksson et al. 1998, Fleg et al. 1983). 2.6.2 Peri-infarction block Peri-infarction block is an ECG abnormality, which usually is indicative of latent coronary artery disease. It develops following myocardial infarction and leads to delayed activation in the region of the infarction. It is frequently associated with extensive scarring of the left ventricle and ventricular late potentials in signal averaged ECG. (Castle & Keane 1965, Flowers et al. 1990, Scherlag et al. 1985a) The pattern is known to increase the risk of malignant ventricular arrhythmias and sudden cardiac death (Flowers et al. 1990, Scherlag et al. 1985b). In the ECG, the peri-infarction block manifests itself as Q waves as a sign of myocardial infarction and slurring or prolongation of the terminal QRS complex. The QRS interval is either normal or borderline prolonged. (First et al. 1950)Due to the terminal QRS changes, peri-infarction block has been proposed to overlap with the identification of J waves (Littmann 2010). 2.6.3 Fragmented QRS complex The fragmented QRS complex (fQRS) includes several different morphologies of the QRS complex, such as the presence of an additional R wave (R’) or notching in the nadir or the R or the S wave in two contiguous leads corresponding to a major coronary artery territory (Das et al. 2006). It has been demonstrated to represent a myocardial scar in patients with coronary artery disease (Das et al. 2008, Homsi et al. 2009), but is also encountered in several other cardiac conditions, such as ARVD and Brugada syndrome (Morita et al. 2008b, Peters et al. 2008, Peters et al. 2012). Generally, the mechanism of fragmentation of the 49 QRS complex is explained by inhomogeneous activation of the ventricles (Flowers et al. 1969, Varriale & Chryssos 1992). There is no unequivocal data on the prevalence and prognosis of fQRS among general population, but there is growing evidence to support its relation to ventricular arrhythmias and SCD in patients with various cardiac conditions. In CAD patients, the all-cause mortality and cardiac event rate have been shown to be significantly higher in patients with fQRS than those without this abnormality (Das et al. 2007). Another study reported a two-fold higher risk of recurrent cardiac events in patients with fQRS compared to controls among patients with Q wave myocardial infarction (Pietrasik et al. 2007). Furthermore, patients with fQRS have been shown to exhibit a significantly decreased time to death in acute coronary syndrome (Das et al. 2008, Das et al. 2009), and the presence of ≥3 leads with fQRS have been associated with significantly increased rates of cardiac death and hospitalization due to heart failure in patients with prior myocardial infarction (Torigoe et al. 2012). In patients with dilated and ischemic cardiomyopathy, the inferior presentation of fQRS or J waves have predicted higher risk of sudden cardiac death (Brenyo et al. 2012, Pei et al. 2012). These results refer to a possibility of overlapping between J waves, manifested as notching of the terminal QRS, and end-QRS fragmentation (Bastiaenen & Behr 2012). Fig. 7. Theoretical examples of various QRS-ST junction abnormalities. 2.7 Summary of the literature In summary, even in the present era, the traditional standard 12-lead electrocardiogram is an important tool in risk stratification of asymptomatic individuals. Sudden cardiac death represents a major health issue, and since the majority of SCDs occur among low risk individuals in the general population, the 50 quest for better risk stratification is clearly warranted. As presented in this concise literature review, ECG has an important role in the recognition of many cardiac conditions and syndromes associated with malignant ventricular arrhythmias. However, there is still much to be clarified, and these gaps in our knowledge can only be elucidated by further investigation of many electrocardiographic parameters. Early repolarization in the ECG has been one of these controversial issues. Several studies investigating early repolarization have pointed to a benign nature for decades, but the recent emphasis on J waves, especially in inferolateral leads, has challenged this old belief. The arrhythmogenic potential of J waves had been recognized earlier in the Brugada syndrome, but the prevalence and prognostic significance of inferolateral J waves in the general population were unknown prior to this thesis. 51 52 3 Aims of the study After the initial case-control studies pointing to the potential arrhythmia risk of subjects with early repolarization ECG pattern, a study protocol was designed to investigate the phenomenon of ERP in the general population. The primary aim was to evaluate the prevalence and prognosis of such ECG pattern, and thereafter to characterize the features of the ECG pattern in more detail. The specific aims for the study were set as follows: I II III IV V VI To assess the prevalence and prognostic significance of the early repolarization pattern in a large Finnish general population with a long follow-up (study I). To identify the clinical features of early repolarization pattern in the general population (study II). To assess the heritability of early repolarization pattern in the general population (study II). To identify the electrocardiographic features of the early repolarization patterns that are associated with arrhythmia risk in the general population (study III). To assess the significance of the early repolarization pattern during acute ischemic coronary events in the general population (study IV). To identify common genetic variants influencing the early repolarization pattern by performing a meta-analysis of genome-wide association studies (study V). 53 54 4 Study populations 4.1 Study population A In the first and third study of the thesis, the study population was composed of a sample of 10 957 men and women aged 30 to 59 years (52.3% men, mean age 44±8 years) drawn from different geographic areas of Finland, who participated in the Social Insurance Institution’s Coronary Heart Disease Study (CHD study) in 1966-72 (participation rate 89% of 12 310 invited subjects). The CHD study was part of a large, prospective Mobile Clinic Health Survey in which the participants underwent a thorough clinical examination including a resting 12lead ECG. The cohort was representative of the middle-aged Finnish population, since the population groups consisted of either the whole population or a random sample of the population of a certain geographic area. Furthermore the geographical areas were chosen from regions with varying death rates. The mortality data was determined when the follow-up was completed in 2007. It was necessary to exclude 93 subjects who had missing data or whose ECG traces were unreadable. Thus, the study population of the first study consisted of 10 864 subjects (52% men, mean age 44±8 years). 4.2 Study populations B and C In the second study, the study population composed of two general population cohorts: the Framingham Heart Study (FHS, study population B) and the Health 2000 Study (H2K, study population C). These populations were also used in the GWAS meta-analysis (study V). The Framingham Heart Study (FHS) is a prospective community-based study initiated in 1948 in the town of Framingham, Massachusetts, United States, to evaluate potential risk factors for coronary heart disease, and 4 095 subjects with available ECGs were included. Originally, a listing of all residents 20 years of age and older, living in Framingham, was used as the basis of the sampling of subjects. The Health 2000 (H2K) Study is a cross-sectional, population-based cohort of 8028 Finnish adults aged ≥30 years, sampled by the Finnish Population Information System in 2000-2001. Full details of the sample procedures have been described elsewhere (Aromaa & Koskinen 2004, Heistaro 2008). Briefly, it 55 included similar interviews and extensive health examinations as utilized in the protocol of the Framingham Heart Study. Both cohorts, the Framingham Heart Study and the Health 2000 Study, were representative of the general population. The final study population of the second study of this thesis consisted of 3 955 subjects from FHS and 5 489 from H2K after exclusions of missing data, poor ECG quality, various unsuitable ECG features for ERP analysis, age over 80, and non-Caucasian race. For the fifth study, 3726 participant from FHS and 2082 from H2K with both GWAS and ECG available were studied. 4.3 Study population D The study population of the fourth study consisted of cases and controls from the same regional area in Northern Finland. The cases were subjects from the Finnish Genetic Study of Arrhythmic Events (FinGesture), which was initiated in 1998, and by 2009, had gathered data from 2732 consecutive victims of out-of-hospital sudden unexpected death in the Oulu University Hospital District, with all of these deaths being autopsy-verified to have been due to an acute coronary event. The SCD victims consisted of those with a witnessed sudden death within 6 hours of onset of the forewarning symptoms. For those with unwitnessed SCD, the time definition was extended to within 24 hours of the time when the victim had been last seen alive and in a normal state of health. Those with any other cause than SCD (accidental or suicidal intoxication) during an acute coronary event were excluded, as well as those subjects with signs of severe heart failure (prior history of hospitalization due to congestive heart failure or acute pulmonary edema at autopsy). The controls consisted of 1565 survivors of an acute coronary event, confirmed by the current guidelines (positive troponin assay), from the same time period and the same geographical area from which the SCD victims were gathered. As the aim was to investigate the early repolarization pattern, subjects were included if a prior standard 12-lead ECG unrelated to the event (SCD or acute coronary event) had been recorded and was available for interpretation. Sixteen percent of 2732 victims of SCD and thirty four percent of 1565 survivors had prior ECG traces available. Consequently, the study population of the fourth study consisted of 432 victims of sudden death and 532 controls. 56 4.4 Study population E In the fifth study, a meta-analysis of genome wide association studies was performed and in addition to populations B and C, another general population sample from Germany was included. This Cooperative Health Research in the Region of Augsburg (KORA) F4 Study was conducted in 2006 to 2008 and included men and women aged between 32 and 81 years. After identical exclusions as applied to populations B and C, 1674 subjects with available GWAS and ECG data were studied. 4.5 Study population F In the third study, two separate athlete samples were examined for ERP prior to the re-evaluation of study population A. ECG tracings were recorded and analyzed from 62 volunteer Finnish young male athletes, aged 13 to 15 years (100% men) and from 503 competitive athletes from the University of Miami, United States. Fifty-one percent of the US athletes were men, with ages ranging from 17 to 24 years, and 34% were african descent. 57 58 5 Study methods 5.1 Study I. Prevalence and long-term prognosis of early repolarization pattern in general population The aim of the first study was to assess the prevalence and long-term prognosis of early repolarization pattern in leads other than V1-V3. At the baseline examinations in 1966-72 all of the studied subjects completed a questionnaire concerning their health habits, risk factors for cardiovascular disease, known diseases and medications. In addition to resting 12-lead ECG and the health questionnaire, blood pressure, body-mass index and serum cholesterol were measured. It should be noted that usage of beta-blockers or other medications causing potential changes in cardiac repolarization was extremely rare during that era. The ECGs were recorded at a paper speed of 50 mm per second and a specially trained nurse measured the basic ECG parameters, such as SokolowLyon index at the time of baseline examinations. The majority of the participants in the original Mobile Health Clinic Survey also participated in re-examinations between 1973 and 1976, in which resting ECGs were recorded in similar way to the baseline evaluation. All of the participants were automatically followed-up until the end of 2007, as Finland maintains extensive administrative registers and every death in the country is recorded. Less than 2% of the studied subjects had moved abroad, but even then in majority of the cases, the survival status could be determined. The causes and rates of death were determined by examining death certificates from Statistics Finland. The mean follow-up was 30±11 years. The primary end point used in the publications was death from cardiac causes, and the secondary end points were death from arrhythmia and death from any cause. Another important feature was the arrhythmic mortality, and all deaths classified as cardiac death were reviewed to identify the cases of sudden death due to arrhythmia. Experienced cardiologists, who were unaware of the ECG data, performed this analysis. After reviewing the data available from death certificates and hospital records, the cardiac deaths were classified as either or not associated with arrhythmia. Definite and probable arrhythmic deaths were included according to the definitions described in Cardiac Arrhythmia Pilot Study (Greene et al. 1989), in which the arrhythmic death was defined as a spontaneous cessation of respiration and circulation with loss of consciousness in one of the following 59 situations: witnessed and instantaneous without new or accelerating symptoms, witnessed and preceded or accompanied by symptoms attributable to myocardial ischemia in the absence of heart failure, witnessed and preceded by symptoms attributable to cardiac arrhythmia, and unwitnessed but without evidence of another cause. If severe congestive heart failure was present, death was not included in the arrhythmia category if death from heart failure was considered to be likely within 4 months before the fatal event. All 10,957 baseline ECGs were re-analyzed. QRS duration and PQ-interval were measured, as well as the presence of early repolarization ECG pattern and Tinversions were recorded by five physicians blinded to the original measurements and outcome data. ERP was defined as J-point elevation in at least two consecutive leads in the inferior (II, III, aVF) or lateral (I, aVL, V4-V6) leads, with or without ST-segment elevation. Terminal QRS notching (a positive J deflection inscribed on the S wave) and slurring (a smooth transition from QRS to ST-segment) of at least 0.1 mV were considered as early repolarization. Additionally the amplitude of the J wave at its highest point was measured and categorized as ≥0.1 mV or >0.2 mV. Post-extrasystolic beats were not used. The ECGs with positive ERP grading were double-checked and the grading was established by consensus. All of the available 542 re-examination ECGs from subjects who presented ERP at baseline were also analyzed. In order to estimate interobserver and intraobserver variations, 556 ECGs were analyzed twice. In the comparative analyses, prolonged QTc interval was defined as at least 440 ms for men and at least 460 ms for women, and left ventricular hypertrophy as SokolowLyon index >35 mm. 5.2 Study II. Clinical features and heritability of early repolarization pattern in the general population In the second publication, the aim was to identify the clinical correlates and to assess the heritability of ERP in general population. At the baseline examinations of the Framingham and Health 2000 survey participants, extensive studies were performed in each individual, including standard resting ECG, blood samples, medical history, physical examination, etc. In the FHS sample, the 25 mm/s paper ECGs were read manually by two investigators, and in the Health 2000 sample, the measurements were performed from digital 50 mm/s ECGs (a single median heart beat for each 12 leads 60 separately) by another two physicians. Post-extrasystolic beats were not used. All interpretations were performed blinded to the clinical data. Within the FHS reviewers, 100 ECGs were cross-analyzed to assess the interobserver variability. In order to estimate the interobserver variability there was cross-analyzing of all available ECGs within the Health 2000 by the reviewers. 5.3 Study III. Long-term prognosis of various early repolarization patterns and the significance of ST-segment in general population The aim of the third publication was to assess the significance of different ERP variants and the significance of ST-segment associated with the pattern, i.e. to identify the subjects with the highest risk of arrhythmic death. Three different cohorts were examined in this publication. The athlete samples were 62 young and healthy volunteer Finnish ice hockey players from Oulu, Finland, and 503 competitive college athletes from University of Miami, United States. The main study population used for mortality assessment was the same middle-aged general population sample as used in study I (study population A). First, a pilot study was performed with the athletes in Finland and United States to assess the prevalence and different patterns of ERP. The ECGs of Finnish athletes were read by two readers independently and the US athletes by a single reader. Thereafter, all the 630 ECGs of middle-aged subjects with positive grading for ERP were re-analyzed to assess the prevalence and long-term prognosis of those patterns and in the general population. Fifty-four subjects from population I had to be excluded from the analyses, as their ST-segment status could not be unequivocally determined due to the poor quality of the ECG tracing or no consensus could be reached. The different patterns of ERP were defined as follows. The J waves were categorized into notching or slurring, similar to the notch/slur definition used in the first publication. Furthermore, the ST-segments following the J waves were categorized into rapidly ascending or horizontal/descending. The ascending STsegment was defined as >0.1 mV ST-elevation 100 ms after the J-point, and consistently the ST-segment was defined as horizontal/descending if this criterion was not met, i.e. ST-elevation was ≤0.1 mV. The Finnish and US ECGs were recorded at paper speed of 50 mm/s and 25 mm/s, respectively. 61 5.4 Study IV. The significance of early repolarization pattern in acute coronary events In the fourth publication, the aim was to investigate the significance of ERP during an acute ischemic coronary event with a case-control layout. First, the electrical medical records were retrospectively reviewed to identify those subjects with a prior ECG available before the event (sudden death or myocardial infarction). If several ECGs were available, the most recent one was utilized in the analyses. Simultaneously, basic demographic parameters and medication data were collected as well as known diseases from those subjects either from digital medical records, medicolegal autopsy records, or from standardized questionnaires completed by the victims’ closest relatives. Consequently, the information in this study represents the known medical history of the subjects, prior to the acute index coronary event. Medication data could not be reliably obtained from 209 (22%) subjects. The ECGs, all recorded at paper speed of 50 mm/s, were analyzed by three investigators independently and blinded to the clinical data and outcome status, and consensus readings were performed if necessary. ERP and the following dominant ST-segment types were defined as in studies I and III. 5.5 Study V. Identification of common genetic variants influencing early repolarization pattern The aim of the fifth study was to conduct a meta-analysis of genome-wide association studies to identify common genetic variants that influence ERP. Three large community based cohorts (study populations B, C and E) were used. The ECG analyses in study populations B and C are described above in Study II. Identical measurements, classifications and exclusion criteria were applied also for study population E. GWA studies in participants with and without ERP were analyzed by logistic regression assuming an additive genetic model and individual cohort results were meta-analyzed. Those findings that reached p-value 1 x 10-5 in independent individuals were further investigated by direct genotyping or in-silico analysis of genome-wide data. 62 5.6 Statistical analysis In all studies, continuous values are presented as mean ± standard deviation (SD) and categorical values as number of subjects or percentages. In categorical variables, groups were compared with Chi-square test. In continuous variables, the groups were compared with the independent samples t-test (two group comparison) or analysis of variance (multiple group comparison). P values were two-tailed, and a P value <0.05 was considered statistically significant. In studies I and III, hazard ratios and 95% confidence intervals were computed with Cox proportional hazard models. These models were adjusted with covariates that had a known association with cardiovascular mortality. The covariates included age, gender, systolic blood pressure, body mass index, heart rate, smoking and ECG signs of left ventricular hypertrophy or coronary artery disease. Kaplan-Meier estimates were used for overall, cardiac and arrhythmic mortality. In the second study, a multivariable logistic regression model was constructed to identify independent determinants of ERP status, forcing in age and sex, using a forward selection procedure (entry p<0.05 in the initial models, retention p<0.05). SAS proc logistic (version 9.2) was used for model selection in both cohorts. Within each of the used stratified samples in the heritability analyses of the second study, odds ratios were obtained predicting the ERP status of the sibling of the index individual using logistic regression, with and without adjustment for age and sex. In the fourth study, logistic regression analysis was used to assess the significance of predictors of SCD between groups after adjustments for age, gender, smoking, body mass index, prior myocardial infarction, history of coronary artery disease, and medication (beta blocker, statin, angiotensin II receptor antagonist and nitroglycerin in model 2). The Hosmer and Lemeshow test was used to assess the goodness-of-fit of the logistic regression models. In the fifth study, primary analyses were performed separately at each site by using logistic regression assuming an additive genetic model. Models were adjusted for age, gender, and RR interval. The meta-analysis of site-specific association results was performed by using the meta-analysis tool Meta Analysis Helper (Willer et al. 2010), and finally the GWAS and replication stage were combined for overall effect estimate. The statistical analyses were performed with SPSS 16.0 (I, III, IV) (SPSS Inc, Chicago IL), and SAS software v 9.1.3 (I, III) and 9.2 (II) (SAS, Cary, NC). 