* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Protein Buffer Systems
Survey
Document related concepts
Transcript
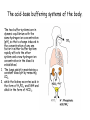
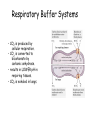
Acid-Base balance and buffer systems in the human body Department of General Chemistry Water and pH relationship In solution water shows a very low dissociation (probability of H+ in water is 1.8 x 10-9) H2O H+ + OH- H+ is actually associated with a cluster of water molecules and exists in solution as H3O+ or H5O2+ or H7O3+ Ionization of Water Ionization of Water H20 + H20 H20 Keq= [H+] [OH-] [H2O] H3O+ + OH- H+ + OHKeq=1.8 X 10-16M [H2O] = 55.5 M [H2O] Keq = [H+] [OH-] (1.8 X 10-16M)(55.5 M ) = [H+] [OH-] 1.0 X 10-14 M2 = [H+] [OH-] = Kw pH Scale Devised by Sorenson (1902) [H+] can range from 1M and 1 X 10-14M using a log scale simplifies notation pH = -log [H+] Water and pH relationship • For Dissociation of water, [H ][OH ] K [H 2 O] • Where [ ] denotes concentration and K is the dissociation constant Water and pH relationship 1 mole of water = 18g 1 L of water contains 1000 ÷ 18 = 55.56 mol (ie pure water = 55.56M) Molar concentration of H+ (or OH-) ions can be calculated [H+] = 1.8x10-9 x 55.56 = 1.0x10-7 In order to avoide using –ve numbers, the [H+] is expressed as pH which is –ve log (base 10) of [H+] pH = - log10 [H+] pH of pure water is 7 Acidic solutions have pH < 7 while basic solutions have pH > 7 Water and pH relationship [H+]=1x10-6 what is the pH? • pH= - log10 [H+] • [H+]=0.24x10-4 what is the pH? 6 4.6 • [H+]=3.4x10-3 what is the pH? 2.5 Calculating pH Effect of pH Shifts in Biological Systems • Biochemical processes & reactions are pHdependent. • Cells & organisms maintain constant specific pH value that is optimal for function. • Changes in charge can alter molecular conformation and activity. • pH sensitive molecules include enzymes, receptors, ligands, ion channels, transporters and structural proteins. Weak Acids and Bases Equilibria •Strong acids / bases – disassociate completely •Weak acids / bases – disassociate only partially •Enzyme activity sensitive to pH • weak acid/bases play important role in protein structure/function Shape of titration curve is same for all weak acids Acid/conjugate base pairs HA + H2O HA A- + H3O+ A- + H+ HA = acid ( donates H+)(Bronstad Acid) A- = Conjugate base (accepts H+)(Bronstad Base) Ka = [H+][A-] [HA] pKa = - log Ka Ka & pKa value describe tendency to loose H+ large Ka = stronger acid small Ka = weaker acid Henderson-Hasselbach Equation Consider the dissociation of a general acid HA HA H + + A- We can define a dissociation constant (K) where [H ][A ] K [HA] Rearranging gives K[HA] [H ] [A ] Taking logarithms on both sides and multiplying by -1 gives: -log[H+] = -logK – log [HA]/[A-] or pH = pK + log [A-]/[HA] Henderson-Hasselbach Equation 1) Ka = [H+][A-] [HA] HA = weak acid A- = Conjugate base 2) [H+] = Ka [HA] [A-] 3) -log[H+] = -log Ka -log [HA] [A-] 4) -log[H+] = -log Ka +log [A-] [HA] 5) pH = pKa +log [A-] [HA] * H-H equation describes the relationship between pH, pKa and buffer concentration Henderson-Hasselbalch Equation • This equation can be used to determine the pH if the pK and ratio of the ionised and unionised forms is known. • The pKa (a for acid) is the –ve log of the dissociation constant of the acid. It is the pH at which the ratio of the ionised and unionised species is equal to 1. ie the molar concentration of the ionised and unionsed species is the same. • Similarly pKb is –ve log of the dissociation constant of the base Regulation of H+ concentration Concentration of hydrogen ions is regulated sequentially by: • Chemical buffer systems –act within seconds • The respiratory center in the brain stem –acts within 1-3 min • Renal