63 64 6 Results 6.1 Prevalence and prognostic significance of early repolarization pattern in a large Finnish general population with a long followup (I) In the first epidemiological study (I), 630 persons were identified (5.8% out of 10,864 middle-aged subjects) displaying the early repolarization pattern, including 3.5% in the inferior and 2.4% in the lateral leads. Higher amplitude J waves (>0.2 mV) were observed in 67 subjects (0.6%). Those with ERP in the inferior leads were more often male, smokers, had a lower body mass-index, lower blood pressure, a lower resting heart rate, a shorter QTc interval, a longer QRS duration, and were more likely to have ECG signs of coronary artery disease according to Minnesota-codes. In contrast, those with ERP in the lateral leads were more often male, had a lower resting heart rate, and were more likely to have ECG signs of left ventricular hypertrophy. Table 2 describes the characteristics of the groups at baseline. During a follow-up of 30±11 years, 6133 (56.5%) subjects had died. Of these deaths, 1969 (32.1%) were due to cardiac causes, and furthermore 795 (40.4%) of the cardiac deaths were classified as arrhythmic deaths. Before and after multivariate adjustments, those with ≥0.1 mV ERP in the inferior leads had an elevated risk of cardiac (adjusted relative risk [RR] 1.28, 95% confidence interval [CI] 1.04 to 1.59) and arrhythmic death (RR 1.43, 95% CI 1.06 to 1.94), but did not possess a higher rate of overall mortality. Those with the higher amplitude ERP (>0.2 mV) had an even higher risk of cardiac (RR 2.98, 95% CI 1.85 to 4.92) and arrhythmic death (RR 3.94, 95% CI 1.96 to 7.90). Those with ≥0.1 ERP in the lateral leads had an elevated risk of cardiac (RR 1.34, 95% CI 1.04 to 1.74) and had a higher rate of overall mortality (RR 1.19, 95% CI 1.02 to 1.39), but did not suffer an elevated risk for arrhythmic death. Table 3 presents the adjusted relative risks of prolonged QTc, left ventricular hypertrophy, and ERP. 65 66 Age (years) ‡ 87±9 10.2 1.1 2.2 Coronary artery disease (%) § Prior myocardial infarction (%) Prior angina pectoris (%) 2.3 1.1 9.7 87±8 408±28 30.8 8.1 4.3 82±12 139±22 0.7* 1.4 14.9* 89±7* 404±26* 35.0 7.7 2.7 80±12* 136±19* 74±14* 25.1±3.3** 6.51±1.23 42.6** 44.2±8.1 44.2** 384 * P value <0.05; ** P value <0.001 compared to group None 1.6 1.1 13.5* 88±7** 406±27 40.1** 7.5 4.0 82±13 137±20 74±14** 25.6±3.4* 6.53±1.26 36.1 44.5±8.3* 44.8** 630 Inferior or lateral leads § The criteria for the diagnosis of coronary artery disease corresponded to Minnesota codes 1.1 to 1.3, 5.1 to 5.2, 4.1 to 4.3, 7.1, and 7.4. 3.0 0.6 11.2 87±7 409±27 49.5** 6.9 5.8 84±14* 139±21 74±15* 26.4±3.7 6.58±1.28 26.1* 45.2±8.7* 45.2* 262 Lateral leads Early repolarization pattern ≥ 0.1mV Inferior leads † P values in this category were adjusted for age. ‡ P values in this category were adjusted for sex. Plus–minus values are means ± SD. QTc denotes QT corrected for heart rate. 408±28 31.2 ECG signs of LVH (%) †‡ QRS duration 8.2 Cardiovascular disease (%) †‡ QTc duration 4.3 82±12 Diastolic blood pressure †‡ Chronotropic medication (%) †‡ 138±22 Systolic blood pressure †‡ 76±15 25.9±3.9 25.9±3.9 76±15 6.50±1.32 6.50±1.32 33.9 44.0±8.5 34.0 44.0±8.5 Heart rate (beats/min) †‡ Body-mass index †‡ Total cholesterol (mmol/liter) †‡ Current smoker (%) †‡ 51.5 10 234 None 52.4 10 864 No. of subjects Male gender (%)† All subjects Characteristics Table 2. Characteristics of the subjects at baseline (I). Table 3. Adjusted relative risk of death from cardiac causes (I). Variable No. Relative risk Prolonged QTc interval 668 1.20 (1.02-1.42) 0.03 3410 1.16 (1.05-1.27) 0.004 Electrocardiographic LVH P value ERP ≥ 0.1mV in inferior leads 384 1.28 (1.04-1.59) 0.03 ERP > 0.2mV in inferior leads 36 2.98 (1.85-4.92) <0.001 Relative risks are presented as RR (95% confidence intervals), and adjusted for age, sex, body mass index, heart rate, smoking status, systolic blood pressure, presence of left ventricular hypertrophy or coronary artery disease on electrocardiography. QTc indicates QT interval corrected for heart rate, LVH left ventricular hypertrophy, and ERP early repolarization pattern. Furthermore, the cumulative survival plots (Figure 8) showed that the mortality rates between those with and without ERP started to diverge only after a notably long follow-up. This study illustrated that ERP was a quite common finding and that it was associated with adverse cardiovascular outcomes in the general population. Figure 9 illustrates an example ECG of a middle-aged subject with ERP in the inferior leads. 67 Fig. 8. Kaplan-meier survival curves (I). 68 Fig. 9. Example ECG of a middle-aged subjects carrying inferior ERP followed by horizontal ST segments. 69 6.2 The clinical correlates and heritability assessment of early repolarization pattern in general population (II) The second study (II) identified a total of 423 persons (4.5% out of 9,444 subjects from general population samples) displaying ERP. The pattern was present in 243/3,955 (6.1%) of FHS subjects, and 180/5,489 (3.3%) of H2K subjects. After stepwise selection using all significant covariates from age- and sexadjusted models, male sex, increased RR interval, decreased systolic BP, decreased Cornell voltage, and increased Sokolow-Lyon voltage were independently associated with ERP status on multivariable regression in FHS cohort. Correspondingly, younger age, male sex, shorter RR-corrected QT interval, shorter QRS duration, lower Cornell voltage, higher Sokolow-Lyon index, and lower systolic blood pressure were independently associated with ERP status in the H2K sample. A family history of SCD or syncope was no more commonly reported in subjects with ERP. The full descriptive data is presented in Table 4, and the results of the multivariate regression model are shown in Table 5. In the Health 2000 sample, the prevalence of ERP was decreasing with increasing age, and there was evidence for a modest age by sex interaction (p=0.048). The prevalence of ERP by gender in different decades of age in the Health 2000 sample is presented in Figure 10. With respect to the heritability assessment in the Framingham sample, there were 1,111 sibling pairs available for analyses. Siblings of those with ERP had an ERP prevalence of 11.6% vs 6.1% in those without the pattern (sibling occurrence risk ratio 1.89), and furthermore, siblings of ERP-positive individuals exhibited an increased odds of ERP before adjustments (OR 2.20, 95% CI 1.00 to 4.90). This study showed quite similar prevalence of ERP as in study I, and demonstrated that ERP has a strong association with clinical factors. ERP also seemed to have a heritable basis, although other confounders such as age, sex or exercise habits could partly explain ERP among siblings. 70 71 120 (31.6) 78.8 (9.3) 121 (12.8) LDL cholesterol, mg/dL Diastolic BP, mmHg Systolic BP, mmHg 73 (4.5%) 140 (114) Triglycerides, mg/dL Beta blocker medication 46.6 (12.5) HDL cholesterol, mg/dL 19 (1.2%) 281 (17.2%) Cardiovascular disease Current smoker 4 (0.2%) Coronary heart disease Hypertension 68 (4.2%) Sokolow-Lyon, mV 10 (0.6%) 2.15 (0.57) 371 (22.7%) Cornell voltage, mV History of AF 1.27 (0.46) QRS duration, msec Diabetes mellitus 414 (21.9) 91.0 (9.51) QTc interval, msec 28.1 (4.7) 155 (22.7) Weight, kg PR interval, msec 89.2 (16.1) Height, m BMI, kg/m2 1004 (164) 1.78 (0.07) RR interval, msec 40.7 (8.7) 1 638 No. of subjects Men Age, years ERP- Characteristic 6 (3.3%) 117 (10.4) 75.2 (8.9) 118 (31.3) 114 (87.2) 48.4 (12.3) 20 (10.9%) 0 (0%) 0 (0%) 3 (1.6%) 4 (2.2%) 22 (12.0%) 2.64 (0.72) 1.07 (0.53) 92.4 (9.6) 406 (21.9) 155 (24.9) 26.8 (4.4) 84.2 (15.2) 1.77 (0.06) 1076 (189) 37.7 (8.8) 184 ERP+ 81 (3.9%) 113 (14.5) 72.8 (9.2) 105 (29.9) 97.6 (63.0) 60.9 (16.0) 303 (14.6%) 15 (0.7%) 5 (0.2%) 9 (0.4%) 56 (2.7%) 248 (12.0%) 1.87 (0.51) 0.84 (0.38) 84.2 (8.8) 426 (21.8) 143 (21.3) 26.1 (6.1) 70.4 (16.9) 1.64 (0.06) 966 (153) 40.1 (8.8) 2 074 ERP59 ERP+ 1 (1.7%) 109 (10.4) 71.0 (7.3) 101 (29.3) 82.1 (35.7) 63.8 (14.9) 10 (17.0%) 0 (0%) 0 (0%) 0 (0%) 0 (0%) 1 (1.7%) 2.15 (0.67) 0.66 (0.4) 85.3 (7.4) 422 (20.9) 141 (19.9) 24.7 (6.1) 67.33 (18.1) 1.65 (0.06) 1030 (174) 36.4 (9.2) Women Framingham Heart Study Table 4. Descriptive data of subjects in the Framingham and Health 2000 (II). 336 (14.7%) 136 (18.5) 85.0 (10.6) 149 (40.1) 162 (111) 46.0 (12.5) 809 (35.3) 156 (6.8) 108 (4.7%) NA 154 (6.7%) 1171 (51.1%) 3.15 (0.78) 1.67 (0.54) 93.2 (9.1) 390 (21.8) 166 (28.5) 27.3 (4.1) 84.8 (14.4) 1.76 (0.07) 987 (170) 50.2 (12.1) 2 291 ERP137 ERP+ 11 (8.0%) 126 (13.9) 81.3 (9.6) 147 (40.3) 136 (92.2) 50.7 (14.5) 64 (46.7%) 4 (2.9%) 3 (2.2%) NA 4 (2.9%) 43 (31.4%) 3.93 (0.87) 1.38 (0.64) 91.7 (8.1) 374 (18.4) 165 (25.9) 25.2 (3.1) 79.4 (11.3) 1.78 (0.06) 1072 (167) 43.2 (10.8) Men 561 (18.6%) 132 (21.6) 80.0 (10.7) 139 (40.3) 123 (63.7) 55.9 (14.5) 732 (24.3%) 128 (4.3%) 56 (1.9%) NA 128 (4.2%) 1270 (42.1%) 2.66 (0.64) 1.34 (0.50) 86.4 (8.2) 403 (21.2) 158 (24.8) 26.8 (5.1) 70.7 (13.7) 1.63 (0.07) 961 (149) 50.8 (13.1) 3 018 ERP- Health 2000 Study 7 (16.3%) 121 (15.9) 74.6 (10.0) 131 (41.2) 104 (41.6) 54.2 (12.7) 16 (37.2%) 4 (9.3%) 1 (2.3%) NA 2 (4.7%) 10 (23.3%) 3.11 (0.74) 1.02 (0.58) 82.3 (7.1) 393 (20.6) 160 (22.6) 26.0 (4.3) 69.5 (13.4) 1.63 (0.07) 1000 (175) 47.0 (14.2) 43 ERP+ Women 72 41 (2.5%) 208 (12.7%) 21 (11.4%) 4 (2.17%) 1 (0.5%) ERP+ 519 (25.0%) 67 (3.23%) 108 (5.2%) 1 (1.7%) 0 (0%) ERP+ 21 (35.6%) Women ERP- NA NA 252 (11.0%) ERP- ERP+ NA NA 12 (8.8%) Men NA NA 524 (17.4%) ERP- Health 2000 Study NA NA 8 (18.6%) ERP+ Women for heart rate with Bazett’s formula. NA indicates data not available, and AF atrial fibrillation/flutter Numbers presented as mean (standard deviation) for continuous variables and number of cases (percentage) for dichotomous variables. QT interval corrected History of syncope Family history of SCD medication 42 (2.6%) QT prolonging Men ERP- Characteristic Framingham Heart Study Table 5. Independent predictors of ERP in multivariate regression model (II). Variable Odds ratio P Framingham Heart Study Age, per 10 years 0.84 (0.71-1.00) 0.05 Male, vs. female 4.10 (2.80-5.80) 1.0x10-14 Cornell voltage, per mV 0.41 (0.28-0.61) 1.4x10-5 Sokolow-Lyon, per mV 3.20 (2.50-4.10) 1.3x10-20 Systolic BP, per 10 mmHg 0.82 (0.72-0.92) 0.001 RR interval, per 100 msec 1.20 (1.10-1.30) 4.3x10-5 Health 2000 Study Age, per 10 years 0.95 (0.80-1.12) 0.51 Male, vs. female 2.74 (1.80-4.20) 5.6x10-6 Cornell voltage, per mV 0.22 (0.15-0.32) 3.9x10-16 Sokolow-Lyon, per mV 4.30 (3.40-5.40) 1.8x10-34 Systolic BP, per 10 mmHg 0.78 (0.69-0.88) 7.8x10-5 QTc interval, per 20 msec 0.61 (0.51-0.72) 1.0x10-8 QRS duration, per 10 msec 0.77 (0.63-0.95) 0.01 Odds ratios are presented as OR (95% confidence intervals) 73 Fig. 10. Prevalence of ERP by gender in the Health 2000 study (II). 74 6.3 The long-term significance of ER ECG pattern subtypes and ST-segment morphology (III) The sequel study (III) to the first observational study (I) explored the prognosis of different ERP subtypes after reviewing a large amount of ECGs from young and healthy athletes. Among the young Finnish athletes, the prevalence of ERP was 43.5% (27 out of 62 persons), and in the US athletes, the prevalence was 30.0% (151/503 persons). Both terminal QRS notching and slurring were present, and ST-segments following the J waves were elevated or rapidly ascending in 96.3% and 85.0% of Finnish and US athletes, respectively. In contrast, in the general middle-aged population, this ST-segment subtype was observed in only 28.5% of subjects with ERP. The dominant form of ST-segment in those middle-aged subjects with ERP was horizontal or descending (71.5%). ERP in the general population sample was classified as notched in 39.6%, slurred in 50.7% and undetermined in 9.7%. Table 6 presents the characteristics of the Finnish athletes. In the general population sample, those with ERP and horizontal/descending ST-segment were older, had a higher prevalence of ECG signs of coronary artery disease, and had a longer QRS duration compared with those without ERP. In contrast, those with ERP and a rapidly ascending ST-segment were more often male, were younger, had lower body mass index, lower heart rate, lower blood pressure, shorter QTc duration, and higher prevalence of ECG signs of left ventricular hypertrophy. Table 7 presents the baseline data in different groups. Subjects with ≥0.1 mV ERP and horizontal or descending ST-segments had a higher risk of sudden arrhythmic death (age- and sex adjusted HR 1.43, 95% CI 1.05 to 1.94) than those without the pattern. Subjects with ERP but rapidly ascending ST-segment did not display a similar elevated risk for arrhythmic death (HR 0.89, 95% CI 0.52 to 1.55). The same trends were found in subjects with inferior ER patterns, as subjects with inferior ERP and horizontal/descending STsegment had a HR of 1.72 (95% CI 1.22 to 2.41) for arrhythmic death after adjustments for age and sex, but in contrast, the ascending ST-segment type was not associated with adverse outcomes. Table 8 summarizes the risk of sudden arrhythmic death. Two example ECGs from subjects displaying ERP with horizontal ST-segments are presented in Figure 11. 75 Table 6. Characteristics of the subjects in the Finnish athlete population (III). Characteristic No ER No. of subjects 35 27 13.3±0.6 13.4±0.6 Age Males, % Heart rate. bpm QRS duration, ms ER + 100 100 71±17 68±11 88±8 84±12* QTc interval,ms 405±17 417±22* PR-interval, ms 145±25 142±20 Sokolow-Lyon index, mV 3.1±0.7 3.6±0.7* QRS-angle, degrees 71±21 74±47 J-point amplitude, mV 0 0.13±0.03** Numbers presented as mean ± standard deviation for continuous variables and percentage for dichotomous variables. QTc indicates QT interval corrected for heart rate. * P-value <0.05; ** P-value <0.001 Subjects with notched ≥0.1 mV ERP (terminal QRS notching independent of the ST-segment patterns) had an age- and sex adjusted HR of 1.58 (95% CI 1.07 to 2.32) for arrhythmic death. Correspondingly subjects with slurred ≥0.1 mV ERP (terminal QRS slurring independent of the ST-segment patterns) had an adjusted HR or 1.19 (95% CI 0.81 to 1.76) for arrhythmic death. In subjects with greater amplitude ERP (>0.2 mV), the notched subtype had an adjusted HR of 2.14 (95% CI 0.89 to 5.16) and those with the slurred subtype had a very high adjusted HR of 5.14 (95% CI 1.92 to 13.76). When only inferior location was investigated (≥0.1 mV), only notched type was significantly associated with arrhythmic death (HR 1.54, 95% CI 1.00 to 2.38). 76 Table 7. Adjusted characteristics of subjects with ERP and different ST segment patterns (III). Characteristic No ERP ERP with ascending ERP with horizontal No. of subjects 10 288 164 412 51.5 88.1** 57.1* 45.5 ± 8.2** / upsloping ST Males (%)1 Age (years)† / descending ST 44.0 ± 8.5 42.6 ± 7.9 Current smoker (%) ‡ 33.8 38.4 36.4 Cholesterol (mmol/l) ‡ 6.50 ± 1.32 6.48 ± 1.32 6.58 ± 1.21 BMI (kg/m2) ‡ 25.7 ± 3.6 25.9 ± 3.9 25.0 ± 2.9* Heart rate (bpm) ‡ 76 ± 15 70 ± 12** 75 ± 14 Systolic blood pressure (mmHg) ‡ 138 ± 22 134 ± 16* 138 ± 21 Diastolic blood pressure (mmHg) ‡ 82 ± 12 79 ± 12** 83 ± 13 Chronotropic medication (%) ‡ 4.3 3.2 4.2 Cardiovascular disease (%) ‡ 8.1 8.7 7.2 Electrocardiographic LVH (%) ‡ 30.8 60.6** 33.2 QTc duration (ms) ‡ 408 ± 28 400 ± 22** 409 ± 28 QRS duration (ms) ‡ 87 ± 8 87 ± 7 89 ± 7** ECG signs of CAD (%) ‡ 9.6 12.2 14.9** History of prior MI (%) ‡ 1.1 0.3 1.3 History of angina pectoris (%) ‡ 2.3 2.5 1.6 1 Adjusted for age; † Adjusted for sex; ‡ Adjusted for age and sex; ± values are means SD * P-value <0.05; ** P-value <0.001 compared to group No ERP CAD denotes coronary artery disease, MI myocardial infarction 77 Fig. 11. Example ECGs of two middle-aged subjects with inferior ERP followed by horizontal/descending ST segments (III). 78 Table 8. Risk of sudden arrhythmic death in different ERP groups according to STsegment morphology (III). Characteristic No. of subjects at No. of arrhythmic risk events 10 288 739 1 horizontal / descending ST 412 43 1.39 (1.02-1.89) ascending ST 164 13 0.87 (0.50-1.52) horizontal / descending ST 265 35 1.73 (1.22-2.44) ascending ST 90 8 1.05 (0.52-2.11) horizontal / descending ST 50 9 2.42 (1.25-4.69) ascending ST 10 1 0.96 (0.13-6.83) No ERP Adjusted HR Inferior / lateral ERP ≥ 0.1mV Inferior ERP ≥ 0.1mV Inferior / lateral ERP > 0.2mV HR denotes hazard ratio, ERP early repolarization pattern and ST ST-segment. Hazard ratios (95% confidence intervals) are adjusted for age, gender, body-mass index, heart rate, QTc duration, QRS duration, ECG signs of left ventricular hypertrophy or ECG signs of coronary artery disease. 79 Fig. 12. Cardiac and arrhythmic death survival plotted for ERP in middle-aged subjects. 80 6.4 Significance of early repolarization pattern during acute coronary events (IV) The fourth study revealed a total prevalence of 10.8% for ERP among 432 victims of sudden cardiac death and 532 survivors of an acute coronary event. The victims of SCD were more often men and smokers, were significantly younger, and had lower body mass index than the survivors of an acute event. Overall, the victims of SCD had a lower prevalence of medical admissions due to a prior myocardial infarction (23% vs 59%, p<0.001) or diagnosed coronary artery disease (44% vs 76%, p<0.001), as well as a reduced usage of cardiovascular medication, as shown in Table 9. ERP was significantly more common in victims of SCD than survivors (14.4% vs. 7.9%, p=0.001). Those with ERP had a lower prevalence of diagnosed diabetes mellitus (p=0.01) and diagnosed coronary artery disease (p=0.04), lower heart rate (p=0.02) and higher Sokolow-Lyon index (p<0.001) than those without the pattern. An inferior manifestation of ERP was more common in victims than survivors (6.7% vs 3.6%, p=0.03). Furthermore, a specific ERP pattern with a horizontal or descending ST-segment was associated with SCD (10.2% vs 5.3%, p=0.004), whereas ERP with ascending ST-segment was not (3.0% vs 2.3%, p=0.46). The electrocardiographic features are presented in Table 10. In a secondary analysis with a subset of patients without documented coronary artery disease (n=309), the results remained essentially the same, as ERP was more common in victims than survivors (16.9% vs 7.2%, p=0.04). Table 11 presents the odds ratios of sudden cardiac death in the evaluated variables. Unadjusted predictors of SCD during acute coronary event were male gender (OR 2.08, 95% CI 1.55 to 2.80), elevated heart rate (OR 1.90, 95% CI 1.46 to 2.47) and ERP (OR 1.85, 95% CI 1.23 to 2.80). In more specific terms, ERP with horizontal/descending ST-segment was associated with SCD (OR 2.04, 95% CI 1.25 to 3.34). In both multivariate models, ERP with horizontal/descending ST-segment remained as a significant predictor of SCD. The group with ERP and ascending ST-segment did not reach statistical significance either before or after adjustments. 81 Table 9. Demographic and clinical characteristics recorded before the event (IV). Characteristic Victims Survivors No. of subjects 432 532 Age, y P-value 66±11 69±12 <0.001 Male, % 79.4 64.9 <0.001 Body mass index 27±6 28±5 0.02 Current/ex smoker, % 43.3 32.3 0.001 <0.001 History of Prior myocardial infarction, % 23.1 59.1 Diabetes mellitus, % 26.6 26.3 0.93 Coronary artery disease, % 47.6 84.4 <0.001 Hypertension, % 50.0 54.0 0.22 Medication Beta-blocker, % 35.1 48.9 <0.001 Statin, % 11.3 32.0 <0.001 Acetylsalicylic acid, % 27.8 28.6 0.79 ACE inhibitor, % 16.0 20.9 0.09 ATII antagonist, % 3.6 10.9 <0.001 Nitroglycerin, % 14.9 25.2 <0.001 ACE indicates angiotensin-converting enzyme; ATII angiotensin II receptor. Table 10. Electrocardiographic variables (IV). Characteristic Victims Survivors No. of subjects 432 532 P-value QRS duration, ms 91±15 88±16 0.01 Heart rate, bpm 73±16 70±15 0.002 QTc, ms 421±38 419±35 0.39 27±9 27±8 0.83 0.03 Sokolow-Lyon, mV PQ-interval, ms 166±30 171±34 Atrial fibrillation, % 5.3 5.8 0.74 Pathologic Q waves, % 5.8 8.3 0.16 ERP in inferior and/or lateral leads, % 14.4 7.9 0.001 0.03 Location specific ERP ERP in inferior leads, % 6.7 3.6 ERP in lateral leads, % 6.0 3.6 0.07 ERP in inferior and lateral leads, % 1.7 0.7 0.13 ERP + horizontal/descending ST segment, % 10.2 5.3 0.004 ERP + ascending ST segment, % 3.0 2.3 0.46 Morphology specific ERP ERP indicates early repolarization pattern; bpm beats per minute 82 83 101 Early repolarization pattern 2.04 (1.25-3.34) 1.34 (0.61-2.98) 1.85 (1.23-2.80) 1.05 (0.73-1.52) 1.39 (0.83-2.33) 2.13 (1.29-3.52) 1.03 (0.46-2.32) 1.77 (1.15-2.70) 0.97 (0.67-1.41) 1.28 (0.89-1.84) 1.89 (1.45-2.46) 1.34 (0.79-2.28) … Adjusted OR* 2.15 (1.20-3.85) 1.12 (0.44-2.85) 1.73 (1.06-2.83) 0.94 (0.58-1.51) 1.27 (0.84-1.93) 1.65 (1.22-2.23) 1.59 (0.87-2.91) … Multivariate model 1† 2.29 (1.16-4.54) 1.67 (0.50-5.49) 2.02 (1.04-3.61) 1.30 (0.76-2.21) 1.17 (0.73-1.89) 1.79 (1.17-2.72) 1.28 (0.68-2.39) … Multivariate model 2‡ ATII receptor antagonist, and nitroglycerin medication. ‡Multivariate model 2 adjusted for age, gender, smoking, body mass index, prior myocardial infarction, history of coronary artery disease, beta blocker, statin, *Adjusted for age and gender; †Multivariate model 1 adjusted for age, gender, smoking, prior myocardial infarction, history of coronary artery disease. for women. Prolonged QRS ≥120ms; Elevated heart rate >80bpm; Left ventricular hypertrophy Sokolow-Lyon index >35mm; Prolonged QTc >450ms for men and >470ms No. indicates number of subjects. Odds ratios presented as OR (95% confidence intervals) 25 135 72 133 Prolonged QTc Left ventricular hypertrophy ERP + horizontal ST segment 1.36 (0.96-1.95) 377 Elevated heart rate ERP + ascending ST segment 1.90 (1.46-2.47) 61 Prolonged QRS duration 2.08 (1.55-2.80) 687 Male gender Unadjusted OR No. Variable Table 11. Odds ratios (OR) for sudden cardiac death during acute coronary event (IV). 6.5 Meta-analysis of genome wide association studies of early repolarization pattern (V) In the fifth study, the GWA study stage combined 7,482 subjects from the three cohorts, and the total prevalence of ERP was 6.0%. A total of 2,523,555 single nucleotide polymorphisms (SNPs) were analyzed for their association with an early repolarization pattern, with 8 loci being identified with p-values under 1 x 10-5. The most interesting SNP (rs17029069) was on chromosome 1p13.2 and maps to KCND3, which encodes the voltagegated potassium channel Kv4.3. Replication of the results of GWA study stage were attempted in independent samples of 7,151 individuals, but none of the identified eight SNPs could be replicated, and when meta-analyzed with primary GWAS results, none of the SNPs reached genome-wide significance (Table 12). 84 85 17 10 1 14 2 1 8 4 rs11653989 rs6585436 rs17029069 rs17097328 rs17012480 rs1541064 rs6988260 rs1382629 Closest gene(s) KIAA1211 FAM110B UBE2U LTBP1 C14orf177 KCND3 PDZD8, EMX2 MGAT5B, SEC14L OR 2.23 0.69 1.38 0.69 1.51 1.46 0.32 0.47 95% CI 1.56–3.18 0.59–0.81 1.20–1.59 0.58–0.81 1.27–1.78 1.25–1.69 0.21–0.50 0.36–0.61 P-value 9.82 x 10-06 8.92 x 10-06 8.54 x 10-06 6.97 x 10-06 1.71 x 10-06 8.50 x 10-07 4.88 x 10-07 6.87 x 10-09 OR 1.48 0.75 1.20 0.83 1.21 1.29 0.48 0.62 1.17–1.87 0.66–0.86 1.08–1.34 0.73–0.94 1.06–1.38 1.15–1.44 0.35–0.66 0.50–0.76 95% CI 9.12 x 10-04 1.72 x 10-05 6.80 x 10-04 3.45 x 10-03 4.03 x 10-03 8.28 x 10-06 6.11 x 10-06 2.80 x 10-06 P-value Overall after replication All results are adjusted for age, sex and RR-interval. Chr indicates chromosome, OR odds ratio, CI confidence interval, and SNP single nucleotide polymorphism. Chr SNP GWAS meta-analysis Table 12. GWA studies association results for the most significant SNPs (V). 86 7 Discussion 7.1 Key findings Study purpose 1. The assessment of prevalence and prognostic significance of early repolarization pattern in general population. There was a prevalence of 5.8% for ERP in the middle-aged general population and an association was found between ERP in the inferior leads and death from cardiac causes. These observations warranted further research on the topic. Study purpose 2. The identification of clinical features and the assessment of the heritability of the early repolarization pattern in general population. A strong association was observed between ERP and several physiological and clinical factors, such as gender, age, blood pressure and ECG voltages. There was also evidence for a heritable basis in general population, as ERP was significantly more prevalent in siblings of individuals with ERP than those without the pattern, which warranted further investigation into the genetic variants predisposing to ERP. Study purpose 3. The identification of electrocardiographic features of early repolarization patterns associated with arrhythmia risk in general population. It was demonstrated that ST-segment morphology variants associated with ERP separated subjects with and without an increased risk of arrhythmic death in middle-aged subjects. The arrhythmia risk at the population level was linked to the presence of ERP and a horizontal or descending ST-segment. Study purpose 4. The assessment of the significance of early repolarization pattern during acute ischemic coronary events in general population. A higher prevalence of ERP was observed in victims of SCD than in survivors of an acute coronary event in ECGs recorded prior to and unrelated to the event. The presence of ER ECG patterns, especially with horizontal or descending ST-segments, seemed to be a marker of increased vulnerability to fatal arrhythmia during acute myocardial ischemia. Study purpose 5. The identification of common genetic variants influencing early repolarization pattern (a meta-analysis of genome-wide association studies). A multicentre meta-analysis of GWA studies was performed in almost 15,000 individuals from community-based cohorts of European ancestry. It failed to reliably identify any genetic variants predisposing to ERP. There was presumably 87 insufficient statistical power, but more importantly these results suggested significant heterogeneity within the ERP phenotype. 7.2 Prevalence of early repolarization patterns (I-IV) Among the 10 864 middle-aged Finnish subjects (I), the overall prevalence of an early repolarization ECG pattern was 5.8%, including 3.5% in inferior and 2.4% in lateral leads. Comparably, in the Health 2000 Finnish general population sample of 5489 subjects (II), the ERP prevalence was 3.3%, and in the Framingham –sample of 3955 subjects from United States (II), the prevalence was 6.1%. Among the examined athlete cohorts (III), the total ERP prevalence was 43.5% in 62 young Finnish athletes, and 30.0% in 503 US collegiate athletes. Among the victims of SCD and survivors of acute coronary event (IV), the total prevalence of ERP was 10.8%. The distributions of ERP in inferior and lateral territory in different cohorts are presented in Figure 13. These numbers were in the same range or somewhat lower than in other recent studies, in which the prevalence of ERP has been reported to lie in a range between 3% and 24% (Haissaguerre et al. 2008, Haruta et al. 2011, Noseworthy et al. 2011a, Reinhard et al. 2011, Rosso et al. 2008, Sinner et al. 2010, Tikkanen et al. 2009a). In athletes, significantly higher prevalences have been reported (Cappato et al. 2010, Junttila et al. 2010, Noseworthy et al. 2011c). The prevalence of ERP seems to vary considerably, depending on the demographic features of the studied population; age, race, gender and physical activity. For instance, in the present studies the ages ranged from 30 to 60 years in study population B, in which an obvious age dependency of ERP was also observed. Interestingly, in the same study, by using uniform methods the prevalence of ERP was estimated to be twice as high in Americans as in the Finnish sample (6.1% vs 3.3%, respectively). One plausible explanation could be that the observation of ERP is dependent on whether the interpretation is done from paper or digital ECG, and whether the paper speed of the ECG is 25 or 50 mm/s. The higher prevalence of ERP in the fourth study can probably be explained by a higher prevalence of cardiac disease within this older population, as structural heart disease might itself increase the presence of ERP (depolarization hypothesis). Although there is less information available on the prevalence of ERP in patients with diagnosed structural heart disease, there is one study claiming that there is a prevalence of 20% for ERP in patients with chronic 88 coronary artery disease (Patel et al. 2010). A recent publication also highlighted the importance of definition, as the prevalence of ERP varied from 2% up to 30% in the same cohort sample depending on the kind of definition being used to categorize ERP (Heng et al. 2012). Fig. 13. Prevalences of J waves in investigated populations. One interesting observation from the first study was the finding that in more than three out of every four subjects, ERP was present in baseline ECG and also in a tracing recorded several years later. This suggested that ERP was a stable rather than a transient phenomenon, but was in conflict with the claim that the ERP amplitude and therefore the recognition of the whole pattern varies from day to day depending on heart rate and neurovegetative tone (Mizumaki et al. 2012). This divergence may well be explained by the possibility that there are structural abnormalities behind the ERP pattern observed in our middle-aged populations, in whom subclinical cardiac disease could be expected. 89 In athletes, the dominant ST-segment type following ERP was rapidly ascending: 96% and 85% of ST-segments were classified as ascending in the Finnish and US athletes, respectively. In the general population (study population A), ascending ST-segment was present in 28.5% of subjects with ERP, and horizontal/descending ST-segment in 71.5%. In a subsequent analysis, the ST segments were classified as follows in Health 2000 and Framingham cohorts: 42.4% horizontal and 57.6% ascending in the Health 2000, 64.2% horizontal and 35.8% ascending in the Framingham (Sinner et al. 2012). These results are consistent with more recent data from a French general population sample, in which the prevalence of ERP was 13.3%, and 66% of the ERP positives displayed horizontal or non-ascendant ST-segment (Rollin et al. 2012). 7.3 Demographic features, clinical correlates and heritability of early repolarization pattern (I-III) In studies I and III (study population A), differences were observed in the demographic characteristics between those with and without ERP. Subjects with ERP in the inferior leads were more often men and smokers, had a lower average resting heart rate, had a lower body mass index, had a lower blood pressure, had a shorter QTc interval and a longer QRS duration, and were more likely to have electrocardiographic signs of coronary artery disease (according to Minnesotacriteria) than those without the pattern (study I). ERP in lateral leads was associated with electrocardiographic evidence of left ventricular hypertrophy (I). The predomination of male gender in the early repolarization pattern has been observed in several other studies. Over two thirds of ERP carriers seem to be men, and they also exhibit greater J wave amplitudes as compared to women. (Haissaguerre et al. 2008, Rosso et al. 2008, Sinner et al. 2010, Tikkanen et al. 2009a) Subjects with various types of ST-segment after the J waves had certain differences in the baseline characteristics (III): Those with a rapidly ascending ST-segment were younger, their heart rate and blood pressure were lower, and they more commonly exhibited ECG signs of left ventricular hypertrophy. In contrast, subjects with horizontal or descending ST-segment after ERP were somewhat older and had longer QRS duration than those without ERP. Overall, these findings from the third study revealed that those subjects with ERP and rapidly ascending ST-segment were healthier and likely to be more physically 90 active than the others, leading to the presumption that the ascending ST-segment ERP variant might be a reflection of athlete-type ECG changes such as left ventricular hypertrophy voltage in lateral precordial leads. This concept has support from a study, in which ERP (mainly ascending ST-segment types, and predominantly in lateral leads) was found to have strong clinical associations among competitive athletes, as the prevalence of ERP increased after intense physical training (Noseworthy et al. 2011c). Furthermore, the ascending STsegment with ERP was the dominant form in healthy young athletes in another major study (Rosso et al. 2011), compared to patients with idiopathic ventricular fibrillation, among which the horizontal ST dominated. The second study (study populations B and C) established the independent predictors of ERP in a multivariable model in unselected, community-based cohorts. The following independent associations of ERP were identified; male gender, lower systolic blood pressure, higher Sokolow-Lyon index, and lower Cornell voltage. Other than the lower Cornell voltage, similar associations with ERP and male gender, lower blood pressure and higher Sokolow-Lyon index have been described elsewhere (Haissaguerre et al. 2008, Nam et al. 2008, Riera et al. 2008b). One noteworthy observation is the considerable age-dependency of early repolarization prevalence among males. The pattern is over-represented among young males compared to females, but the high prevalence seems to decline rapidly during middle age (Noseworthy et al. 2011b), which points to a strong influence of testosterone as a modifier of the ERP presence. A similar phenomenon has also been observed in Brugada syndrome (Shimizu et al. 2007). Among competitive athletes, the early repolarization pattern has strong associations with clinical factors. A causal role between exercise and ERP was observed in a cohort of collegiate athletes in whom intense physical training increased the prevalence of ERP from 37% to nearly 53% (Noseworthy et al. 2011c), but unfortunately no data is available of the distribution of ST-segment types in these athletes. In this set of athletes, the increased occurrence of early repolarization was independent of the echocardiographic findings related to athletes’ heart remodelling (Noseworthy et al. 2011c). The prevalence of early repolarization pattern was 30% in another collegiate athlete cohort in the US; of these 85% exhibited the ascending ST-variant (Junttila et al. 2010, Tikkanen et al. 2011). The second publication demonstrated that the ERP observed in the general population might have a heritable basis, with a doubled increased risk in siblings 91 of an individual with ERP. The sibling recurrence risk was attenuated in the older age stratum. Whether this reflects a chance effect due to the lower prevalence in this age group or a smaller contribution of genetic factors in relation to environmental and non-genetic clinical factors at older ages will require further investigation. Subsequently, similar observations have been published in other populations. In UK families, a 2.5-fold occurrence of ERP was observed in the offspring if at least one parent was carrying ERP (Reinhard et al. 2011), and subsequently the familial occurrence of ERP was also demonstrated among the relatives of sudden arrhythmic death syndrome probands (Nunn et al. 2011). Further discussion on the genetics of ERP will be continued in chapter 6.6. 7.4 Prognosis of early repolarization pattern and the significance of ST-segment morphology (I, III) As presented in the literature review, in the milestone publication of Haissaguerre and colleagues (Haissaguerre et al. 2008), there was an obvious overrepresentation of early repolarization ECG pattern in those with idiopathic VF (31%) compared to controls (5%), and the J waves were greater in amplitude in these cases than in the controls. Furthermore, those with extreme J waves (>5mm) had the highest occurrences of ventricular fibrillation episodes during the followup. These findings were shortly thereafter confirmed in multiple case-control studies (Nam et al. 2008, Rosso et al. 2008). Nam and colleagues reported ERP to be present in 58% in IVF patients vs 3% in controls. Rosso and colleagues similarly described a significantly increased prevalence of ERP in IVF patients when they were compared to matched controls (42% vs 13%). These findings of JERP occurring significantly more frequently among IVF survivors than in matched controls have now been confirmed in several other case-control studies (Abe et al. 2010, Derval et al. 2011, Merchant et al. 2009). The first study of this thesis revealed that the early repolarization pattern in the inferior leads was associated with a significantly increased risk of death from cardiac causes in middle-aged subjects among the general population. The results of this publication were consistent with the observations from previous casecontrol studies. The presence of high amplitude (>0.2mV) J waves in the inferior leads was a stronger predictor of death from cardiac causes than other well-known electrocardiographic risk markers, such as the QTc interval and signs of left ventricular hypertrophy. ERP remained an independent prognostic marker even 92 after adjustments for several other risk factors. The observations of inferior manifestation being a stronger predictor of cardiac death than lateral ER patterns and the prognostic value of the J wave amplitude were similar to phenomena also reported in survivors of primary ventricular fibrillation (Rosso et al. 2008). Although the average long-term prognosis of subjects with ERP was observed to be significantly worse than among the community, the mechanisms accounting for these higher mortality rates could not be investigated with the present kind of retrospective study design. An interesting discovery from the first study was the fact that the survival curves for cardiac mortality started to diverge 15 years after the baseline recordings (in the early 1980s) and continued to diverge at a constant rate throughout the follow-up period, even though the treatment and prognosis for patients with cardiac disease overall have improved during recent years. This led to the hypothesis that ERP may be a trigger of fatal events during myocardial ischemia (study IV). The third study was the first to show that inferolateral ERP in a standard 12lead ECG was not uniformly associated with an increased risk of arrhythmic death. It was demonstrated that differences in the ST-segment morphology following the J waves had important prognostic significance. The highest risk for arrhythmic death was observed with the combination of ERP in the inferior limb leads, high amplitude (>0.2 mV) J waves, and a dominant horizontal or descending ST-segment after the J waves. The rapidly ascending ST-segment after EPR was the dominant type encountered in athletes, and notably less common in the middle-aged general population. Moreover, this pattern was not associated with adverse outcomes. This finding was consistent with the concept that ERP in athletes is generally benign, although there might be some exceptions to this rule (Cappato et al. 2010). The present observations on the significance of the ST segment morphology have subsequently been confirmed in a series of IVF patients (Rosso et al. 2011) and in another general population sample (Rollin et al. 2012). Recently, this ERP pattern with horizontal/descending ST-segments in inferior or inferolateral leads was observed as a marker of higher risk of arrhythmic events in patients with malignant arrhythmias, if the pattern persisted during exercise (Bastiaenen et al. 2013). It was not possible to identify any parameter to distinguish between terminal QRS notching and slurring (independent of the following ST-segment morphology) in the analyses of the third study, as the results were contradictory. When all ERP cases were pooled together, subjects with a notched J wave had a 93 worse prognosis, but in other comparisons, the results were ambiguous. A high amplitude slurred ERP type in the inferior leads was associated with the highest risk of arrhythmic death in combination with the horizontal/descending STsegment. However, it should be acknowledged that the small sample size in these subgroups precluded any firm conclusions from being drawn from these analyses despite the fact that statistical significance was reached. Prior to this publication, terminal left precordial QRS notching had been reported to be more prevalent in the malignant (idiopathic ventricular tachycardia/fibrillation) than in the benign variants of ERP (Merchant et al. 2009). Similarly, a notched ERP was found to be more prevalent in patients with idiopathic ventricular fibrillation than in matched control subjects, whereas the presence of slurred ERP did not differ between the groups (Rosso et al. 2008). Furthermore, recently, ERP with the notching pattern followed by horizontal or descending ST segments was associated with a nearly 9-fold risk for suffering cardiovascular death in a French population (Rollin et al. 2012), whereas the slurring pattern was not associated with increased mortality rates. 7.5 Significance of early repolarization pattern during acute coronary events (IV) The fourth study observed that there was a significant and independent association in the general population sample between ERP, documented in a random ECG recording prior and remote to the event and the risk of sudden cardiac death at the time of an ischemic event. The risk for sudden death was doubled even after multivariate adjustments, with the highest risk being observed for ERP with horizontal or descending ST-segments, which supported the working hypothesis. Although there were demographic and clinical differences between the victims and controls, the association between ERP and SCD was independent of those factors suggesting, although not confirming, the vulnerability of individuals with ERP to suffer malignant arrhythmias during ischemia. Furthermore, the SCD victims had a lower prevalence of previously diagnosed cardiac disease in support the concept that ERP increases the risk of SCD in those subjects who suffer sudden death as their first manifestation of cardiac disease. The prevalence was notably high in this elderly population as compared to values from previous publications, which may well be explained by 94 the higher prevalence of cardiac disease in the studied subjects, if structural heart disease itself is thought to increase the presence of ERP. The results of the fourth study provided one plausible mechanistic link between ERP and risk of SCD: the presence of ERP might act as a factor facilitating the occurrence of fatal arrhythmia during an acute ischemic event. This proposal agrees with previous results from the first study, in which arrhythmic death among middle-aged subjects with ERP occurred at a relatively high age (>55 years). In this age group, SCD has been most commonly caused by an acute ischemic coronary event in the Finnish population (Hookana et al. 2011a). ERP with a non-ascending ST-segment has also been demonstrated to increase the incidence of cardiac events in vasospastic angina patients without fixed coronary artery disease (Oh et al. 2012). Vasospastic angina is a consequence of a coronary artery spasm, which provokes transient myocardial ischemia. Furthermore, the risk for ventricular fibrillation during the first 48 hours after myocardial infarction has recently been shown to be higher in patients who exhibited ERP with horizontal/descending ST segments in the pre-morbid ECG (Naruse et al. 2012, Rudic et al. 2012). In patients with ST-elevation myocardial infarction (STEMI), previous ERP was more common in patients with subsequent ventricular tachyarrhythmias (26% vs 4%), especially when present in the inferior leads (Patel et al. 2012). In another recent study examining acute STEMI patients, the presence of ERP was associated with ventricular arrhythmias and VF (Nakayama et al. 2013). When these observations are combined, one can speculate that patients with the early repolarization ECG pattern suffer an increased risk for ventricular fibrillation during events such as those present in myocardial ischemia that further shorten the action potential and increase the repolarization dispersion. 7.6 Genetics of early repolarization pattern (V) The fifth study of this thesis, a multicenter meta-analysis of GWAS attempted to identify if single nucleotide polymorphisms were associated and evaluated ERP in almost 15 000 individuals from the general population. Although the initial results of GWAS suggested promising results e.g. with KCND3, which encodes the voltage-gated potassium channel, none of the signals could be replicated. This might have been due to the lack of statistical power, but the negative results are more likely related to the possibility that the characteristic early repolarization 95 ECG pattern could be caused by multiple differential underlying changes. Further studies to identify genetic loci associated with ERP are required. Elsewhere, several independent studies have pointed to a heritable basis for early repolarization ECG pattern. In an investigation from UK families, a 2.5 fold occurrence of ERP was observed in the offspring if at least one parent was carrying ERP (Reinhard et al. 2011). In the second study of this thesis, similarly increased odds of ERP were observed in the siblings of ERP positive individuals (Noseworthy et al. 2011a). These estimates are within the same range as reported for other heritable ECG parameters such as the PQ and QT intervals (Hanson et al. 1989), for which chromosomal loci have been identified through genome wide association studies (Newton-Cheh et al. 2009, Pfeufer et al. 2009, Pfeufer et al. 2010). Furthermore, recently the familial occurrence of ERP was demonstrated among the relatives of sudden arrhythmic death syndrome probands (Nunn et al. 2011). However, it should be noted that these observational results of increased occurrence in siblings could also be explained by shared clinical and behavioural factors, such as exercise habits within the same family, since physical activity is known to strongly influence at least the ascending ST -phenotype of early repolarization pattern. Nevertheless, recently, the transmission of early repolarization was found to be compatible with an autosomal dominant form of inheritance in four families affected with ERP (Gourraud et al. 2013). Several mutations have been reported in individuals with ERP and sudden cardiac arrest, i.e. those with the early repolarization syndrome. A single study has described mutations in the cardiac L-type calcium channel among probands diagnosed with Brugada syndrome, idiopathic ventricular fibrillation and early repolarization syndrome (Burashnikov et al. 2010). The L-type calcium channel mutations have been previously associated with Brugada and short QT syndromes (Antzelevitch et al. 2007b, Cordeiro et al. 2009). KCNJ8 is nother susceptibility gene for J-wave syndromes; this gene encodes the cardiac ATP-sensitive potassium (KATP) channel (Barajas-Martinez et al. 2012, Delaney et al. 2012, Haissaguerre et al. 2009a, Medeiros-Domingo et al. 2010). Some other variants have been identified (Watanabe et al. 2011) affecting genes that encode sodium, potassium, and calcium currents, thus supporting the repolarization abnormality hypothesis as the underlying cause of ERP (Antzelevitch 2012). However, in the recent study on familial ERS (Gourraud et al. 2013), none of these mutations could be detected, suggesting further heterogeneity of ERS and IVF. 96 7.7 Early repolarization – overall evidence As already mentioned in the literature review, the terms early repolarization syndrome and early repolarization ECG pattern should be acknowledged as being distinct from each other. The syndrome of early repolarization should be used only to refer to patients with resuscitated cardiac arrest, documented VF or polymorphic VT, or possibly in the future, in relatives with genetic mutations associated with early repolarization syndrome. By contrast, the early repolarization pattern is an ECG variant commonly observed in several conditions and asymptomatic states. The vast majority of individuals investigated during this thesis work were asymptomatic carriers of ERP. This pattern seems to be associated with adverse events over the long term, but the risk of VF is extremely low if there are no other symptoms suggestive of a malignant etiology. 7.7.1 The overall evidence of ERP among general population in the literature In the general population, a total of six large population-based studies have been published, of which one is the initial study of this thesis. Two of these studies have concluded that the early repolarization pattern is benign (Klatsky et al. 2003b, Uberoi et al. 2011), whereas four have associated the pattern with adverse outcomes including cardiovascular and arrhythmic death (Haruta et al. 2011, Olson et al. 2011, Sinner et al. 2010, Tikkanen et al. 2009b). In 2003, Klatsky and colleagues found that individuals with early repolarization, defined as ST segment elevation ≥0.1mV, displayed no increased risk of death or hospitalization during a follow up of 2 years (Klatsky et al. 2003b). In this cohort, only 29% of patients with ST segment elevation had also J waves present. Recently, another group found no significant associations between early repolarization and cardiac mortality, again mainly focusing on ST segment elevation (Uberoi et al. 2011), in fact they observed decreased mortality rates associated with ST-elevations. In 2009, the first publication of this thesis indicated increased cardiac and arrhythmic mortality rates in middle-aged subjects with the early repolarization pattern, especially those with inferior and high amplitude J waves. These observations were soon confirmed in a German population of 1,945 subjects, in which the early repolarization pattern was associated with a two to four fold increased risk of cardiac mortality and inferior manifestation of ERP elevated the 97 risk even more extensively (Sinner et al. 2010). Thereafter Haruta with his colleagues stated that ERP was associated with a decreased risk of cardiac and allcause mortality, but a significantly elevated risk of unexpected sudden death in 5,976 atomic-bomb survivors in Japan (Haruta et al. 2011). The observed risk of unexplained death was again a doubling, given the fact that the prevalence was strikingly high (23.9%) when compared to previous publications. In another prospective population-based study on 15,141 subjects (Olson et al. 2011), a prevalence of 12.3% was observed for J waves, and these were associated with a doubled risk of sudden cardiac death in whites and in females, but not in blacks or males. Recently, a prevalence of 13% and nearly a nine-fold risk for cardiovascular death was observed in a French general population sample of 1,161 subjects (Rollin et al. 2012). The prognostic value of early repolarization pattern has also been evaluated among patients with chronic coronary artery disease and an implanted cardioverter-defibrillator. With a case-control setup, the prevalence of early repolarization pattern was significantly higher among patients who had appropriate ICD therapy for ventricular arrhythmias than in those without such events (32 vs 8%) (Patel et al. 2010). In another study, the J-point amplitude recorded in right ventricular intracardiac implantable cardioverter-defibrillator electrogram was significantly augmented immediately before the onset of polymorphic ventricular tachycardia or VF in patients with structural heart disease (Tereshchenko et al. 2012). The prognostic significance of ERP in the general population seems to fluctuate according to the definitions and methods being used. The most apparent and significant difference between these studies has been that those describing a benign nature have focused mainly on ST-segment elevation in contrast to the others with the higher mortality rates, which have placed more recent emphasis on the J waves regardless of the presence or absence of ST-segment elevation. Overall, the current evidence suggests that ERP is an independent predictor of adverse outcomes at the community level. In a recent meta-analysis of publications on ERP prognosis (Wu et al. 2012), the estimated absolute difference of subjects with J waves was 70 cases of arrhythmic death per 100,000 subjects per year, indicating that although the ECG variant is associated with arrhythmias, the absolute risk is very low. 98 7.7.2 Analogies with QT syndromes The inherited short and long QT syndromes are associated with a high risk of sudden cardiac death, and furthermore, the prolongation of the QT interval is a risk factor for SCD also in the population level. However, there is significant overlapping between the normal limits of the QT interval and the associated mutations. Furthermore, the mortality risk among the general population is moderate (Chugh et al. 2009, Straus et al. 2006, Tikkanen et al. 2009b). Similarly early repolarization syndrome (J waves in the verified absence of structural heart disease) is associated with a high risk of IVF and sudden death, and the early repolarization pattern (idiopathic occurrence of J waves in asymptomatic healthy individual with or without possible cardiac condition) have been linked with a moderate risk of sudden death in the general population. Thus, one might consider ERP to be analogous to the normal variation of the QT interval, whereas the so-called syndrome of early repolarization might be analogous to the situation in individuals affected by long or short QT syndromes. There are also several analogies in the risk factors of ventricular arrhythmias in LQTS and early repolarization: QT prolongation is comparable to augmentation of J waves, and fever to hypothermia. Additionally, in both LQTS and early repolarization, bradycardia and acute myocardial ischemia seem to increase the risk for arrhythmias. Similarly in early repolarization and QT syndromes, it is important to search for additional markers to identify those patients who are at a high risk of suffering adverse events, since all of these conditions are present in a wide range of patients with abnormal ECG patterns which are not all associated with a similar risk. 7.7.3 Association with Brugada syndrome The early repolarization syndrome and Brugada syndrome have been speculated to represent a continuous spectrum of diseases under the term J wave syndromes (Anzelevitch & Yan 2010). There are indeed several significant similarities between BrS and early repolarization pattern in IVF patients: the average age of the first event is under 40 years, a significant male predominance is present in both phenomena, there is a strong temporal variation in the expression of the patterns, and furthermore the pattern is normalized by quinidine exposure, catecholamine stimulation and faster heart rates. (Myerburg & Castellanos 2008) 99 In fact, inferolateral ERP (J waves) have been reported to be present in as high as 11-15% of Brugada patients, and furthermore their presence has been shown to have a strong adverse effect on prognosis (Kamakura et al. 2009, Letsas et al. 2008, Sarkozy et al. 2009). Recently, ERP in multiple leads followed by horizontal ST-segments were claimed to be highly arrhythmogenic substrates in BrS patients (Takagi et al. 2012). As discussed in the literature review, there is recent data pointing to the possibility of delayed depolarization in the right ventricular outflow tract as a mechanism contributing to J waves, arrhythmia expression, and possibly both phenomena in BrS (Nademanee et al. 2011, Wilde et al. 2010). Similarly to the situation in most diseases, it is very likely that Brugada syndrome and early repolarization are not fully attributable to one single mechanism, but possibly they are the outcomes of several various underlying pathophysiologies. 7.7.4 Early repolarization or delayed depolarization? As mentioned earlier, there has been an on-going debate on whether the pattern of early repolarization is truly due to accelerated repolarization or is it rather because there is delayed depolarization present (Antzelevitch et al. 2011, Borggrefe & Schimpf 2010, Littmann 2010, Littmann & Tenczer 2011, Surawicz & Macfarlane 2011, Wellens 2008). It is highly probable that individuals with ERP consist of a heterogeneous group of individuals with diverse clinical backgrounds and different electrophysiologic mechanisms. In IVF patients with marked J waves and without any signs of structural heart disease, accelerated repolarization is likely to be the mechanism; Steep repolarization gradients have been demonstrated with electrocardiographic imaging (Ghosh et al. 2009), suggesting that J waves are markers of an increased dispersion of repolarization. In contrast, in many elderly patients, the pattern might be caused by abnormal depolarization due to underlying subtle heart disease as postulated by Littman in his letter (Littmann 2010). The depolarization concept is supported by the findings from a large cohort in which ERP with the presence of Q waves or T wave inversions predicted cardiovascular death, but plain ERP was not associated with any increased risk (Uberoi et al. 2012). In fact, another study recently demonstrated that patients with inferior ER have a high prevalence of late potentials in signal averaged ECG (SAECG) and that some of the J waves seem to persist during 100 isoprenaline challenge, which is suggestive of an involvement of depolarization abnormalities in a subset of patients with early repolarization (Roten et al. 2012a). This finding was supported by the expected normalization of ERP in the lateral leads during beta-adrenergic (isoprenaline) stimulation (Roten et al. 2012a). Furthermore, Abe and colleagues demonstrated that the repolarization abnormality markers such as T-wave alternans or QT dispersion were not associated with J waves in IVF patients, but the incidence of late potentials, which are reflections of a depolarization abnormality, was higher in those with ERP than those without this pattern (Abe et al. 2010). Augmentation of J waves prior to the onset of ventricular fibrillation has been demonstrated in case reports and larger scale studies, indicative of a causal relationship between ERP and VF (Bernard et al. 2009, Nam et al. 2008, Shinohara et al. 2006b). Recently, another study presented data that the augmentation of J waves occurred immediately before spontaneous VF episodes, but not before the onset of monomorphic ventricular tachycardia in patients with ICDs (Tereshchenko et al. 2012). These observations strongly support the previous hypotheses that different pathophysiologies might be underlying the various regional J waves. Regional differences in ion channel characteristics are possible, but there is more likely to be involvement of depolarization defects. Bastiaenen and colleagues found that 60% of patients in whom ERP persisted during exercise displayed evidence of subtle structural heart disease (Bastiaenen et al. 2013). To further provoke discussion, Littmann and Tenczer have suggested even an accessory pathway conduction to possibly underlie the pattern (Littmann & Tenczer 2011), as recently two cases were presented with unmasking of ERP in pre-exication (Nagao et al. 2011). Based on these observations, the patient population with inferolateral ERP seem to be reflecting a broad range of various pathophysiologies. Hoogendijk and colleagues have discussed possible explanations between early repolarization pattern and VF in their recent editorial (Hoogendijk et al. 2012). Changes in the cardiac action potential, as presented in the literature review, have generally been believed to underlie the early repolarization pattern in both healthy individuals and in those with idiopathic ventricular fibrillation. However, this explanation is at odds with the fact that idiopathic ventricular fibrillation is a very rare condition, but early repolarization pattern and thus these abnormal currents can be observed in a significantly larger proportion of healthy individuals. It has also been speculated that the mechanism causing early repolarization pattern might be the same as that responsible for ventricular 101 fibrillation by providing a trigger or substrate for VF through the loss of the action potential dome and the initiation of re-entrant arrhythmias, as discussed in chapter 2.1.4 of this thesis. The J waves might serve as a way of identifying of those individuals with a deep notch in the action potential and who have at least a hypothetical risk of suffering re-entry arrhythmias. Furthermore, ERP in idiopathic VF patients may only mimic the early repolarization pattern in healthy individuals and reflect activation abnormalities. Hoogendijk and colleagues also proposed, that a deeper notch in the action potential (suggested to underlie also J waves) would indicate a shift in the balance towards more repolarizing current meaning that there would be less depolarizing current available in the adjacent cells, hence impairing conduction and theoretically resulting in increased susceptibility to unidirectional block and VF during transient arrhythmogenic conditions, such as ischemia (Hoogendijk et al. 2012). This theory would partly explain the association between ERP and arrhythmic mortality at the population level and is consistent with the findings of increased risk of SCD during ischemic events in those with ERP (study IV). Overall, all of these pieces of evidence demonstrate that the exact mechanisms underlying early repolarization pattern are far from clear and require further work. 7.7.5 Markers of higher risk of adverse events Similarly to other arrhythmia syndromes, syncope, aborted SCD and a family history of SCD are major markers of increased risk also in the case of early repolarization. Watanabe and colleagues reported younger age, family history of SCD and history of electrical storms as the most significant markers for arrhythmia recurrences in ERS patients (Watanabe et al. 2012). Overall, not all manifestations of ERP across the ECG leads are equally proarrhythmic, since the localization and ST segment morphology seems to play an important prognostic role. In asymptomatic healthy individuals, J-point elevation is most prominently seen in mid-precordial leads (V2 to V4), especially in young male athletes. Terminal QRS notching accompanied by ST-elevation in left-precordial (V4 to V6) and inferior (II, III and aVF) leads is likewise a common finding among young healthy individuals. In contrast, evidence is accumulating in support of the finding that terminal QRS changes in the absence of ST-elevation confer the highest risk of adverse events. As discussed, ERP with horizontal or descending ST segments has been shown to possess the highest risk of adverse 102 outcomes in IVF patient samples, as well as in general population samples (Bastiaenen et al. 2013, Rollin et al. 2012, Rosso et al. 2011, Tikkanen et al. 2011). Terminal QRS notching or slurring without ST-elevation are most often seen in the inferior leads, but are frequent also in left limb leads (I, aVL). They are seen less often in left-precordial leads, and practically never in midprecordials. Figure 15 demonstrates ERP in two young, healthy athletes (study III), and Figure 16 illustrates the typical ECGs of two middle-aged persons with J waves and different ST segment morphologies. A recent study, already discussed in the previous chapter, also demonstrated that there might be significant differences in the pathophysiological mechanism of inferior and lateral ERP manifestations, as lateral J waves invariably normalized during beta-adrenergic stimulation, but precordial and inferior J waves persisted in a subset of patients (Roten et al. 2012a). The amplitude of J waves also impacts on the prognostic evaluation of ERP. In case-control studies, the amplitude of the J waves has been significantly greater in patients with idiopathic ventricular fibrillation than controls (Abe et al. 2010, Haissaguerre et al. 2008, Nam et al. 2008, Rosso et al. 2008) and high amplitude J waves have been shown to possess prognostic significance among healthy individuals in the general population (studies I, III). The augmentation of J waves has also been demonstrated to predispose ventricular arrhythmias (Kitazawa et al. 2011, Nam et al. 2008, Tereshchenko et al. 2012). Furthermore, the dynamicity of ERP, observed as an augmentation after sudden R-R interval prolongation, has been demonstrated to be highly specific for idiopathic ventricular fibrillation (Aizawa et al. 2012). Therefore transient augmentation of J waves should be regarded as a sign of a risk of suffering fatal arrhythmias. Figure 14 describes the various early repolarization patterns and the associated risks in a simplified matter. An attempt has been made to find associations between common markers of ventricular arrhythmogenesis (ventricular late potentials in signal averaged ECG, depressed heart rate variability and presence of ventricular ectopy) and early repolarization pattern in patients referred to ambulatory 24-hour ECG recording (Holter) with negative results (Soliman et al. 2011). These results suggest that the arrhythmogenic potential in ERP is not likely to be mediated through pathological pathways, which could be detected by these markers, although the definition of ERP used in this study emphasized ST elevation (not the presence of J waves), and was based on an automated recognition of the pattern. Another recent study reported the Tpeak-Tend interval to be significantly increased and the Tp-Te/QT 103 ratio to be significantly increased in patients with the early repolarization pattern as compared to their age- and sex matched controls (Karim Talib et al. 2012). The Tpeak-Tend and Tp-Te/QT ratio are known markers of spatial dispersion of the repolarization underlying the development of high-risk ventricular arrhythmias in various cardiac conditions. Fig. 14. Early repolarization patterns and magnitude of sudden cardiac death risk. 104 Fig. 15. Inferior J waves followed by rapidly ascending ST segments in two young and healthy athletes. 105 Fig. 16. Four example ECGs of two middle-aged subjects carrying J waves with (A,C) and without (B,D) ST segment elevation in two distinct patient populations. 106 7.8 Screening for early repolarization and patient management Thus far there are no validated techniques to provoke ERP, or reliable methods recognition of the malignant patterns of early repolarization. The amplitude of J waves in infero-lateral leads is strongly associated with vagal tone and hypothermia; ventricular pauses and bradycardia tend to increase the amplitude of the J waves (Bastiaenen et al. 2010, Gross 2010, Kaneko et al. 2010, Wilhelm et al. 2010), and thus one could hypothesize that Holter monitoring could possibly unmask pathological J waves during bradycardia. Furthermore, in patients with early repolarization and ICD, the incidence of ventricular tachyarrhythmias has been shown to undergo significant circadian variation with nighttime peaks (Kim et al. 2012). Sodium-channel blockade, the method used to unmask the malign Brugada pattern, has recently been shown to attenuate infero-lateral J waves in patients with idiopathic VF conversely to the situation in patients with Brugada syndrome (Kawata et al. 2012, Roten et al. 2012b). A recent report suggested that the Valsalva maneuver might be a potential method to reveal the ECG pattern in family members of sufferers of early repolarization syndrome, but the prognostic value of this test is yet uncertain (Gourraud et al. 2013). Gourraud and colleagues demonstrated that the Valsalva maneuver revealed an early repolarization pattern in 28% of unaffected relatives, and increased the J wave amplitude in 85% of ERP affected relatives. One could argue that genetic testing may be a potential definite screening tool in family members of early repolarization syndrome, but it is likely that it will not be available in the very near future. All of the major automatic commercial programs for ECG analyzes are still using the traditional definition of early repolarization with its high take-off ST segment elevations. Those algorithms do not yet permit the recognition of ERP (J waves in infero-lateral leads), and therefore are of little or even no help for the clinician attempting to identify the malignant form of early repolarization. With the present knowledge, it is not possible to offer any detailed counselling of treatment of individuals with early repolarization ECG pattern. Overall, in asymptomatic subjects, screening for ERP is not legitimate, as the associated arrhythmia risk is extremely low. However, in patients with idiopathic VF or resuscitated cardiac arrest in whom there is no obvious etiology, the correct diagnosis of early repolarization has clinical importance, as patients with ERP are exposed to frequent shocks and electrical storms after the implantation of a cardioverter-defibrillator, and these events can be prevented by quinidine 107 (Haissaguerre et al. 2009c). Further longitudinal as well as observational studies on early repolarization patterns are required. Currently the only reasonable recommendation is that individuals with ERP should reduce their long-term risk for ischemic cardiac events by minimizing all modifiable risk factors for coronary artery disease, due to their possible higher risk of suffering arrhythmias during myocardial ischemia. 7.9 Remaining questions and future directions Many questions in ERP pathogenesis and magnitude of risk remain unanswered. The interest in early repolarization has grown tremendously in recent years resulting in a huge increase in the number of publications, and therefore it can be anticipated that novel data on this topic will emerge in the following years. The turnaround in the attitude from viewing early repolarization as a benign towards a malign phenomenon has raised the problem of confusing terminology (Perez et al. 2012, Surawicz & Macfarlane 2011), as already discussed in this thesis. Although the used terminology surrounding the terms J waves and early repolarization can be misleading in many ways, this recently described electrocardiographic pattern, referred to as early repolarization or J waves, has been shown to have the potential for arrhythmia prediction in several case-control and general population samples. The so-called J waves and early repolarization patterns, i.e. terminal QRS changes, appear to reflect a continuum of risk for arrhythmias. Regardless of the origin of these patterns, this phenomenon appears to have significant usefulness in risk prediction in a number of clinical circumstances. Whether ERP could or should be applied in prospective screening programs is another question. As previously discussed (Hlatky et al. 2009), the evaluation of novel risk markers, such as ERP, needs to be conducted in several phases including prospective validation, proof of incremental value when added to standard risk markers, and the assessment of outcomes after patient management. Understanding ERP and other electrophysiological phenomena, and their value in the mixture of old and novel risk markers, is still an ongoing challenge that certainly will continue in the years ahead. 108 8 Strengths and limitations of the thesis The most significant strength of all studies of the thesis was that all of the cohorts’ ECGs were analysed and interpreted one by one, and inter-observer as well as intra-observer estimations were performed. In none of the studies were any automatic digital analysers used, which has tended to be the standard methodology used in many of the other ECG investigations during recent years. Another major strength of studies I and III was the large population based sample size and the unique long follow-up period with a vast amount of data available. Furthermore, the classification of cardiovascular deaths into sudden arrhythmic deaths and other deaths by experienced cardiologists from the description of the death certificate was a unique feature of study population A. The second study had similar strengths: a large community based cohort and a uniform adjudication process between the two populations (B and C), as well as the inter-/intraobserver analyses. One major limitation of this thesis is the observational and retrospective nature of all of its studies, and therefore any conclusions drawn of the underlying mechanisms remain only speculative. The general population samples (study populations A, B and C), as well as the case-control populations (study population D), included only middle-aged and elderly subjects for the prognostic assessments, which can be considered as a methodological limitation. It is not appropriate to generalize the prognostic results directly to younger populations. Furthermore, all populations consisted of individuals of European ancestry, and therefore the findings may not be extrapolated to other races. The small sample size and the case-control nature of the cross-sectional fourth study are potential limitations, and therefore one cannot generalize the results drawn from these results (study IV). Prospective follow-up studies would give more reliable information in this respect. Furthermore, it should be noted that several demographic and clinical differences were present between the case and control groups due to the lower incidence of prior medical admissions in those suffering sudden cardiac death. One important potential limitation is the lack of a definitive way of measuring the J-point. The J-point elevation in the studies conducted in this thesis was measured either from the highest point of terminal QRS notching or slurring (studies I and III), or from the point of apparent QRS offset according to adjacent leads (studies II and IV). Additionally, defining the point of QRS offset is not 109 always straightforward, as the duration of the QRS complex varies within different areas of the myocardium. These can cause potential variations in the measurements of the J-point between publications, and therefore visual interpretation by a physician is always required. This is not an easy task, as the majority of general practitioners consider the interpretation of the standard ECG to be very challenging. A minor limitation of all the studies in this thesis is the small number of example ECGs in the publications, which probably could have been higher in order to illustrate the typical electrocardiographic features associated with adverse outcomes. The ECGs presented in the first study are subject to speculation of overlapping with peri-infarction block (Littmann 2010), which cannot definitely be ruled out, but was probably present in only a minority of the positive cases displaying the early repolarization pattern. Nevertheless, as discussed earlier in this thesis, several pathophysiological phenomena might be underlying the typical early repolarization pattern and the aetiology cannot be defined from a planar ECG. The focus of this thesis was to investigate early repolarization pattern in a standard ECG, regardless of its exact etiology. The understanding of the results of the publications in this thesis among general physicians around the globe has proved to be difficult. The first publication (study I) caused much confusion and alarm that what had been thought of as a common but completely benign electrocardiographic finding, could be life-threatening. The classical definition of ascending ST-segments with high take-off especially in the anterior leads became confused with the recent definition of emphasis on J waves in infero-lateral leads. Fortunately, the findings in the third study relieved some of the pressure on the clinical interpretation of the relevance of incidental early repolarization patterns. Regardless of the academic study designs in this thesis, as well as in other publications reporting significant and independent ECG risk factors in specific patient groups and general population, it is necessary to rationally evaluate the actual clinical impact of these observations. As discussed in a recent review, screening of these ECG abnormalities is somewhat questionable (Chou et al. 2011), as there is no data available on how ECG screening could affect clinical outcomes as compared to no screening. In addition to statistical multivariate models, there is usually no evidence for any value of adding ECG abnormalities to the conventional risk factors. Furthermore, it is not possible to draw any straightforward conclusions about whether an ECG abnormality should change 110 the status of a patient from a low-risk to a higher risk category. For example, a doubling in the risk of sudden death in a younger patient does not mean that the patient should be considered at high-risk when the absolute risk for adverse events (fatal arrhythmias) in this patient group is extremely low. 111 112 9 Conclusions and clinical implications The results of this thesis have added new valuable information on this potentially life threatening arrhythmogenic ECG variant. Until the results of this thesis and its accompanying publications, there was no data available on the prevalence and long-term prognosis of early repolarization ECG pattern (infero-lateral J waves). The results of this thesis suggest that the long considered benign early repolarization, i.e. ST-segment elevation in the absence of conduction defect or chest pain, should probably continue to be assessed as a benign phenomenon in young and healthy individuals with no other cause to suspect cardiac illness. More importantly, these results have demonstrated that ERP with horizontal or descending ST-segments, i.e. terminal QRS changes especially in the inferior leads, are associated with markedly increased cardiac mortality in the general population, and that individuals carrying this variant are at an increased risk of suffering fatal arrhythmia during myocardial ischemia. In conclusion, J waves and ST-segment elevation should be considered as independent ECG patterns carrying divergent prognostic significance instead of regarding them as a benign marker of good health. Although the early repolarization ECG pattern increases the relative risk of arrhythmic events at the population level, the absolute risk in an individual is low and therefore ERP generally should not be interpreted as a high-risk marker. Although clinical awareness is warranted, the majority of individuals carrying this ECG pattern have a benign prognosis. The only reasonable recommendation as a consequence of the results of this thesis is that all modifiable risk factors for coronary artery disease should be addressed in patients with ERP to reduce their long-term risk for fatal events. To date, there is no experimental evidence of any benefits of screening for ERP in asymptomatic subjects. 113 114 References Abe A, Ikeda T, Tsukada T, Ishiguro H, Miwa Y, Miyakoshi M, Mera H, Yusu S & Yoshino H (2010) Circadian variation of late potentials in idiopathic ventricular fibrillation associated with J waves: insights into alternative pathophysiology and risk stratification. Heart Rhythm 7(5): 675–682. Adabag AS, Casey SA, Kuskowski MA, Zenovich AG & Maron BJ (2005) Spectrum and prognostic significance of arrhythmias on ambulatory Holter electrocardiogram in hypertrophic cardiomyopathy. J Am Coll Cardiol 45(5): 697–704. Adabag AS, Peterson G, Apple FS, Titus J, King R & Luepker RV (2010) Etiology of sudden death in the community: results of anatomical, metabolic, and genetic evaluation. Am Heart J 159(1): 33–39. Aizawa Y, Sato A, Watanabe H, Chinushi M, Furushima H, Horie M, Kaneko Y, Imaizumi T, Okubo K, Watanabe I, Shinozaki T, Aizawa Y, Fukuda K, Joo K & Haissaguerre M (2012) Dynamicity of the J-wave in idiopathic ventricular fibrillation with a special reference to pause-dependent augmentation of the J-wave. J Am Coll Cardiol 59(22): 1948–1953. Algra A, Tijssen JG, Roelandt JR, Pool J & Lubsen J (1991) QTc prolongation measured by standard 12-lead electrocardiography is an independent risk factor for sudden death due to cardiac arrest. Circulation 83(6): 1888–1894. Anttonen O, Junttila MJ, Rissanen H, Reunanen A, Viitasalo M & Huikuri HV (2007) Prevalence and prognostic significance of short QT interval in a middle-aged Finnish population. Circulation 116(7): 714–720. Antzelevitch C (2012) Genetic, molecular and cellular mechanisms underlying the J wave syndromes. Circ J 76(5): 1054–1065. Antzelevitch C, Brugada P, Borggrefe M, Brugada J, Brugada R, Corrado D, Gussak I, LeMarec H, Nademanee K, Perez Riera AR, Shimizu W, Schulze-Bahr E, Tan H & Wilde A (2005a) Brugada syndrome: report of the second consensus conference. Heart Rhythm 2(4): 429–440. Antzelevitch C, Brugada P, Borggrefe M, Brugada J, Brugada R, Corrado D, Gussak I, LeMarec H, Nademanee K, Perez Riera AR, Shimizu W, Schulze-Bahr E, Tan H & Wilde A (2005b) Brugada syndrome: report of the second consensus conference: endorsed by the Heart Rhythm Society and the European Heart Rhythm Association. Circulation 111(5): 659–670. Antzelevitch C, Pollevick GD, Cordeiro JM, Casis O, Sanguinetti MC, Aizawa Y, Guerchicoff A, Pfeiffer R, Oliva A, Wollnik B, Gelber P, Bonaros EP,Jr, Burashnikov E, Wu Y, Sargent JD, Schickel S, Oberheiden R, Bhatia A, Hsu LF, Haissaguerre M, Schimpf R, Borggrefe M & Wolpert C (2007a) Loss-of-function mutations in the cardiac calcium channel underlie a new clinical entity characterized by ST-segment elevation, short QT intervals, and sudden cardiac death. Circulation 115(4): 442–449. 115 Antzelevitch C, Pollevick GD, Cordeiro JM, Casis O, Sanguinetti MC, Aizawa Y, Guerchicoff A, Pfeiffer R, Oliva A, Wollnik B, Gelber P, Bonaros EP,Jr, Burashnikov E, Wu Y, Sargent JD, Schickel S, Oberheiden R, Bhatia A, Hsu LF, Haissaguerre M, Schimpf R, Borggrefe M & Wolpert C (2007b) Loss-of-function mutations in the cardiac calcium channel underlie a new clinical entity characterized by ST-segment elevation, short QT intervals, and sudden cardiac death. Circulation 115(4): 442–449. Antzelevitch C, Sicouri S, Litovsky SH, Lukas A, Krishnan SC, Di Diego JM, Gintant GA & Liu DW (1991) Heterogeneity within the ventricular wall. Electrophysiology and pharmacology of epicardial, endocardial, and M cells. Circ Res 69(6): 1427–1449. Antzelevitch C & Yan GX (2010) J wave syndromes. Heart Rhythm 7(4):549-558. Antzelevitch C, Yan GX & Viskin S (2011) Rationale for the use of the terms J-wave syndromes and early repolarization. J Am Coll Cardiol 57(15): 1587–1590. Arking DE, Junttila MJ, Goyette P, Huertas-Vazquez A, Eijgelsheim M, Blom MT, Newton-Cheh C, Reinier K, Teodorescu C, Uy-Evanado A, Carter-Monroe N, Kaikkonen KS, Kortelainen ML, Boucher G, Lagace C, Moes A, Zhao X, Kolodgie F, Rivadeneira F, Hofman A, Witteman JC, Uitterlinden AG, Marsman RF, Pazoki R, Bardai A, Koster RW, Dehghan A, Hwang SJ, Bhatnagar P, Post W, Hilton G, Prineas RJ, Li M, Kottgen A, Ehret G, Boerwinkle E, Coresh J, Kao WH, Psaty BM, Tomaselli GF, Sotoodehnia N, Siscovick DS, Burke GL, Marban E, Spooner PM, Cupples LA, Jui J, Gunson K, Kesaniemi YA, Wilde AA, Tardif JC, O'Donnell CJ, Bezzina CR, Virmani R, Stricker BH, Tan HL, Albert CM, Chakravarti A, Rioux JD, Huikuri HV & Chugh SS (2011) Identification of a sudden cardiac death susceptibility locus at 2q24.2 through genome-wide association in European ancestry individuals. PLoS Genet 7(6): e1002158. Aro AL, Anttonen O, Tikkanen JT, Junttila MJ, Kerola T, Rissanen HA, Reunanen A & Huikuri HV (2011) Intraventricular conduction delay in a standard 12-lead electrocardiogram as a predictor of mortality in the general population. Circ Arrhythm Electrophysiol 4(5): 704–710. Aro AL, Anttonen O, Tikkanen JT, Junttila MJ, Kerola T, Rissanen HA, Reunanen A & Huikuri HV (2012) Prevalence and prognostic significance of T-wave inversions in right precordial leads of a 12-lead electrocardiogram in the middle-aged subjects. Circulation 125(21): 2572–2577. Arola A, Jokinen E, Ruuskanen O, Saraste M, Pesonen E, Kuusela AL, Tikanoja T, Paavilainen T & Simell O (1997) Epidemiology of idiopathic cardiomyopathies in children and adolescents. A nationwide study in Finland. Am J Epidemiol 146(5): 385–393. Aromaa A & Koskinen S (2004) Health and functional capacity in Finland. Baseline results of the Health 2000 Health Examination Survey. Publication number B12/2004. Helsinki: Publications of the National Public Health Institute. 116 Atarashi H, Ogawa S, Harumi K, Hayakawa H, Sugimoto T, Okada R, Murayama M & Toyama J (1996) Characteristics of patients with right bundle branch block and STsegment elevation in right precordial leads. Idiopathic Ventricular Fibrillation Investigators. Am J Cardiol 78(5): 581–583. Atarashi H, Ogawa S, Harumi K, Sugimoto T, Inoue H, Murayama M, Toyama J, Hayakawa H & Idiopathic Ventricular Fibrillation Investigators (2001) Three-year follow-up of patients with right bundle branch block and ST segment elevation in the right precordial leads: Japanese Registry of Brugada Syndrome. Idiopathic Ventricular Fibrillation Investigators. J Am Coll Cardiol 37(7): 1916–1920. Barajas-Martinez H, Hu D, Ferrer T, Onetti CG, Wu Y, Burashnikov E, Boyle M, Surman T, Urrutia J, Veltmann C, Schimpf R, Borggrefe M, Wolpert C, Ibrahim BB, SanchezChapula JA, Winters S, Haissaguerre M & Antzelevitch C (2012) Molecular genetic and functional association of Brugada and early repolarization syndromes with S422L missense mutation in KCNJ8. Heart Rhythm 9(4): 548–555. Bastiaenen R & Behr ER (2012) Benign or malignant, early or delayed: the changing face of early repolarization. Europace 14(1): 5–7. Bastiaenen R, Hedley PL, Christiansen M & Behr E (2010) Therapeutic hypothermia and VF storm in early repolarization syndrome. Heart Rhythm . Bastiaenen R, Raju H, Sharma S, Papadakis M, Chandra N, Muggenthaler M, Govindan M, Batchvarov VN & Behr ER (2013) Characterization of early repolarization during ajmaline provocation and exercise tolerance testing. Heart Rhythm 10(2): 247–254. Bayes de Luna A, Coumel P & Leclercq JF (1989) Ambulatory sudden cardiac death: mechanisms of production of fatal arrhythmia on the basis of data from 157 cases. Am Heart J 117(1): 151–159. Behr E, Wood DA, Wright M, Syrris P, Sheppard MN, Casey A, Davies MJ, McKenna W & Sudden Arrhythmic Death Syndrome Steering Group (2003) Cardiological assessment of first-degree relatives in sudden arrhythmic death syndrome. Lancet 362(9394): 1457–1459. Behr ER, Dalageorgou C, Christiansen M, Syrris P, Hughes S, Tome Esteban MT, Rowland E, Jeffery S & McKenna WJ (2008) Sudden arrhythmic death syndrome: familial evaluation identifies inheritable heart disease in the majority of families. Eur Heart J 29(13): 1670–1680. Belhassen B, Glick A & Viskin S (2004) Efficacy of quinidine in high-risk patients with Brugada syndrome. Circulation 110(13): 1731–1737. Belhassen B, Glick A & Viskin S (2009) Excellent long-term reproducibility of the electrophysiologic efficacy of quinidine in patients with idiopathic ventricular fibrillation and Brugada syndrome. Pacing Clin Electrophysiol 32(3): 294–301. Belhassen B & Viskin S (1993) Idiopathic ventricular tachycardia and fibrillation. J Cardiovasc Electrophysiol 4(3): 356–368. Bellocq C, van Ginneken AC, Bezzina CR, Alders M, Escande D, Mannens MM, Baro I & Wilde AA (2004) Mutation in the KCNQ1 gene leading to the short QT-interval syndrome. Circulation 109(20): 2394–2397. 117 Bernard A, Genee O, Grimard C, Sacher F, Fauchier L & Babuty D (2009) Electrical storm reversible by isoproterenol infusion in a striking case of early repolarization. J Interv Card Electrophysiol 25(2): 123–127. Bode-Schnurbus L, Bocker D, Block M, Gradaus R, Heinecke A, Breithardt G & Borggrefe M (2003) QRS duration: a simple marker for predicting cardiac mortality in ICD patients with heart failure. Heart 89(10): 1157–1162. Bongioanni S, Bianchi F, Migliardi A, Gnavi R, Pron PG, Casetta M & Conte MR (2007) Relation of QRS duration to mortality in a community-based cohort with hypertrophic cardiomyopathy. Am J Cardiol 100(3): 503–506. Borggrefe M & Schimpf R (2010) J-wave syndromes caused by repolarization or depolarization mechanisms a debated issue among experimental and clinical electrophysiologists. J Am Coll Cardiol 55(8): 798–800. Brenyo A, Pietrasik G, Barsheshet A, Huang DT, Polonsky B, McNitt S, Moss AJ & Zareba W (2012) QRS Fragmentation and the Risk of Sudden Cardiac Death in MADIT II. J Cardiovasc Electrophysiol . Brugada J, Brugada R, Antzelevitch C, Towbin J, Nademanee K & Brugada P (2002) Long-term follow-up of individuals with the electrocardiographic pattern of right bundle-branch block and ST-segment elevation in precordial leads V1 to V3. Circulation 105(1): 73–78. Brugada J, Brugada R & Brugada P (2003) Determinants of sudden cardiac death in individuals with the electrocardiographic pattern of Brugada syndrome and no previous cardiac arrest. Circulation 108(25): 3092–3096. Brugada P & Brugada J (1992) Right bundle branch block, persistent ST segment elevation and sudden cardiac death: a distinct clinical and electrocardiographic syndrome. A multicenter report. J Am Coll Cardiol 20(6): 1391–1396. Brugada R, Brugada J, Antzelevitch C, Kirsch GE, Potenza D, Towbin JA & Brugada P (2000) Sodium channel blockers identify risk for sudden death in patients with STsegment elevation and right bundle branch block but structurally normal hearts. Circulation 101(5): 510–515. Brugada R, Hong K, Dumaine R, Cordeiro J, Gaita F, Borggrefe M, Menendez TM, Brugada J, Pollevick GD, Wolpert C, Burashnikov E, Matsuo K, Wu YS, Guerchicoff A, Bianchi F, Giustetto C, Schimpf R, Brugada P & Antzelevitch C (2004) Sudden death associated with short-QT syndrome linked to mutations in HERG. Circulation 109(1): 30–35. Burashnikov E, Pfeiffer R, Barajas-Martinez H, Delpon E, Hu D, Desai M, Borggrefe M, Haissaguerre M, Kanter R, Pollevick GD, Guerchicoff A, Laino R, Marieb M, Nademanee K, Nam GB, Robles R, Schimpf R, Stapleton DD, Viskin S, Winters S, Wolpert C, Zimmern S, Veltmann C & Antzelevitch C (2010) Mutations in the cardiac L-type calcium channel associated with inherited J-wave syndromes and sudden cardiac death. Heart Rhythm 7(12): 1872–1882. 118 Byrne R, Constant O, Smyth Y, Callagy G, Nash P, Daly K & Crowley J (2008) Multiple source surveillance incidence and aetiology of out-of-hospital sudden cardiac death in a rural population in the West of Ireland. Eur Heart J 29(11): 1418–1423. Cappato R, Furlanello F, Giovinazzo V, Infusino T, Lupo P, Pittalis M, Foresti S, De Ambroggi G, Ali H, Bianco E, Riccamboni R, Butera G, Ricci C, Ranucci M, Pelliccia A & De Ambroggi L (2010) J Wave, QRS Slurring, and ST Elevation in Athletes With Cardiac Arrest in the Absence of Heart Disease: Marker of Risk or Innocent Bystander? Circ Arrhythm Electrophysiol 3(4): 305–311. Castle CH & Keane WM (1965) Electrocardiographic "Peri-Infarction Block". a Clinical and Pathologic Correlation. Circulation 31: 403–408. Chen Q, Kirsch GE, Zhang D, Brugada R, Brugada J, Brugada P, Potenza D, Moya A, Borggrefe M, Breithardt G, Ortiz-Lopez R, Wang Z, Antzelevitch C, O'Brien RE, Schulze-Bahr E, Keating MT, Towbin JA & Wang Q (1998) Genetic basis and molecular mechanism for idiopathic ventricular fibrillation. Nature 392(6673): 293– 296. Chou R, Arora B, Dana T, Fu R, Walker M & Humphrey L (2011) Screening asymptomatic adults with resting or exercise electrocardiography: a review of the evidence for the U.S. Preventive Services Task Force. Ann Intern Med 155(6): 375– 385. Chugh SS, Jui J, Gunson K, Stecker EC, John BT, Thompson B, Ilias N, Vickers C, Dogra V, Daya M, Kron J, Zheng ZJ, Mensah G & McAnulty J (2004) Current burden of sudden cardiac death: multiple source surveillance versus retrospective death certificate-based review in a large U.S. community. J Am Coll Cardiol 44(6): 1268– 1275. Chugh SS, Kelly KL & Titus JL (2000a) Sudden cardiac death with apparently normal heart. Circulation 102(6): 649–654. Chugh SS, Kelly KL & Titus JL (2000b) Sudden cardiac death with apparently normal heart. Circulation 102(6): 649–654. Chugh SS, Reinier K, Singh T, Uy-Evanado A, Socoteanu C, Peters D, Mariani R, Gunson K & Jui J (2009) Determinants of prolonged QT interval and their contribution to sudden death risk in coronary artery disease: the Oregon Sudden Unexpected Death Study. Circulation 119(5): 663–670. Cleland JG, Chattopadhyay S, Khand A, Houghton T & Kaye GC (2002) Prevalence and incidence of arrhythmias and sudden death in heart failure. Heart Fail Rev 7(3): 229– 242. Cordeiro JM, Marieb M, Pfeiffer R, Calloe K, Burashnikov E & Antzelevitch C (2009) Accelerated inactivation of the L-type calcium current due to a mutation in CACNB2b underlies Brugada syndrome. J Mol Cell Cardiol 46(5): 695–703. Corrado D, Basso C, Buja G, Nava A, Rossi L & Thiene G (2001) Right bundle branch block, right precordial st-segment elevation, and sudden death in young people. Circulation 103(5): 710–717. Corrado D, Basso C & Thiene G (2000) Arrhythmogenic right ventricular cardiomyopathy: diagnosis, prognosis, and treatment. Heart 83(5): 588–595. 119 Corrado D, Basso C, Thiene G, McKenna WJ, Davies MJ, Fontaliran F, Nava A, Silvestri F, Blomstrom-Lundqvist C, Wlodarska EK, Fontaine G & Camerini F (1997) Spectrum of clinicopathologic manifestations of arrhythmogenic right ventricular cardiomyopathy/dysplasia: a multicenter study. J Am Coll Cardiol 30(6): 1512–1520. Corrado D, Nava A, Buja G, Martini B, Fasoli G, Oselladore L, Turrini P & Thiene G (1996) Familial cardiomyopathy underlies syndrome of right bundle branch block, ST segment elevation and sudden death. J Am Coll Cardiol 27(2): 443–448. Corrado D, Pelliccia A, Heidbuchel H, Sharma S, Link M, Basso C, Biffi A, Buja G, Delise P, Gussac I, Anastasakis A, Borjesson M, Bjornstad HH, Carre F, Deligiannis A, Dugmore D, Fagard R, Hoogsteen J, Mellwig KP, Panhuyzen-Goedkoop N, Solberg E, Vanhees L, Drezner J, Estes NA,3rd, Iliceto S, Maron BJ, Peidro R, Schwartz PJ, Stein R, Thiene G, Zeppilli P, McKenna WJ & Section of Sports Cardiology, European Association of Cardiovascular Prevention and Rehabilitation (2010) Recommendations for interpretation of 12-lead electrocardiogram in the athlete. Eur Heart J 31(2): 243–259. Cox MG, van der Smagt JJ, Wilde AA, Wiesfeld AC, Atsma DE, Nelen MR, Rodriguez LM, Loh P, Cramer MJ, Doevendans PA, van Tintelen JP, de Bakker JM & Hauer RN (2009) New ECG criteria in arrhythmogenic right ventricular dysplasia/cardiomyopathy. Circ Arrhythm Electrophysiol 2(5): 524–530. Dalal D, Nasir K, Bomma C, Prakasa K, Tandri H, Piccini J, Roguin A, Tichnell C, James C, Russell SD, Judge DP, Abraham T, Spevak PJ, Bluemke DA & Calkins H (2005) Arrhythmogenic right ventricular dysplasia: a United States experience. Circulation 112(25): 3823–3832. Das MK, Khan B, Jacob S, Kumar A & Mahenthiran J (2006) Significance of a fragmented QRS complex versus a Q wave in patients with coronary artery disease. Circulation 113(21): 2495–2501. Das MK, Michael MA, Suradi H, Peng J, Sinha A, Shen C, Mahenthiran J & Kovacs RJ (2009) Usefulness of fragmented QRS on a 12-lead electrocardiogram in acute coronary syndrome for predicting mortality. Am J Cardiol 104(12): 1631–1637. Das MK, Saha C, El Masry H, Peng J, Dandamudi G, Mahenthiran J, McHenry P & Zipes DP (2007) Fragmented QRS on a 12-lead ECG: a predictor of mortality and cardiac events in patients with coronary artery disease. Heart Rhythm 4(11): 1385–1392. Das MK, Suradi H, Maskoun W, Michael MA, Shen C, Peng J, Dandamudi G & Mahenthiran J (2008) Fragmented wide QRS on a 12-lead ECG: a sign of myocardial scar and poor prognosis. Circ Arrhythm Electrophysiol 1(4): 258–268. de Vreede-Swagemakers JJ, Gorgels AP, Dubois-Arbouw WI, van Ree JW, Daemen MJ, Houben LG & Wellens HJ (1997) Out-of-hospital cardiac arrest in the 1990's: a population-based study in the Maastricht area on incidence, characteristics and survival. J Am Coll Cardiol 30(6): 1500–1505. Dec GW & Fuster V (1994) Idiopathic dilated cardiomyopathy. N Engl J Med 331(23): 1564–1575. 120 Dekker LR, Bezzina CR, Henriques JP, Tanck MW, Koch KT, Alings MW, Arnold AE, de Boer MJ, Gorgels AP, Michels HR, Verkerk A, Verheugt FW, Zijlstra F & Wilde AA (2006) Familial sudden death is an important risk factor for primary ventricular fibrillation: a case-control study in acute myocardial infarction patients. Circulation 114(11): 1140–1145. Delaney JT, Muhammad R, Blair MA, Kor K, Fish FA, Roden DM & Darbar D (2012) A KCNJ8 mutation associated with early repolarization and atrial fibrillation. Europace . Derval N, Simpson CS, Birnie DH, Healey JS, Chauhan V, Champagne J, Gardner M, Sanatani S, Yee R, Skanes AC, Gula LJ, Leong-Sit P, Ahmad K, Gollob MH, Haissaguerre M, Klein GJ & Krahn AD (2011) Prevalence and characteristics of early repolarization in the CASPER registry: cardiac arrest survivors with preserved ejection fraction registry. J Am Coll Cardiol 58(7): 722–728. Desai AD, Yaw TS, Yamazaki T, Kaykha A, Chun S & Froelicher VF (2006) Prognostic Significance of Quantitative QRS Duration. Am J Med 119(7): 600–606. Drory Y, Turetz Y, Hiss Y, Lev B, Fisman EZ, Pines A & Kramer MR (1991) Sudden unexpected death in persons less than 40 years of age. Am J Cardiol 68(13): 1388– 1392. Eckardt L, Probst V, Smits JP, Bahr ES, Wolpert C, Schimpf R, Wichter T, Boisseau P, Heinecke A, Breithardt G, Borggrefe M, LeMarec H, Bocker D & Wilde AA (2005) Long-term prognosis of individuals with right precordial ST-segment-elevation Brugada syndrome. Circulation 111(3): 257–263. Efremidis M, Letsas KP, Weber R, Gavrielatos G, Filippatos GS, Sideris A & Kardaras F (2009) Recurrent syncope associated with a distinct ECG pattern consisting of short QT interval, early repolarization and atrioventricular block. Clin Res Cardiol 98(12): 807–810. Elhendy A, Hammill SC, Mahoney DW & Pellikka PA (2005) Relation of QRS duration on the surface 12-lead electrocardiogram with mortality in patients with known or suspected coronary artery disease. Am J Cardiol 96(8): 1082–1088. Elliott P, Andersson B, Arbustini E, Bilinska Z, Cecchi F, Charron P, Dubourg O, Kuhl U, Maisch B, McKenna WJ, Monserrat L, Pankuweit S, Rapezzi C, Seferovic P, Tavazzi L & Keren A (2008a) Classification of the cardiomyopathies: a position statement from the European Society Of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J 29(2): 270–276. Elliott P, Andersson B, Arbustini E, Bilinska Z, Cecchi F, Charron P, Dubourg O, Kuhl U, Maisch B, McKenna WJ, Monserrat L, Pankuweit S, Rapezzi C, Seferovic P, Tavazzi L & Keren A (2008b) Classification of the cardiomyopathies: a position statement from the European Society Of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J 29(2): 270–276. Elliott PM, Gimeno JR, Thaman R, Shah J, Ward D, Dickie S, Tome Esteban MT & McKenna WJ (2006) Historical trends in reported survival rates in patients with hypertrophic cardiomyopathy. Heart 92(6): 785–791. Eriksson P, Hansson PO, Eriksson H & Dellborg M (1998) Bundle-branch block in a general male population: the study of men born 1913. Circulation 98(22): 2494–2500. 121 Eriksson P, Wilhelmsen L & Rosengren A (2005) Bundle-branch block in middle-aged men: risk of complications and death over 28 years. The Primary Prevention Study in Goteborg, Sweden. Eur Heart J 26(21): 2300–2306. European Heart Rhythm Association, Heart Rhythm Society, Zipes DP, Camm AJ, Borggrefe M, Buxton AE, Chaitman B, Fromer M, Gregoratos G, Klein G, Moss AJ, Myerburg RJ, Priori SG, Quinones MA, Roden DM, Silka MJ, Tracy C, Smith SC,Jr, Jacobs AK, Adams CD, Antman EM, Anderson JL, Hunt SA, Halperin JL, Nishimura R, Ornato JP, Page RL, Riegel B, Priori SG, Blanc JJ, Budaj A, Camm AJ, Dean V, Deckers JW, Despres C, Dickstein K, Lekakis J, McGregor K, Metra M, Morais J, Osterspey A, Tamargo JL, Zamorano JL, American College of Cardiology, American Heart Association Task Force & European Society of Cardiology Committee for Practice Guidelines (2006) ACC/AHA/ESC 2006 guidelines for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: a report of the American College of Cardiology/American Heart Association Task Force and the European Society of Cardiology Committee for Practice Guidelines (Writing Committee to Develop Guidelines for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death). J Am Coll Cardiol 48(5): e247–346. Fahy GJ, Pinski SL, Miller DP, McCabe N, Pye C, Walsh MJ & Robinson K (1996) Natural history of isolated bundle branch block. Am J Cardiol 77(14): 1185–1190. Fananapazir L, Tracy CM, Leon MB, Winkler JB, Cannon RO,3rd, Bonow RO, Maron BJ & Epstein SE (1989) Electrophysiologic abnormalities in patients with hypertrophic cardiomyopathy. A consecutive analysis in 155 patients. Circulation 80(5): 1259– 1268. Fenichel NN (1962) A long term study of concave RS-T elevation--a normal variant of the electrocardiogram. Angiology 13: 360–366. Fenichel NN (1962) A long term study of concave RS-T elevation--a normal variant of the electrocardiogram. Angiology 13: 360–366. First SR, Bayley RH & Bedford DR (1950) Peri-infarction block; electrocardiographic abnormality occasionally resembling bundle branch block and local ventricular block of other types. Circulation 2(1): 31–36. Fish JM & Antzelevitch C (2004) Link between hypothermia and the Brugada syndrome. J Cardiovasc Electrophysiol 15(8): 942–944. Fleg J, Das D & Lakatta E (1983) Right bundle branch block: long-term prognosis in apparently healthy men. J Am Coll Cardiol 1(3): 887–892. Flowers NC, Horan LG, Thomas JR & Tolleson WJ (1969) The anatomic basis for highfrequency components in the electrocardiogram. Circulation 39(4): 531–539. Flowers NC, Horan LG, Wylds AC, Crawford W, Sridharan MR, Horan CP & Cliff SF (1990) Relation of peri-infarction block to ventricular late potentials in patients with inferior wall myocardial infarction. Am J Cardiol 66(5): 568–574. Fontaine G, Fontaliran F, Hebert JL, Chemla D, Zenati O, Lecarpentier Y & Frank R (1999) Arrhythmogenic right ventricular dysplasia. Annu Rev Med 50: 17–35. 122 Funada A, Hayashi K, Ino H, Fujino N, Uchiyama K, Sakata K, Masuta E, Sakamoto Y, Tsubokawa T & Yamagishi M (2008) Assessment of QT intervals and prevalence of short QT syndrome in Japan. Clin Cardiol 31(6): 270–274. Fuster V, Gersh BJ, Giuliani ER, Tajik AJ, Brandenburg RO & Frye RL (1981) The natural history of idiopathic dilated cardiomyopathy. Am J Cardiol 47(3): 525–531. Gaita F, Giustetto C, Bianchi F, Wolpert C, Schimpf R, Riccardi R, Grossi S, Richiardi E & Borggrefe M (2003a) Short QT Syndrome: a familial cause of sudden death. Circulation 108(8): 965–970. Gaita F, Giustetto C, Bianchi F, Wolpert C, Schimpf R, Riccardi R, Grossi S, Richiardi E & Borggrefe M (2003b) Short QT Syndrome: a familial cause of sudden death. Circulation 108(8): 965–970. Gallagher MM, Forleo GB, Behr ER, Magliano G, De Luca L, Morgia V, De Liberato F & Romeo F (2008) Prevalence and significance of Brugada-type ECG in 12,012 apparently healthy European subjects. Int J Cardiol 130(1): 44–48. Gallagher MM, Magliano G, Yap YG, Padula M, Morgia V, Postorino C, Di Liberato F, Leo R, Borzi M & Romeo F (2006) Distribution and prognostic significance of QT intervals in the lowest half centile in 12,012 apparently healthy persons. Am J Cardiol 98(7): 933–935. Ghosh S, Cooper DH, Vijayakumar R, Zhang J, Pollak S, Haissaguerre M & Rudy Y (2009) Early repolarization associated with sudden death: Insights from noninvasive electrocardiographic imaging. Heart Rhythm . Gibbons LW, Cooper KH, Martin RP & Pollock ML (1977) Medical examination and electrocardiographic analysis of elite distance runners. Ann N Y Acad Sci 301: 283– 296. Giustetto C, Drago S, Demarchi PG, Dalmasso P, Bianchi F, Masi AS, Carvalho P, Occhetta E, Rossetti G, Riccardi R, Bertona R, Gaita F & Italian Association of Arrhythmology and Cardiostimulation (AIAC)-Piedmont Section (2009) Risk stratification of the patients with Brugada type electrocardiogram: a community-based prospective study. Europace 11(4): 507–513. Goldenberg I, Horr S, Moss AJ, Lopes CM, Barsheshet A, McNitt S, Zareba W, Andrews ML, Robinson JL, Locati EH, Ackerman MJ, Benhorin J, Kaufman ES, Napolitano C, Platonov PG, Priori SG, Qi M, Schwartz PJ, Shimizu W, Towbin JA, Vincent GM, Wilde AA & Zhang L (2011) Risk for life-threatening cardiac events in patients with genotype-confirmed long-QT syndrome and normal-range corrected QT intervals. J Am Coll Cardiol 57(1): 51–59. Goldman MJ (1953a) RS-T segment elevation in mid- and left precordial leads as a normal variant. Am Heart J 46(6): 817–820. Goldman MJ (1953b) RS-T segment elevation in mid- and left precordial leads as a normal variant. Am Heart J 46(6): 817–820. Goldman MJ (1960) Normal variants in the electrocardiogram leading to cardiac invalidism. Am Heart J 59: 71–77. Gollob MH, Redpath CJ & Roberts JD (2011) The short QT syndrome: proposed diagnostic criteria. J Am Coll Cardiol 57(7): 802–812. 123 Gourraud JB, Le Scouarnec S, Sacher F, Chatel S, Derval N, Portero V, Chavernac P, Sandoval JE, Mabo P, Redon R, Schott JJ, Le Marec H, Haissaguerre M & Probst V (2013) Identification of large families in early repolarization syndrome. J Am Coll Cardiol 61(2): 164–172. Grant RP, Eestes EH,Jr & Doyle JT (1951) Spatial vector electrocardiography; the clinical characteristics of S-T and T vectors. Circulation 3(2): 182–197. Greene HL, Richardson DW, Barker AH, Roden DM, Capone RJ, Echt DS, Friedman LM, Gillespie MJ, Hallstrom AP & Verter J (1989) Classification of deaths after myocardial infarction as arrhythmic or nonarrhythmic (the Cardiac Arrhythmia Pilot Study). Am J Cardiol 63(1): 1–6. Gross GJ (2010) Early repolarization and ventricular fibrillation: Vagally familiar? Heart Rhythm . Grusin H (1954) Peculiarities of the African's electrocardiogram and the changes observed in serial studies. Circulation 9(6): 860–867. Gussak I & Antzelevitch C (2000) Early repolarization syndrome: clinical characteristics and possible cellular and ionic mechanisms. J Electrocardiol 33(4): 299–309. Gussak I & Bjerregaard P (2005) Short QT syndrome--5 years of progress. J Electrocardiol 38(4): 375–377. Gussak I, Brugada P, Brugada J, Wright RS, Kopecky SL, Chaitman BR & Bjerregaard P (2000) Idiopathic short QT interval: a new clinical syndrome? Cardiology 94(2): 99– 102. Haissaguerre M, Chatel S, Sacher F, Weerasooriya R, Probst V, Loussouarn G, Horlitz M, Liersch R, Schulze-Bahr E, Wilde A, Kaab S, Koster J, Rudy Y, Le Marec H & Schott JJ (2009a) Ventricular fibrillation with prominent early repolarization associated with a rare variant of KCNJ8/KATP channel. J Cardiovasc Electrophysiol 20(1): 93–98. Haissaguerre M, Derval N, Sacher F, Jesel L, Deisenhofer I, de Roy L, Pasquie JL, Nogami A, Babuty D, Yli-Mayry S, De Chillou C, Scanu P, Mabo P, Matsuo S, Probst V, Le Scouarnec S, Defaye P, Schlaepfer J, Rostock T, Lacroix D, Lamaison D, Lavergne T, Aizawa Y, Englund A, Anselme F, O'Neill M, Hocini M, Lim KT, Knecht S, Veenhuyzen GD, Bordachar P, Chauvin M, Jais P, Coureau G, Chene G, Klein GJ & Clementy J (2008) Sudden cardiac arrest associated with early repolarization. N Engl J Med 358(19): 2016–2023. Haissaguerre M, Sacher F, Nogami A, Komiya N, Bernard A, Probst V, Yli-Mayry S, Defaye P, Aizawa Y, Frank R, Mantovan R, Cappato R, Wolpert C, Leenhardt A, de Roy L, Heidbuchel H, Deisenhofer I, Arentz T, Pasquie JL, Weerasooriya R, Hocini M, Jais P, Derval N, Bordachar P & Clementy J (2009b) Characteristics of recurrent ventricular fibrillation associated with inferolateral early repolarization role of drug therapy. J Am Coll Cardiol 53(7): 612–619. 124 Haissaguerre M, Sacher F, Nogami A, Komiya N, Bernard A, Probst V, Yli-Mayry S, Defaye P, Aizawa Y, Frank R, Mantovan R, Cappato R, Wolpert C, Leenhardt A, de Roy L, Heidbuchel H, Deisenhofer I, Arentz T, Pasquie JL, Weerasooriya R, Hocini M, Jais P, Derval N, Bordachar P & Clementy J (2009c) Characteristics of recurrent ventricular fibrillation associated with inferolateral early repolarization role of drug therapy. J Am Coll Cardiol 53(7): 612–619. Hanson B, Tuna N, Bouchard T, Heston L, Eckert E, Lykken D, Segal N & Rich S (1989) Genetic factors in the electrocardiogram and heart rate of twins reared apart and together. Am J Cardiol 63(9): 606–609. Haruta D, Matsuo K, Tsuneto A, Ichimaru S, Hida A, Sera N, Imaizumi M, Nakashima E, Maemura K & Akahoshi M (2011) Incidence and prognostic value of early repolarization pattern in the 12-lead electrocardiogram. Circulation 123(25): 2931– 2937. Hedley PL, Jorgensen P, Schlamowitz S, Wangari R, Moolman-Smook J, Brink PA, Kanters JK, Corfield VA & Christiansen M (2009) The genetic basis of long QT and short QT syndromes: a mutation update. Hum Mutat 30(11): 1486–1511. Heistaro S (2008) Methodology report, Health 2000 survey. KTL national Public Health Institute, Finland. Heng SJ, Clark EN & Macfarlane PW (2012) End QRS Notching or Slurring in the Electrocardiogram: Influence on the Definition of "Early Repolarization". J Am Coll Cardiol . Hermida JS, Denjoy I, Clerc J, Extramiana F, Jarry G, Milliez P, Guicheney P, Di Fusco S, Rey JL, Cauchemez B & Leenhardt A (2004) Hydroquinidine therapy in Brugada syndrome. J Am Coll Cardiol 43(10): 1853–1860. Hermida JS, Lemoine JL, Aoun FB, Jarry G, Rey JL & Quiret JC (2000) Prevalence of the brugada syndrome in an apparently healthy population. Am J Cardiol 86(1): 91–94. Hisamatsu K, Kusano KF, Morita H, Takenaka S, Nagase S, Nakamura K, Emori T, Matsubara H, Mikouchi H, Nishizaki Y & Ohe T (2004) Relationships between depolarization abnormality and repolarization abnormality in patients with Brugada syndrome: using body surface signal-averaged electrocardiography and body surface maps. J Cardiovasc Electrophysiol 15(8): 870–876. Hlaing T, DiMino T, Kowey PR & Yan GX (2005) ECG repolarization waves: their genesis and clinical implications. Ann Noninvasive Electrocardiol 10(2): 211–223. Hlatky MA, Greenland P, Arnett DK, Ballantyne CM, Criqui MH, Elkind MS, Go AS, Harrell FE,Jr, Hong Y, Howard BV, Howard VJ, Hsue PY, Kramer CM, McConnell JP, Normand SL, O'Donnell CJ, Smith SC,Jr, Wilson PW & American Heart Association Expert Panel on Subclinical Atherosclerotic Diseases and Emerging Risk Factors and the Stroke Council (2009) Criteria for evaluation of novel markers of cardiovascular risk: a scientific statement from the American Heart Association. Circulation 119(17): 2408–2416. Homsi M, Alsayed L, Safadi B, Mahenthiran J & Das MK (2009) Fragmented QRS complexes on 12-lead ECG: a marker of cardiac sarcoidosis as detected by gadolinium cardiac magnetic resonance imaging. Ann Noninvasive Electrocardiol 14(4): 319–326. 125 Hoogendijk MG, Opthof T, Postema PG, Wilde AA, de Bakker JM & Coronel R (2010) The Brugada ECG pattern: a marker of channelopathy, structural heart disease, or neither? Toward a unifying mechanism of the Brugada syndrome. Circ Arrhythm Electrophysiol 3(3): 283–290. Hoogendijk MG, Potse M & Coronel R (2012) Early repolarization patterns: the good, the bad, and the ugly? Heart Rhythm 9(2): 230–231. Hookana E, Junttila MJ, Puurunen VP, Tikkanen JT, Kaikkonen KS, Kortelainen ML, Myerburg RJ & Huikuri HV (2011a) Causes of nonischemic sudden cardiac death in the current era. Heart Rhythm 8(10): 1570–1575. Hookana E, Junttila MJ, Puurunen VP, Tikkanen JT, Kaikkonen KS, Kortelainen ML, Myerburg RJ & Huikuri HV (2011b) Causes of nonischemic sudden cardiac death in the current era. Heart Rhythm 8(10): 1570–1575. Huikuri HV, Castellanos A & Myerburg RJ (2001) Sudden death due to cardiac arrhythmias. N Engl J Med 345(20): 1473–1482. Ikeda T, Sakurada H, Sakabe K, Sakata T, Takami M, Tezuka N, Nakae T, Noro M, Enjoji Y, Tejima T, Sugi K & Yamaguchi T (2001) Assessment of noninvasive markers in identifying patients at risk in the Brugada syndrome: insight into risk stratification. J Am Coll Cardiol 37(6): 1628–1634. Iuliano S, Fisher SG, Karasik PE, Fletcher RD, Singh SN & Department of Veterans Affairs Survival Trial of Antiarrhythmic Therapy in Congestive Heart Failure (2002) QRS duration and mortality in patients with congestive heart failure. Am Heart J 143(6): 1085–1091. Jain R, Dalal D, Daly A, Tichnell C, James C, Evenson A, Jain R, Abraham T, Tan BY, Tandri H, Russell SD, Judge D & Calkins H (2009) Electrocardiographic features of arrhythmogenic right ventricular dysplasia. Circulation 120(6): 477–487. John BT, Tamarappoo BK, Titus JL, Edwards WD, Shen WK & Chugh SS (2004) Global remodeling of the ventricular interstitium in idiopathic myocardial fibrosis and sudden cardiac death. Heart Rhythm 1(2): 141–149. Johnson RA & Palacios I (1982a) Dilated cardiomyopathies of the adult (first of two parts). N Engl J Med 307(17): 1051–1058. Johnson RA & Palacios I (1982b) Dilated cardiomyopathies of the adult (second of two parts). N Engl J Med 307(18): 1119–1126. Josephson M & Wellens HJ (2004) Implantable defibrillators and sudden cardiac death. Circulation 109(22): 2685–2691. Jouven X, Desnos M, Guerot C & Ducimetiere P (1999) Predicting sudden death in the population: the Paris Prospective Study I. Circulation 99(15): 1978–1983. Junttila MJ, Brugada P, Hong K, Lizotte E, DE Zutter M, Sarkozy A, Brugada J, Benito B, Perkiomaki JS, Makikallio TH, Huikuri HV & Brugada R (2008a) Differences in 12lead electrocardiogram between symptomatic and asymptomatic Brugada syndrome patients. J Cardiovasc Electrophysiol 19(4): 380–383. 126 Junttila MJ, Gonzalez M, Lizotte E, Benito B, Vernooy K, Sarkozy A, Huikuri HV, Brugada P, Brugada J & Brugada R (2008b) Induced Brugada-type electrocardiogram, a sign for imminent malignant arrhythmias. Circulation 117(14): 1890–1893. Junttila MJ, Raatikainen MJ, Karjalainen J, Kauma H, Kesaniemi YA & Huikuri HV (2004) Prevalence and prognosis of subjects with Brugada-type ECG pattern in a young and middle-aged Finnish population. Eur Heart J 25(10): 874–878. Junttila MJ, Sager SJ, Freiser M, McGonagle S, Castellanos A & Myerburg RJ (2010) Inferolateral early repolarization in athletes. J Interv Card Electrophysiol . Kaikkonen KS, Kortelainen ML, Linna E & Huikuri HV (2006) Family history and the risk of sudden cardiac death as a manifestation of an acute coronary event. Circulation 114(14): 1462–1467. Kalla H, Yan GX & Marinchak R (2000) Ventricular fibrillation in a patient with prominent J (Osborn) waves and ST segment elevation in the inferior electrocardiographic leads: a Brugada syndrome variant? J Cardiovasc Electrophysiol 11(1): 95–98. Kamakura S, Ohe T, Nakazawa K, Aizawa Y, Shimizu A, Horie M, Ogawa S, Okumura K, Tsuchihashi K, Sugi K, Makita N, Hagiwara N, Inoue H, Atarashi H, Aihara N, Shimizu W, Kurita T, Suyama K, Noda T, Satomi K, Okamura H, Tomoike H & Brugada Syndrome Investigators in Japan (2009) Long-term prognosis of probands with Brugada-pattern ST-elevation in leads V1-V3. Circ Arrhythm Electrophysiol 2(5): 495–503. Kambara H & Phillips J (1976a) Long-term evaluation of early repolarization syndrome (normal variant RS-T segment elevation). Am J Cardiol 38(2): 157–156. Kambara H & Phillips J (1976b) Long-term evaluation of early repolarization syndrome (normal variant RS-T segment elevation). Am J Cardiol 38(2): 157–156. Kaneko Y, Nakajima T, Irie T & Kurabayashi M (2010) Augmentation of J wave following ventricular pause in "benign" early repolarization. Intern Med 49(15): 1671. Karim Talib A, Sato N, Sakamoto N, Tanabe Y, Takeuchi T, Saijo Y, Kawamura Y & Hasebe N (2012) Enhanced Transmural Dispersion of Repolarization in Patients with J Wave Syndromes. J Cardiovasc Electrophysiol . Kashani A & Barold SS (2005) Significance of QRS complex duration in patients with heart failure. J Am Coll Cardiol 46(12): 2183–2192. Kawata H, Noda T, Yamada Y, Okamura H, Satomi K, Aiba T, Takaki H, Aihara N, Isobe M, Kamakura S & Shimizu W (2012) Effect of sodium-channel blockade on early repolarization in inferior/lateral leads in patients with idiopathic ventricular fibrillation and Brugada syndrome. Heart Rhythm 9(1): 77–83. Kim SH, Nam GB, Baek S, Oh Choi H, Hun Kim K, Choi KJ, Joung B, Pak HN, Lee MH, Soon Kim S, Park SJ, Keun On Y, Soo Kim J, Oh IY, Choi EK, Oh S, Choi YS, Il Choi J, Weon Park S, Kim YH, Young Lee M, Euy Lim H, Lee YS, Cho Y, Kim J, Lee DI, Kyoung Cho D & Kim YH (2012) Circadian and Seasonal Variations of Ventricular Tachyarrhythmias in Patients with Early Repolarization Syndrome and Brugada Syndrome: Analysis of Patients with Implantable Cardioverter Defibrillator. J Cardiovasc Electrophysiol 23(7): 757–763. 127 Kitazawa H, Wakasugi T, Sugimoto T, Yamamoto K, Yoshii S & Aizawa Y (2011) Evolving J waves prior to ventricular fibrillation postoperative coronary bypass. Intern Med 50(20): 2337–2340. Klatsky AL, Oehm R, Cooper RA, Udaltsova N & Armstrong MA (2003a) The early repolarization normal variant electrocardiogram: correlates and consequences. Am J Med 115(3): 171–177. Klatsky AL, Oehm R, Cooper RA, Udaltsova N & Armstrong MA (2003b) The early repolarization normal variant electrocardiogram: correlates and consequences. Am J Med 115(3): 171–177. Kobza R, Roos M, Niggli B, Abacherli R, Lupi GA, Frey F, Schmid JJ & Erne P (2009) Prevalence of long and short QT in a young population of 41,767 predominantly male Swiss conscripts. Heart Rhythm 6(5): 652–657. Komiya N, Imanishi R, Kawano H, Shibata R, Moriya M, Fukae S, Doi Y, Nakao K, Seto S & Yano K (2006) Ventricular fibrillation in a patient with prominent j wave in the inferior and lateral electrocardiographic leads after gastrostomy. Pacing Clin Electrophysiol 29(9): 1022–1024. Krantz MJ & Lowery CM (2005) Images in clinical medicine. Giant Osborn waves in hypothermia. N Engl J Med 352(2): 184. Kurl S, Makikallio TH, Rautaharju P, Kiviniemi V & Laukkanen JA (2012) Duration of QRS complex in resting electrocardiogram is a predictor of sudden cardiac death in men. Circulation 125(21): 2588–2594. Leenhardt A, Lucet V, Denjoy I, Grau F, Ngoc DD & Coumel P (1995) Catecholaminergic polymorphic ventricular tachycardia in children. A 7-year follow-up of 21 patients. Circulation 91(5): 1512–1519. Lehnart SE, Ackerman MJ, Benson DW,Jr, Brugada R, Clancy CE, Donahue JK, George AL,Jr, Grant AO, Groft SC, January CT, Lathrop DA, Lederer WJ, Makielski JC, Mohler PJ, Moss A, Nerbonne JM, Olson TM, Przywara DA, Towbin JA, Wang LH & Marks AR (2007) Inherited arrhythmias: a National Heart, Lung, and Blood Institute and Office of Rare Diseases workshop consensus report about the diagnosis, phenotyping, molecular mechanisms, and therapeutic approaches for primary cardiomyopathies of gene mutations affecting ion channel function. Circulation 116(20): 2325–2345. Lepeschkin E & Surawicz B (1952) The measurement of the duration of the QRS interval. Am Heart J 44(1): 80–88. Letsas KP, Sacher F, Probst V, Weber R, Knecht S, Kalusche D, Haissaguerre M & Arentz T (2008) Prevalence of early repolarization pattern in inferolateral leads in patients with Brugada syndrome. Heart Rhythm 5(12): 1685–1689. Littmann L (2010) Early repolarization on electrocardiography. N Engl J Med 362(12): 1148; author reply 1148. Littmann L & Tenczer J (2011) Widening spectrum of the J-wave syndromes. J Electrocardiol . 128 Luu M, Stevenson WG, Stevenson LW, Baron K & Walden J (1989) Diverse mechanisms of unexpected cardiac arrest in advanced heart failure. Circulation 80(6): 1675–1680. Manolio TA, Baughman KL, Rodeheffer R, Pearson TA, Bristow JD, Michels VV, Abelmann WH & Harlan WR (1992) Prevalence and etiology of idiopathic dilated cardiomyopathy (summary of a National Heart, Lung, and Blood Institute workshop. Am J Cardiol 69(17): 1458–1466. Marcus FI (2005) Prevalence of T-wave inversion beyond V1 in young normal individuals and usefulness for the diagnosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia. Am J Cardiol 95(9): 1070–1071. Marcus FI & Fontaine G (1995) Arrhythmogenic right ventricular dysplasia/cardiomyopathy: a review. Pacing Clin Electrophysiol 18(6): 1298–1314. Marcus FI, Fontaine GH, Guiraudon G, Frank R, Laurenceau JL, Malergue C & Grosgogeat Y (1982) Right ventricular dysplasia: a report of 24 adult cases. Circulation 65(2): 384–398. Marcus FI, McKenna WJ, Sherrill D, Basso C, Bauce B, Bluemke DA, Calkins H, Corrado D, Cox MG, Daubert JP, Fontaine G, Gear K, Hauer R, Nava A, Picard MH, Protonotarios N, Saffitz JE, Sanborn DM, Steinberg JS, Tandri H, Thiene G, Towbin JA, Tsatsopoulou A, Wichter T & Zareba W (2010) Diagnosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia: proposed modification of the task force criteria. Circulation 121(13): 1533–1541. Maron BJ & Maron MS (2012) Hypertrophic cardiomyopathy. Lancet . Maron BJ, Olivotto I, Spirito P, Casey SA, Bellone P, Gohman TE, Graham KJ, Burton DA & Cecchi F (2000) Epidemiology of hypertrophic cardiomyopathy-related death: revisited in a large non-referral-based patient population. Circulation 102(8): 858–864. Maron BJ, Thompson PD, Puffer JC, McGrew CA, Strong WB, Douglas PS, Clark LT, Mitten MJ, Crawford MD, Atkins DL, Driscoll DJ & Epstein AE (1998) Cardiovascular preparticipation screening of competitive athletes: addendum: an addendum to a statement for health professionals from the Sudden Death Committee (Council on Clinical Cardiology) and the Congenital Cardiac Defects Committee (Council on Cardiovascular Disease in the Young), American Heart Association. Circulation 97(22): 2294. Martini B, Nava A, Thiene G, Buja GF, Canciani B, Scognamiglio R, Daliento L & Dalla Volta S (1989) Ventricular fibrillation without apparent heart disease: description of six cases. Am Heart J 118(6): 1203–1209. Matsuo K, Akahoshi M, Nakashima E, Suyama A, Seto S, Hayano M & Yano K (2001) The prevalence, incidence and prognostic value of the Brugada-type electrocardiogram: a population-based study of four decades. J Am Coll Cardiol 38(3): 765–770. Matsuo K, Kurita T, Inagaki M, Kakishita M, Aihara N, Shimizu W, Taguchi A, Suyama K, Kamakura S & Shimomura K (1999) The circadian pattern of the development of ventricular fibrillation in patients with Brugada syndrome. Eur Heart J 20(6): 465–470. 129 Matsuo K, Shimizu W, Kurita T, Inagaki M, Aihara N & Kamakura S (1998) Dynamic changes of 12-lead electrocardiograms in a patient with Brugada syndrome. J Cardiovasc Electrophysiol 9(5): 508–512. Maury P, Extramiana F, Sbragia P, Giustetto C, Schimpf R, Duparc A, Wolpert C, Denjoy I, Delay M, Borggrefe M & Gaita F (2008) Short QT syndrome. Update on a recent entity. Arch Cardiovasc Dis 101(11–12): 779–786. McKenna WJ, Thiene G, Nava A, Fontaliran F, Blomstrom-Lundqvist C, Fontaine G & Camerini F (1994) Diagnosis of arrhythmogenic right ventricular dysplasia/cardiomyopathy. Task Force of the Working Group Myocardial and Pericardial Disease of the European Society of Cardiology and of the Scientific Council on Cardiomyopathies of the International Society and Federation of Cardiology. Br Heart J 71(3): 215–218. Medeiros-Domingo A, Tan BH, Crotti L, Tester DJ, Eckhardt L, Cuoretti A, Kroboth SL, Song C, Zhou Q, Kopp D, Schwartz PJ, Makielski JC & Ackerman MJ (2010) Gainof-function mutation S422L in the KCNJ8-encoded cardiac K(ATP) channel Kir6.1 as a pathogenic substrate for J-wave syndromes. Heart Rhythm 7(10): 1466–1471. Merchant FM, Noseworthy PA, Weiner RB, Singh SM, Ruskin JN & Reddy VY (2009) Ability of terminal QRS notching to distinguish benign from malignant electrocardiographic forms of early repolarization. Am J Cardiol 104(10): 1402–1406. Miller WL, Ballman KV, Hodge DO, Rodeheffer RJ & Hammill SC (2005) Risk factor implications of incidentally discovered uncomplicated bundle branch block. Mayo Clin Proc 80(12): 1585–1590. Miller WL, Hodge DO & Hammill SC (2008) Association of uncomplicated electrocardiographic conduction blocks with subsequent cardiac morbidity in a community-based population (Olmsted County, Minnesota). Am J Cardiol 101(1): 102–106. Miyamoto A, Hayashi H, Yoshino T, Kawaguchi T, Taniguchi A, Itoh H, Sugimoto Y, Itoh M, Makiyama T, Xue JQ, Murakami Y & Horie M (2012) Clinical and electrocardiographic characteristics of patients with short QT interval in a large hospital-based population. Heart Rhythm 9(1): 66–74. Miyasaka Y, Tsuji H, Yamada K, Tokunaga S, Saito D, Imuro Y, Matsumoto N & Iwasaka T (2001) Prevalence and mortality of the Brugada-type electrocardiogram in one city in Japan. J Am Coll Cardiol 38(3): 771–774. Mizumaki K, Nishida K, Iwamoto J, Nakatani Y, Yamaguchi Y, Sakamoto T, Tsuneda T, Kataoka N & Inoue H (2012) Vagal activity modulates spontaneous augmentation of J-wave elevation in patients with idiopathic ventricular fibrillation. Heart Rhythm 9(2): 249–255. Morita H, Kusano KF, Miura D, Nagase S, Nakamura K, Morita ST, Ohe T, Zipes DP & Wu J (2008a) Fragmented QRS as a marker of conduction abnormality and a predictor of prognosis of Brugada syndrome. Circulation 118(17): 1697–1704. 130 Morita H, Kusano KF, Miura D, Nagase S, Nakamura K, Morita ST, Ohe T, Zipes DP & Wu J (2008b) Fragmented QRS as a marker of conduction abnormality and a predictor of prognosis of Brugada syndrome. Circulation 118(17): 1697–1704. Morita H, Morita ST, Nagase S, Banba K, Nishii N, Tani Y, Watanabe A, Nakamura K, Kusano KF, Emori T, Matsubara H, Hina K, Kita T & Ohe T (2003) Ventricular arrhythmia induced by sodium channel blocker in patients with Brugada syndrome. J Am Coll Cardiol 42(9): 1624–1631. Moriya M, Seto S, Yano K & Akahoshi M (2007) Two cases of short QT interval. Pacing Clin Electrophysiol 30(12): 1522–1526. Moss AJ, Schwartz PJ, Crampton RS, Tzivoni D, Locati EH, MacCluer J, Hall WJ, Weitkamp L, Vincent GM & Garson A,Jr (1991) The long QT syndrome. Prospective longitudinal study of 328 families. Circulation 84(3): 1136–1144. Moukabary T (2007) Willem Einthoven (1860–1927): Father of electrocardiography. Cardiol J 14(3): 316–317. Myerburg RJ (2001) Sudden cardiac death: exploring the limits of our knowledge. J Cardiovasc Electrophysiol 12(3): 369–381. Myerburg RJ (2002) Scientific gaps in the prediction and prevention of sudden cardiac death. J Cardiovasc Electrophysiol 13(7): 709–723. Myerburg RJ & Castellanos A (2008) Early repolarization and sudden cardiac arrest: theme or variation on a theme? Nat Clin Pract Cardiovasc Med 5(12): 760–761. Myerburg RJ & Vetter VL (2007) Electrocardiograms should be included in preparticipation screening of athletes. Circulation 116(22): 2616–26; discussion 2626. Myers GB & Klein HA (1947a) Normal variations in multiple precordial leads. Am Heart J 34(6): 785–808. Myers GB & Klein HA (1947b) Normal variations in multiple precordial leads. Am Heart J 34(6): 785–808. Myojo T, Sato N, Nimura A, Matsuo A, Taniguchi O, Nakamura H, Karim Talib A, Sakamoto N, Takeuchi T, Kawamura Y & Hasebe N (2012) Recurrent ventricular fibrillation related to hypokalemia in early repolarization syndrome. Pacing Clin Electrophysiol 35(8): e234–8. Nademanee K, Veerakul G, Chandanamattha P, Chaothawee L, Ariyachaipanich A, Jirasirirojanakorn K, Likittanasombat K, Bhuripanyo K & Ngarmukos T (2011) Prevention of ventricular fibrillation episodes in Brugada syndrome by catheter ablation over the anterior right ventricular outflow tract epicardium. Circulation 123(12): 1270–1279. Nademanee K, Veerakul G, Nimmannit S, Chaowakul V, Bhuripanyo K, Likittanasombat K, Tunsanga K, Kuasirikul S, Malasit P, Tansupasawadikul S & Tatsanavivat P (1997) Arrhythmogenic marker for the sudden unexplained death syndrome in Thai men. Circulation 96(8): 2595–2600. Nagao S, Hayashi Y, Yagihara N, Sato A, Watanabe H, Furushima H, Chinushi M & Aizawa Y (2011) Preexcitation unmasks J waves: 2 cases. J Electrocardiol 44(3): 359–362. 131 Nakayama M, Sato M, Kitazawa H, Saito A, Ikeda Y, Fujita S, Fuse K, Takahashi M, Takarada K, Oguro T, Matsushita H, Okabe M, Yamashina A & Aizawa Y (2013) Jwaves in patients with an acute ST-elevation myocardial infarction who underwent successful percutaneous coronary intervention: prevalence, pathogenesis, and clinical implication. Europace 15(1): 109–115. Nam GB, Kim YH & Antzelevitch C (2008) Augmentation of J waves and electrical storms in patients with early repolarization. N Engl J Med 358(19): 2078–2079. Naruse Y, Tada H, Harimura Y, Hayashi M, Noguchi Y, Sato A, Yoshida K, Sekiguchi Y & Aonuma K (2012) Early repolarization is an independent predictor of occurrences of ventricular fibrillation in the very early phase of acute myocardial infarction. Circ Arrhythm Electrophysiol 5(3): 506–513. Newton-Cheh C, Eijgelsheim M, Rice KM, de Bakker PI, Yin X, Estrada K, Bis JC, Marciante K, Rivadeneira F, Noseworthy PA, Sotoodehnia N, Smith NL, Rotter JI, Kors JA, Witteman JC, Hofman A, Heckbert SR, O'Donnell CJ, Uitterlinden AG, Psaty BM, Lumley T, Larson MG & Stricker BH (2009) Common variants at ten loci influence QT interval duration in the QTGEN Study. Nat Genet 41(4): 399–406. Nikolic G & Marriott HJ (1985) Left bundle branch block with right axis deviation: a marker of congestive cardiomyopathy. J Electrocardiol 18(4): 395–404. Norman MW & McKenna WJ (1999) Arrhythmogenic right ventricular cardiomyopathy: perspectives on disease. Z Kardiol 88(8): 550–554. Noseworthy PA, Tikkanen JT, Porthan K, Oikarinen L, Pietila A, Harald K, Peloso GM, Merchant FM, Jula A, Vaananen H, Hwang SJ, O'Donnell CJ, Salomaa V, NewtonCheh C & Huikuri HV (2011a) The early repolarization pattern in the general population: clinical correlates and heritability. J Am Coll Cardiol 57(22): 2284–2289. Noseworthy PA, Tikkanen JT, Porthan K, Oikarinen L, Pietila A, Harald K, Peloso GM, Merchant FM, Jula A, Vaananen H, Hwang SJ, O'Donnell CJ, Salomaa V, NewtonCheh C & Huikuri HV (2011b) The early repolarization pattern in the general population: clinical correlates and heritability. J Am Coll Cardiol 57(22): 2284–2289. Noseworthy PA, Weiner R, Kim J, Keelara V, Wang F, Berkstresser B, Wood MJ, Wang TJ, Picard MH, Hutter AM,Jr, Newton-Cheh C & Baggish AL (2011c) Early repolarization pattern in competitive athletes: clinical correlates and the effects of exercise training. Circ Arrhythm Electrophysiol 4(4): 432–440. Nunn LM, Bhar-Amato J, Lowe MD, Macfarlane PW, Rogers P, McKenna WJ, Elliott PM & Lambiase PD (2011) Prevalence of J-point elevation in sudden arrhythmic death syndrome families. J Am Coll Cardiol 58(3): 286–290. Oh CM, Oh J, Shin DH, Hwang HJ, Kim BK, Pak HN, Lee MH & Joung B (2012) Early repolarization pattern predicts cardiac death and fatal arrhythmia in patients with vasospastic angina. Int J Cardiol . 132 Oikarinen L, Nieminen MS, Viitasalo M, Toivonen L, Jern S, Dahlof B, Devereux RB, Okin PM & LIFE Study Investigators (2004) QRS duration and QT interval predict mortality in hypertensive patients with left ventricular hypertrophy: the Losartan Intervention for Endpoint Reduction in Hypertension Study. Hypertension 43(5): 1029–1034. Olson KA, Viera AJ, Soliman EZ, Crow RS & Rosamond WD (2011) Long-term prognosis associated with J-point elevation in a large middle-aged biracial cohort: the ARIC study. Eur Heart J 32(24): 3098–3106. O'Malley PG & Redberg RF (2010) Risk refinement, reclassification, and treatment thresholds in primary prevention of cardiovascular disease: incremental progress but significant gaps remain. Arch Intern Med 170(17): 1602–1603. Osborn JJ (1953) Experimental hypothermia; respiratory and blood pH changes in relation to cardiac function. Am J Physiol 175(3): 389–398. Papadakis M, Sharma S, Cox S, Sheppard MN, Panoulas VF & Behr ER (2009) The magnitude of sudden cardiac death in the young: a death certificate-based review in England and Wales. Europace 11(10): 1353–1358. Patel RB, Ilkhanoff L, Ng J, Chokshi M, Mouchli A, Chacko SJ, Subacius H, Bhojraj S, Goldberger JJ & Kadish AH (2012) Clinical characteristics and prevalence of early repolarization associated with ventricular arrhythmias following acute ST-elevation myocardial infarction. Am J Cardiol 110(5): 615–620. Patel RB, Ng J, Reddy V, Chokshi M, Parikh K, Subacius H, Alsheikh-Ali AA, Nguyen T, Link MS, Goldberger JJ, Ilkhanoff L & Kadish AH (2010) Early repolarization associated with ventricular arrhythmias in patients with chronic coronary artery disease. Circ Arrhythm Electrophysiol 3(5): 489–495. Pei J, Li N, Gao Y, Wang Z, Li X, Zhang Y, Chen J, Zhang P, Cao K & Pu J (2012) The J wave and fragmented QRS complexes in inferior leads associated with sudden cardiac death in patients with chronic heart failure. Europace 14(8): 1180–1187. Perez MV, Friday K & Froelicher V (2012) Semantic Confusion: The Case of Early Repolarization and the J Point. Am J Med . Peters S, Truemmel M & Koehler B (2012) Prognostic value of QRS fragmentation in patients with arrhythmogenic right ventricular cardiomyopathy/dysplasia. J Cardiovasc Med (Hagerstown) 13(5): 295–298. Peters S, Trummel M & Koehler B (2008) QRS fragmentation in standard ECG as a diagnostic marker of arrhythmogenic right ventricular dysplasia-cardiomyopathy. Heart Rhythm 5(10): 1417–1421. Pfeufer A, Sanna S, Arking DE, Muller M, Gateva V, Fuchsberger C, Ehret GB, Orru M, Pattaro C, Kottgen A, Perz S, Usala G, Barbalic M, Li M, Putz B, Scuteri A, Prineas RJ, Sinner MF, Gieger C, Najjar SS, Kao WH, Muhleisen TW, Dei M, Happle C, Mohlenkamp S, Crisponi L, Erbel R, Jockel KH, Naitza S, Steinbeck G, Marroni F, Hicks AA, Lakatta E, Muller-Myhsok B, Pramstaller PP, Wichmann HE, Schlessinger D, Boerwinkle E, Meitinger T, Uda M, Coresh J, Kaab S, Abecasis GR & Chakravarti A (2009) Common variants at ten loci modulate the QT interval duration in the QTSCD Study. Nat Genet 41(4): 407–414. 133 Pfeufer A, van Noord C, Marciante KD, Arking DE, Larson MG, Smith AV, Tarasov KV, Muller M, Sotoodehnia N, Sinner MF, Verwoert GC, Li M, Kao WH, Kottgen A, Coresh J, Bis JC, Psaty BM, Rice K, Rotter JI, Rivadeneira F, Hofman A, Kors JA, Stricker BH, Uitterlinden AG, van Duijn CM, Beckmann BM, Sauter W, Gieger C, Lubitz SA, Newton-Cheh C, Wang TJ, Magnani JW, Schnabel RB, Chung MK, Barnard J, Smith JD, Van Wagoner DR, Vasan RS, Aspelund T, Eiriksdottir G, Harris TB, Launer LJ, Najjar SS, Lakatta E, Schlessinger D, Uda M, Abecasis GR, MullerMyhsok B, Ehret GB, Boerwinkle E, Chakravarti A, Soliman EZ, Lunetta KL, Perz S, Wichmann HE, Meitinger T, Levy D, Gudnason V, Ellinor PT, Sanna S, Kaab S, Witteman JC, Alonso A, Benjamin EJ & Heckbert SR (2010) Genome-wide association study of PR interval. Nat Genet 42(2): 153–159. Pietrasik G, Goldenberg I, Zdzienicka J, Moss AJ & Zareba W (2007) Prognostic significance of fragmented QRS complex for predicting the risk of recurrent cardiac events in patients with Q-wave myocardial infarction. Am J Cardiol 100(4): 583–586. Priori S (1997) Survivors of out-of-hospital cardiac arrest with apparently normal heart. Need for definition and standardized clinical evaluation. Consensus Statement of the Joint Steering Committees of the Unexplained Cardiac Arrest Registry of Europe and of the Idiopathic Ventricular Fibrillation Registry of the United States. Circulation 95(1): 265–272. Priori SG (2004) Inherited arrhythmogenic diseases: the complexity beyond monogenic disorders. Circ Res 94(2): 140–145. Priori SG, Aliot E, Blomstrom-Lundqvist C, Bossaert L, Breithardt G, Brugada P, Camm AJ, Cappato R, Cobbe SM, Di Mario C, Maron BJ, McKenna WJ, Pedersen AK, Ravens U, Schwartz PJ, Trusz-Gluza M, Vardas P, Wellens HJ & Zipes DP (2001) Task Force on Sudden Cardiac Death of the European Society of Cardiology. Eur Heart J 22(16): 1374–1450. Priori SG, Napolitano C, Gasparini M, Pappone C, Della Bella P, Giordano U, Bloise R, Giustetto C, De Nardis R, Grillo M, Ronchetti E, Faggiano G & Nastoli J (2002) Natural history of Brugada syndrome: insights for risk stratification and management. Circulation 105(11): 1342–1347. Priori SG, Napolitano C, Schwartz PJ, Grillo M, Bloise R, Ronchetti E, Moncalvo C, Tulipani C, Veia A, Bottelli G & Nastoli J (2004) Association of long QT syndrome loci and cardiac events among patients treated with beta-blockers. JAMA 292(11): 1341–1344. Priori SG, Pandit SV, Rivolta I, Berenfeld O, Ronchetti E, Dhamoon A, Napolitano C, Anumonwo J, di Barletta MR, Gudapakkam S, Bosi G, Stramba-Badiale M & Jalife J (2005) A novel form of short QT syndrome (SQT3) is caused by a mutation in the KCNJ2 gene. Circ Res 96(7): 800–807. Priori SG, Schwartz PJ, Napolitano C, Bloise R, Ronchetti E, Grillo M, Vicentini A, Spazzolini C, Nastoli J, Bottelli G, Folli R & Cappelletti D (2003) Risk stratification in the long-QT syndrome. N Engl J Med 348(19): 1866–1874. 134 Probst V, Veltmann C, Eckardt L, Meregalli PG, Gaita F, Tan HL, Babuty D, Sacher F, Giustetto C, Schulze-Bahr E, Borggrefe M, Haissaguerre M, Mabo P, Le Marec H, Wolpert C & Wilde AA (2010a) Long-term prognosis of patients diagnosed with Brugada syndrome: Results from the FINGER Brugada Syndrome Registry. Circulation 121(5): 635–643. Probst V, Veltmann C, Eckardt L, Meregalli PG, Gaita F, Tan HL, Babuty D, Sacher F, Giustetto C, Schulze-Bahr E, Borggrefe M, Haissaguerre M, Mabo P, Le Marec H, Wolpert C & Wilde AA (2010b) Long-term prognosis of patients diagnosed with Brugada syndrome: Results from the FINGER Brugada Syndrome Registry. Circulation 121(5): 635–643. Puranik R, Chow CK, Duflou JA, Kilborn MJ & McGuire MA (2005) Sudden death in the young. Heart Rhythm 2(12): 1277–1282. Rautaharju PM, Surawicz B, Gettes LS, Bailey JJ, Childers R, Deal BJ, Gorgels A, Hancock EW, Josephson M, Kligfield P, Kors JA, Macfarlane P, Mason JW, Mirvis DM, Okin P, Pahlm O, van Herpen G, Wagner GS, Wellens H, American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology, American College of Cardiology Foundation & Heart Rhythm Society (2009a) AHA/ACCF/HRS recommendations for the standardization and interpretation of the electrocardiogram: part IV: the ST segment, T and U waves, and the QT interval: a scientific statement from the American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society. Endorsed by the International Society for Computerized Electrocardiology. J Am Coll Cardiol 53(11): 982–991. Rautaharju PM, Surawicz B, Gettes LS, Bailey JJ, Childers R, Deal BJ, Gorgels A, Hancock EW, Josephson M, Kligfield P, Kors JA, Macfarlane P, Mason JW, Mirvis DM, Okin P, Pahlm O, van Herpen G, Wagner GS, Wellens H, American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology, American College of Cardiology Foundation & Heart Rhythm Society (2009b) AHA/ACCF/HRS recommendations for the standardization and interpretation of the electrocardiogram: part IV: the ST segment, T and U waves, and the QT interval: a scientific statement from the American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society. Endorsed by the International Society for Computerized Electrocardiology. J Am Coll Cardiol 53(11): 982–991. Reinhard W, Kaess BM, Debiec R, Nelson CP, Stark K, Tobin MD, Macfarlane PW, Tomaszewski M, Samani NJ & Hengstenberg C (2011) Heritability of early repolarization: a population-based study. Circ Cardiovasc Genet 4(2): 134–138. 135 Richardson P, McKenna W, Bristow M, Maisch B, Mautner B, O'Connell J, Olsen E, Thiene G, Goodwin J, Gyarfas I, Martin I & Nordet P (1996) Report of the 1995 World Health Organization/International Society and Federation of Cardiology Task Force on the Definition and Classification of cardiomyopathies. Circulation 93(5): 841–842. Richter S, Sarkozy A, Paparella G, Henkens S, Boussy T, Chierchia GB, Brugada R, Brugada J & Brugada P (2010) Number of electrocardiogram leads displaying the diagnostic coved-type pattern in Brugada syndrome: a diagnostic consensus criterion to be revised. Eur Heart J 31(11): 1357–1364. Riera AR, Uchida AH, Schapachnik E, Dubner S, Zhang L, Celso Ferreira F & Ferreira C (2008a) Early repolarization variant: epidemiological aspects, mechanism, and differential diagnosis. Cardiol J 15(1): 4–16. Riera AR, Uchida AH, Schapachnik E, Dubner S, Zhang L, Celso Ferreira F & Ferreira C (2008b) Early repolarization variant: epidemiological aspects, mechanism, and differential diagnosis. Cardiol J 15(1): 4–16. Rollin A, Maury P, Bongard V, Sacher F, Delay M, Duparc A, Mondoly P, Carrie D, Ferrieres J & Ruidavets JB (2012) Prevalence, Prognosis, and Identification of the Malignant Form of Early Repolarization Pattern in a Population-Based Study. Am J Cardiol . Rosso R, Glikson E, Belhassen B, Katz A, Halkin A, Steinvil A & Viskin S (2011) Distinguishing "benign" from "malignant early repolarization": The value of the STsegment morphology. Heart Rhythm . Rosso R, Kogan E, Belhassen B, Rozovski U, Scheinman MM, Zeltser D, Halkin A, Steinvil A, Heller K, Glikson M, Katz A & Viskin S (2008) J-point elevation in survivors of primary ventricular fibrillation and matched control subjects: incidence and clinical significance. J Am Coll Cardiol 52(15): 1231–1238. Roten L, Derval N, Sacher F, Pascale P, Scherr D, Komatsu Y, Ramoul K, Daly M, Denis A, Shah AJ, Hocini M, Jais P & Haissaguerre M (2012a) Heterogeneous Response of J wave Syndromes to Beta-Adrenergic Stimulation. Heart Rhythm . Roten L, Derval N, Sacher F, Pascale P, Wilton SB, Scherr D, Shah A, Pedersen ME, Jadidi AS, Miyazaki S, Knecht S, Hocini M, Jais P & Haissaguerre M (2012b) Ajmaline attenuates electrocardiogram characteristics of inferolateral early repolarization. Heart Rhythm 9(2): 232–239. Rotman M & Triebwasser JH (1975) A clinical and follow-up study of right and left bundle branch block. Circulation 51(3): 477–484. Rowin EJ, Maron BJ, Appelbaum E, Link MS, Gibson CM, Lesser JR, Haas TS, Udelson JE, Manning WJ & Maron MS (2012) Significance of False Negative Electrocardiograms in Preparticipation Screening of Athletes for Hypertrophic Cardiomyopathy. Am J Cardiol . Rubart M & Zipes DP (2005) Mechanisms of sudden cardiac death. J Clin Invest 115(9): 2305-2315. 136 Rudic B, Veltmann C, Kuntz E, Behnes M, Elmas E, Konrad T, Kuschyk J, Weiss C, Borggrefe M & Schimpf R (2012) Early repolarization pattern is associated with ventricular fibrillation in patients with acute myocardial infarction. Heart Rhythm . Sahara M, Sagara K, Yamashita T, Abe T, Kirigaya H, Nakada M, Iinuma H, Fu LT & Watanabe H (2002) J wave and ST segment elevation in the inferior leads: a latent type of variant Brugada syndrome? Jpn Heart J 43(1): 55–60. Sarkozy A, Boussy T, Kourgiannides G, Chierchia GB, Richter S, De Potter T, Geelen P, Wellens F, Spreeuwenberg MD & Brugada P (2007) Long-term follow-up of primary prophylactic implantable cardioverter-defibrillator therapy in Brugada syndrome. Eur Heart J 28(3): 334–344. Sarkozy A, Chierchia GB, Paparella G, Boussy T, De Asmundis C, Roos M, Henkens S, Kaufman L, Buyl R, Brugada R, Brugada J & Brugada P (2009) Inferior and lateral electrocardiographic repolarization abnormalities in Brugada syndrome. Circ Arrhythm Electrophysiol 2(2): 154–161. Savage DD, Seides SF, Clark CE, Henry WL, Maron BJ, Robinson FC & Epstein SE (1978) Electrocardiographic findings in patients with obstructive and nonobstructive hypertrophic cardiomyopathy. Circulation 58(3 Pt 1): 402–408. Scherlag BJ, Gunn CG, Berbari EJ & Lazzara R (1985a) Peri-infarction block (1950)-late potentials (1980): their relationship, significance and diagnostic implications. Am J Cardiol 55(6): 839–841. Scherlag BJ, Gunn CG, Berbari EJ & Lazzara R (1985b) Peri-infarction block (1950)-late potentials (1980): their relationship, significance and diagnostic implications. Am J Cardiol 55(6): 839–841. Schwartz PJ, Moss AJ, Vincent GM & Crampton RS (1993) Diagnostic criteria for the long QT syndrome. An update. Circulation 88(2): 782–784. Schwartz PJ, Priori SG, Spazzolini C, Moss AJ, Vincent GM, Napolitano C, Denjoy I, Guicheney P, Breithardt G, Keating MT, Towbin JA, Beggs AH, Brink P, Wilde AA, Toivonen L, Zareba W, Robinson JL, Timothy KW, Corfield V, Wattanasirichaigoon D, Corbett C, Haverkamp W, Schulze-Bahr E, Lehmann MH, Schwartz K, Coumel P & Bloise R (2001) Genotype-phenotype correlation in the long-QT syndrome: genespecific triggers for life-threatening arrhythmias. Circulation 103(1): 89–95. Schwartz PJ, Stramba-Badiale M, Crotti L, Pedrazzini M, Besana A, Bosi G, Gabbarini F, Goulene K, Insolia R, Mannarino S, Mosca F, Nespoli L, Rimini A, Rosati E, Salice P & Spazzolini C (2009) Prevalence of the congenital long-QT syndrome. Circulation 120(18): 1761–1767. Shimizu W, Antzelevitch C, Suyama K, Kurita T, Taguchi A, Aihara N, Takaki H, Sunagawa K & Kamakura S (2000) Effect of sodium channel blockers on ST segment, QRS duration, and corrected QT interval in patients with Brugada syndrome. J Cardiovasc Electrophysiol 11(12): 1320–1329. Shimizu W, Matsuo K, Kokubo Y, Satomi K, Kurita T, Noda T, Nagaya N, Suyama K, Aihara N, Kamakura S, Inamoto N, Akahoshi M & Tomoike H (2007) Sex hormone and gender difference--role of testosterone on male predominance in Brugada syndrome. J Cardiovasc Electrophysiol 18(4): 415–421. 137 Shinohara T, Takahashi N, Saikawa T & Yoshimatsu H (2006a) Characterization of J wave in a patient with idiopathic ventricular fibrillation. Heart Rhythm 3(9): 1082–1084. Shinohara T, Takahashi N, Saikawa T & Yoshimatsu H (2006b) Characterization of J wave in a patient with idiopathic ventricular fibrillation. Heart Rhythm 3(9): 1082– 1084. Shipley R & Hallaran W (1936) The four lead electrocardiogram in 200 normal menand women. Am Heart J 11: 325–345. Sinner MF, Pfeufer A, Perz S, Schulze-Bahr E, Monnig G, Eckardt L, Beckmann BM, Wichmann HE, Breithardt G, Steinbeck G, Fabritz L, Kaab S & Kirchhof P (2009) Spontaneous Brugada electrocardiogram patterns are rare in the German general population: results from the KORA study. Europace 11(10): 1338–1344. Sinner MF, Porthan K, Noseworthy PA, Havulinna AS, Tikkanen J, Muller-Nurasyid M, Peloso G, Ulivi S, Beckmann BM, Brockhaus AC, Cooper RR, Gasparini P, Hengstenberg C, Hwang SJ, Iorio A, Junttila MJ, Klopp N, Kahonen M, Laaksonen MA, Lehtimaki T, Lichtner P, Lyytikainen LP, Martens E, Meisinger C, Meitinger T, Merchant FM, Nieminen MS, Peters A, Pietila A, Perz S, Oikarinen L, Raitakari O, Reinhard W, Silander K, Thorand B, Wichmann HE, Sinagra G, Viikari J, O'Donnell CJ, Ellinor PT, Huikuri HV, Kaab S, Newton-Cheh C & Salomaa V (2012) A metaanalysis of genome-wide association studies of the electrocardiographic early repolarization pattern. Heart Rhythm . Sinner MF, Reinhard W, Muller M, Beckmann BM, Martens E, Perz S, Pfeufer A, Winogradow J, Stark K, Meisinger C, Wichmann HE, Peters A, Riegger GA, Steinbeck G, Hengstenberg C & Kaab S (2010) Association of early repolarization pattern on ecg with risk of cardiac and all-cause mortality: a population-based prospective cohort study (MONICA/KORA). PLoS Med 7(7): e1000314. Soliman EZ, Elsalam MA & Li Y (2011) Early repolarization and markers of ventricular arrhythmogenesis in patients referred to ambulatory 24-hour ECG recording. Int J Cardiol . 138 Sotoodehnia N, Isaacs A, de Bakker PI, Dorr M, Newton-Cheh C, Nolte IM, van der Harst P, Muller M, Eijgelsheim M, Alonso A, Hicks AA, Padmanabhan S, Hayward C, Smith AV, Polasek O, Giovannone S, Fu J, Magnani JW, Marciante KD, Pfeufer A, Gharib SA, Teumer A, Li M, Bis JC, Rivadeneira F, Aspelund T, Kottgen A, Johnson T, Rice K, Sie MP, Wang YA, Klopp N, Fuchsberger C, Wild SH, Mateo Leach I, Estrada K, Volker U, Wright AF, Asselbergs FW, Qu J, Chakravarti A, Sinner MF, Kors JA, Petersmann A, Harris TB, Soliman EZ, Munroe PB, Psaty BM, Oostra BA, Cupples LA, Perz S, de Boer RA, Uitterlinden AG, Volzke H, Spector TD, Liu FY, Boerwinkle E, Dominiczak AF, Rotter JI, van Herpen G, Levy D, Wichmann HE, van Gilst WH, Witteman JC, Kroemer HK, Kao WH, Heckbert SR, Meitinger T, Hofman A, Campbell H, Folsom AR, van Veldhuisen DJ, Schwienbacher C, O'Donnell CJ, Volpato CB, Caulfield MJ, Connell JM, Launer L, Lu X, Franke L, Fehrmann RS, te Meerman G, Groen HJ, Weersma RK, van den Berg LH, Wijmenga C, Ophoff RA, Navis G, Rudan I, Snieder H, Wilson JF, Pramstaller PP, Siscovick DS, Wang TJ, Gudnason V, van Duijn CM, Felix SB, Fishman GI, Jamshidi Y, Stricker BH, Samani NJ, Kaab S & Arking DE (2010) Common variants in 22 loci are associated with QRS duration and cardiac ventricular conduction. Nat Genet 42(12): 1068–1076. Sridharan MR & Horan LG (1984) Electrocardiographic J wave of hypercalcemia. Am J Cardiol 54(6): 672–673. Straus SM, Kors JA, De Bruin ML, van der Hooft CS, Hofman A, Heeringa J, Deckers JW, Kingma JH, Sturkenboom MC, Stricker BH & Witteman JC (2006) Prolonged QTc interval and risk of sudden cardiac death in a population of older adults. J Am Coll Cardiol 47(2): 362–367. Sumitomo N, Harada K, Nagashima M, Yasuda T, Nakamura Y, Aragaki Y, Saito A, Kurosaki K, Jouo K, Koujiro M, Konishi S, Matsuoka S, Oono T, Hayakawa S, Miura M, Ushinohama H, Shibata T & Niimura I (2003) Catecholaminergic polymorphic ventricular tachycardia: electrocardiographic characteristics and optimal therapeutic strategies to prevent sudden death. Heart 89(1): 66–70. Surawicz B & Macfarlane PW (2011) Inappropriate and confusing electrocardiographic terms: J-wave syndromes and early repolarization. J Am Coll Cardiol 57(15): 1584– 1586. Takagi M, Aihara N, Takaki H, Taguchi A, Shimizu W, Kurita T, Suyama K & Kamakura S (2000) Clinical characteristics of patients with spontaneous or inducible ventricular fibrillation without apparent heart disease presenting with J wave and ST segment elevation in inferior leads. J Cardiovasc Electrophysiol 11(8): 844–848. Takagi M, Aonuma K, Sekiguchi Y, Yokoyama Y, Aihara N & Hiraoka M (2012) The Prognostic Value of Early Repolarization (J wave) and ST-segment Morphology after J wave in Brugada Syndrome: Multi-center Study in Japan. Heart Rhythm . Tatsumi H, Takagi M, Nakagawa E, Yamashita H & Yoshiyama M (2006) Risk stratification in patients with Brugada syndrome: analysis of daily fluctuations in 12lead electrocardiogram (ECG) and signal-averaged electrocardiogram (SAECG). J Cardiovasc Electrophysiol 17(7): 705–711. 139 Teodorescu C, Reinier K, Uy-Evanado A, Navarro J, Mariani R, Gunson K, Jui J & Chugh SS (2011) Prolonged QRS duration on the resting ECG is associated with sudden death risk in coronary disease, independent of prolonged ventricular repolarization. Heart Rhythm 8(10): 1562–1567. Tereshchenko LG, McCabe A, Han L, Sur S, Huang T, Marine JE, Cheng A, Spragg DD, Sinha S, Calkins H, Stein K, Tomaselli GF & Berger RD (2012) Intracardiac J-point elevation before the onset of polymorphic ventricular tachycardia and ventricular fibrillation in patients with an implantable cardioverter-defibrillator. Heart Rhythm . Tester DJ, Will ML, Haglund CM & Ackerman MJ (2006) Effect of clinical phenotype on yield of long QT syndrome genetic testing. J Am Coll Cardiol 47(4): 764–768. Thiene G, Nava A, Corrado D, Rossi L & Pennelli N (1988) Right ventricular cardiomyopathy and sudden death in young people. N Engl J Med 318(3): 129–133. Tikkanen JT, Anttonen O, Junttila MJ, Aro AL, Kerola T, Rissanen HA, Reunanen A & Huikuri HV (2009a) Long-term outcome associated with early repolarization on electrocardiography. N Engl J Med 361(26): 2529–2537. Tikkanen JT, Anttonen O, Junttila MJ, Aro AL, Kerola T, Rissanen HA, Reunanen A & Huikuri HV (2009b) Long-term outcome associated with early repolarization on electrocardiography. N Engl J Med 361(26): 2529–2537. Tikkanen JT, Junttila MJ, Anttonen O, Aro AL, Luttinen S, Kerola T, Sager SJ, Rissanen HA, Myerburg RJ, Reunanen A & Huikuri HV (2011) Early repolarization: electrocardiographic phenotypes associated with favorable long-term outcome. Circulation 123(23): 2666–2673. Torigoe K, Tamura A, Kawano Y, Shinozaki K, Kotoku M & Kadota J (2012) The number of leads with fragmented QRS is independently associated with cardiac death or hospitalization for heart failure in patients with prior myocardial infarction. J Cardiol 59(1): 36–41. Tsunoda Y, Takeishi Y, Nozaki N, Kitahara T & Kubota I (2004) Presence of intermittent J waves in multiple leads in relation to episode of atrial and ventricular fibrillation. J Electrocardiol 37(4): 311–314. Uberoi A, Jain NA, Perez M, Weinkopff A, Ashley E, Hadley D, Turakhia MP & Froelicher V (2011) Early repolarization in an ambulatory clinical population. Circulation 124(20): 2208–2214. Uberoi A, Sallam K, Perez M, Jain NA, Ashley E & Froelicher V (2012) Prognostic implications of q waves and T-wave inversion associated with early repolarization. Mayo Clin Proc 87(7): 614–619. Vaillancourt C, Stiell IG & Canadian Cardiovascular Outcomes Research Team (2004) Cardiac arrest care and emergency medical services in Canada. Can J Cardiol 20(11): 1081–1090. Varriale P & Chryssos BE (1992) The RSR' complex not related to right bundle branch block: diagnostic value as a sign of myocardial infarction scar. Am Heart J 123(2): 369–376. 140 Veltmann C, Schimpf R, Echternach C, Eckardt L, Kuschyk J, Streitner F, Spehl S, Borggrefe M & Wolpert C (2006) A prospective study on spontaneous fluctuations between diagnostic and non-diagnostic ECGs in Brugada syndrome: implications for correct phenotyping and risk stratification. Eur Heart J 27(21): 2544–2552. Viskin S & Belhassen B (1990) Idiopathic ventricular fibrillation. Am Heart J 120(3): 661–671. Viskin S, Zeltser D, Ish-Shalom M, Katz A, Glikson M, Justo D, Tekes-Manova D & Belhassen B (2004) Is idiopathic ventricular fibrillation a short QT syndrome? Comparison of QT intervals of patients with idiopathic ventricular fibrillation and healthy controls. Heart Rhythm 1(5): 587–591. Waller AD (1887) A Demonstration on Man of Electromotive Changes accompanying the Heart's Beat. J Physiol 8(5): 229–234. Wang NC, Maggioni AP, Konstam MA, Zannad F, Krasa HB, Burnett JC,Jr, Grinfeld L, Swedberg K, Udelson JE, Cook T, Traver B, Zimmer C, Orlandi C, Gheorghiade M & Efficacy of Vasopressin Antagonism in Heart Failure Outcome Study With Tolvaptan (EVEREST) Investigators (2008) Clinical implications of QRS duration in patients hospitalized with worsening heart failure and reduced left ventricular ejection fraction. JAMA 299(22): 2656–2666. Wasserburger RH & Alt WJ (1961) The normal RS-T segment elevation variant. Am J Cardiol 8: 184–192. Watanabe H, Makiyama T, Koyama T, Kannankeril PJ, Seto S, Okamura K, Oda H, Itoh H, Okada M, Tanabe N, Yagihara N, Kamakura S, Horie M, Aizawa Y & Shimizu W (2010) High prevalence of early repolarization in short QT syndrome. Heart Rhythm 7(5): 647–652. Watanabe H, Nogami A, Ohkubo K, Kawata H, Hayashi Y, Ishikawa T, Makiyama T, Nagao S, Yagihara N, Takehara N, Kawamura Y, Sato A, Okamura K, Hosaka Y, Sato M, Fukae S, Chinushi M, Oda H, Okabe M, Kimura A, Maemura K, Watanabe I, Kamakura S, Horie M, Aizawa Y, Shimizu W & Makita N (2011) Electrocardiographic characteristics and SCN5A mutations in idiopathic ventricular fibrillation associated with early repolarization. Circ Arrhythm Electrophysiol 4(6): 874–881. Watanabe H, Nogami A, Ohkubo K, Kawata H, Hayashi Y, Ishikawa T, Makiyama T, Nagao S, Yagihara N, Takehara N, Kawamura Y, Sato A, Okamura K, Hosaka Y, Sato M, Fukae S, Chinushi M, Oda H, Okabe M, Kimura A, Maemura K, Watanabe I, Kamakura S, Horie M, Aizawa Y, Shimizu W & Makita N (2012) Clinical characteristics and risk of arrhythmia recurrences in patients with idiopathic ventricular fibrillation associated with early repolarization. Int J Cardiol 159(3): 238– 240. Wellens HJ (2008) Early repolarization revisited. N Engl J Med 358(19): 2063–2065. Wellens HJ, de Vreede J & Gorgels AP (1995) Sudden cardiac death. How to reduce the number of victims? Eur Heart J 16 Suppl G: 7–9. 141 Wilde AA, Antzelevitch C, Borggrefe M, Brugada J, Brugada R, Brugada P, Corrado D, Hauer RN, Kass RS, Nademanee K, Priori SG, Towbin JA & Study Group on the Molecular Basis of Arrhythmias of the European Society of Cardiology (2002) Proposed diagnostic criteria for the Brugada syndrome: consensus report. Circulation 106(19): 2514–2519. Wilde AA, Postema PG, Di Diego JM, Viskin S, Morita H, Fish JM & Antzelevitch C (2010) The pathophysiological mechanism underlying Brugada syndrome: depolarization versus repolarization. J Mol Cell Cardiol 49(4): 543–553. Wilhelm M, Brem MH, Rost C, Klinghammer L, Hennig FF, Daniel WG & Flachskampf F (2010) Early repolarization, left ventricular diastolic function, and left atrial size in professional soccer players. Am J Cardiol 106(4): 569–574. Willer CJ, Li Y & Abecasis GR (2010) METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 26(17): 2190–2191. Wu SH, Lin XX, Cheng YJ, Qiang CC & Zhang J (2012) Early Repolarization Pattern and Risk for Arrhythmia Death: A Meta-Analysis. J Am Coll Cardiol . Yan GX & Antzelevitch C (1996a) Cellular basis for the electrocardiographic J wave. Circulation 93(2): 372–379. Yan GX & Antzelevitch C (1996b) Cellular basis for the electrocardiographic J wave. Circulation 93(2): 372–379. Yan GX & Antzelevitch C (1999) Cellular basis for the Brugada syndrome and other mechanisms of arrhythmogenesis associated with ST-segment elevation. Circulation 100(15): 1660–1666. Yan GX, Lankipalli RS, Burke JF, Musco S & Kowey PR (2003) Ventricular repolarization components on the electrocardiogram: cellular basis and clinical significance. J Am Coll Cardiol 42(3): 401–409. Zimetbaum PJ, Buxton AE, Batsford W, Fisher JD, Hafley GE, Lee KL, O'Toole MF, Page RL, Reynolds M & Josephson ME (2004) Electrocardiographic predictors of arrhythmic death and total mortality in the multicenter unsustained tachycardia trial. Circulation 110(7): 766–769. Zipes DP & Wellens HJ (1998) Sudden cardiac death. Circulation 98(21): 2334–2351. 142 Original publications This thesis is based on the following original articles, which are referred to in the text by their Roman numerals. I Tikkanen JT, Anttonen O, Junttila MJ, Aro AL, Kerola T, Rissanen HA, Reunanen A & Huikuri HV (2009) Long-term outcome associated with early repolarization on electrocardiography. N Engl J Med 361: 2529–2537 II Noseworthy PA*, Tikkanen JT*, Porthan K*, Oikarinen L, Pietilä A, Harald K, Peloso GM, Merchant FM, Jula A, Väänänen H, Hwang SJ, O’Donnel CJ, Salomaa V, Newton-Cheh C & Huikuri HV (2011) The early repolarization pattern in the general population: clinical correlates and heritability. J Am Coll Cardiol 57: 2284–2289 III Tikkanen JT, Junttila MJ, Anttonen O, Aro AL, Luttinen S, Kerola T, Sager SJ, Rissanen HA, Myerburg RJ, Reunanen A & Huikuri HV (2011) Early repolarization: electrocardiographic phenotypes associated with favorable long-term outcome. Circulation 123: 2666–2673 IV Tikkanen JT, Wichmann V, Junttila MJ, Rainio M, Hookana E, Lappi O, Kortelainen ML, Anttonen O & Huikuri HV (2012) Association of early repolarization and sudden cardiac death during an acute coronary event. Circ Arrh Electrophysiol 5: 714–718. V Sinner MF*, Porthan K*, Noseworthy PA*, Havulinna AS*, Tikkanen JT*, MüllerNurasyid M, Peloso G, Ulivi S, Beckmann BM, Brockhaus AC, Cooper RR, Gasparini P, Hengstenberg C, Hwang SJ, Iorio A, Junttila MJ, Klopp N, Kähönen M, Laaksonen MA, Lehtimäki T, Lichtner P, Lyytikäinen LP, Martens E, Meisinger C, Meitinger T, Merchant FM, Nieminen MS, Peters A, Pietilä A, Perz S, Oikarinen L, Raitakari O, Reinhard W, Silander K, Thorand B, Wichmann HE, Sinagra G, Viikari J, O'Donnell CJ, Ellinor PT, Huikuri HV, Kääb S, Newton-Cheh C & Salomaa V (2012) A metaanalysis of genome-wide association studies of the electrocardiographic early repolarization pattern. Heart Rhythm 9: 1627–1634. Reprinted with permission from Massachusetts Medical Society (I), Elsevier (II, V), and Wolters Kluwer Health (III, IV). Original publications are not included in the electronical version of the dissertation. 143 144 ACTA UNIVERSITATIS OULUENSIS SERIES D MEDICA 1196. Koivikko, Minna (2013) Cardiac autonomic regulation and repolarisation during hypoglycaemia in type 1 diabetes 1197. Aro, Ellinoora (2013) Prolyl 4-hydroxylases, key enzymes regulating hypoxia response and collagen synthesis : the roles of specific isoenzymes in the control of erythropoiesis and skeletogenesis 1198. Koivumäki, Janne (2013) Biomechanical modeling of proximal femur : development of finite element models to simulate fractures 1199. Paavola, Liisa (2013) Salla disease – rare but diverse : a clinical follow-up study of a Finnish patient sample 1200. Vesterinen, Soili (2013) Osastonhoitajien johtamistyylit osana johtamiskulttuuria 1201. Huusko, Tuija (2013) Genetic and molecular background of ascending aortic aneurysms 1202. Pisto, Pauliina (2013) Fat accumulation in liver and muscle : association with adipokines and risk of cardiovascular events 1203. Yannopoulos, Fredrik (2013) Remote ischemic preconditioning as a means to protect the brain against hypothermic circulatory arrest : an experimental study on piglets 1204. Arvonen, Miika (2013) Intestinal immune activation in juvenile idiopathic arthritis 1205. Ahonkallio, Sari (2013) Endometrial thermal ablation : a choice for treatment of heavy menstrual bleeding 1206. Penttilä, Matti (2013) Duration of untreated psychosis : association with clinical and social outcomes and brain morphology in schizophrenia 1207. Wikström, Erika (2013) Epidemiology of Chlamydia trachomatis infection in Finland during 1983–2009 1208. Jokela, Tiina (2013) Analyses of kidney organogenesis through in vitro and in vivo approaches : generation of conditional Wnt4 mouse models and a method for applying inducible Cre-recombination for kidney organ culture 1209. Hietikko, Elina (2013) Genetic and clinical features of familial Meniere’s disease in Northern Ostrobothnia and Kainuu 1210. Abou Elseoud, Ahmed (2013) Exploring functional brain networks using independent component analysis : functional brain networks connectivity 1211. Aro, Aapo (2013) Electrocardiographic risk markers of sudden cardiac death in middle-aged subjects Book orders: Granum: Virtual book store http://granum.uta.fi/granum/ D 1212 OULU 2013 UNIV ER S IT Y OF OULU P. O. B R[ 00 FI-90014 UNIVERSITY OF OULU FINLAND U N I V E R S I TAT I S S E R I E S SCIENTIAE RERUM NATURALIUM Professor Esa Hohtola HUMANIORA University Lecturer Santeri Palviainen TECHNICA Postdoctoral research fellow Sanna Taskila MEDICA Professor Olli Vuolteenaho ACTA EARLY REPOLARIZATION IN THE INFEROLATERAL LEADS OF THE ELECTROCARDIOGRAM PREVALENCE, PROGNOSIS AND CHARACTERISTICS SCIENTIAE RERUM SOCIALIUM University Lecturer Hannu Heikkinen SCRIPTA ACADEMICA Director Sinikka Eskelinen OECONOMICA Professor Jari Juga EDITOR IN CHIEF Professor Olli Vuolteenaho PUBLICATIONS EDITOR Publications Editor Kirsti Nurkkala ISBN 978-952-62-0230-3 (Paperback) ISBN 978-952-62-0231-0 (PDF) ISSN 0355-3221 (Print) ISSN 1796-2234 (Online) U N I V E R S I T AT I S O U L U E N S I S Jani Tikkanen E D I T O R S Jani Tikkanen A B C D E F G O U L U E N S I S ACTA A C TA D 1212 UNIVERSITY OF OULU GRADUATE SCHOOL; UNIVERSITY OF OULU, FACULTY OF MEDICINE, INSTITUTE OF CLINICAL MEDICINE, DEPARTMENT OF INTERNAL MEDICINE; OULU UNIVERSITY HOSPITAL D MEDICA