mechanisms –require hours to days to effect pH changes Sources of hydrogen ions anaerobic and aerobic respiration of glucose incomplete oxidation of fatty acids oxidation of sulfur-containing amino acids hydrolysis of phosphoproteins and nucleic acids Buffers • Definition: A weak acid plus its conjugate base that cause a solution to resist changes in pH when an acid or base are added Effectiveness of a buffer is determined by: 1) the pH of the solution, buffers work best within 1 pH unit of their pKa 2) the concentration of the buffer; the more present, the greater the buffering capacity Buffer capacity • The buffer capacity of a system is already defined as the amount of strong acid or base added to one litre (l) of the system in order to change the pH one unit The Major Body Buffer Systems The Major Body Buffer Systems Site Buffer System Comment ISF Bicarbonate For metabolic acids Phosphate Not important because concentration too low Protein Not important because concentration too low Bicarbonate Important for metabolic acids Haemoglobin Important for carbon dioxide Plasma protein Minor buffer Phosphate Concentration too low Proteins Important buffer Phosphates Important buffer Phosphate Responsible for most of 'Titratable Acidity' Ammonia Important - formation of NH4+ Ca carbonate In prolonged metabolic acidosis Blood ICF Urine Bone • Carbonic Acid – Bicarbonate Buffer System ~ Most important in the ECF Bicarbonate buffer system • Present in intra-and extracellular fluid – Bicarbonate ion acts as weak base, carbonic acid acts as a weak acid – Bicarbonate ions combine with excess hydrogen ions to form carbonic acid – Carbonic acid dissociates to release bicarbonate ions and hydrogen ions H+ + HCO3- H2CO3 H+ + HCO3- The blood buffering system, simplified Phosphate Buffer system ~ Important in ICF & urine Phosphate buffer system • Important in intracellular fluid and urine pH regulation • Consists of two phosphate ions – Monohydrogenphosphate ions act as a weak base and combine with hydrogen ions to form dihydrogenphosphate – Dihydrogenphosphate dissociates to release hydrogen ions H+ + HPO4-2 H2PO4- H+ + HPO4-2 Phosphate has three ionizable H+ and three pKas Protein Buffer Systems ~ Important in ECF and ICF ~ Interact with other buffer systems Protein buffer system • Consists of Plasma Proteins (albumin, hemoglobin) • Remember proteins are just chains of AAThe exposed amine group of the AA (NH2) accepts H+ ions when conditions are acidic • The exposed carboxyl group of AA can release H+ ions when conditions are basic Proteins can act as Acids or Bases Hemoglobin is an important blood buffer particularly for buffering CO2 • Protein buffers in blood include haemoglobin (150g/l) and plasma proteins (70g/l). Buffering is by the imidazole group of the histidine residues which has a pKa of about 6.8. This is suitable for effective buffering at physiological pH. • Haemoglobin is quantitatively about 6 times more important then the plasma proteins as it is present in about twice the concentration and contains about three times the number of histidine residues per molecule. For example if blood pH changed from 7.5 to 6.5, haemoglobin would buffer 27.5 mmol/l of H+ and total plasma protein buffering would account for only 4.2 mmol/l of H+. The acid-base buffering systems of the body. The two buffer systems are in dynamic equilibrium with the same hydrogen ion concentration (pH), so that a change induced in the concentration of any one factor in either buffer system rapidly affects the other system and a new hydrogen ion concentration in the blood is established. 1. The lungs assist in maintaining a constant blood pH by removing CO2, 2. while the kidney excretes acid in the form of H2PO4- and NH4 and alkali in the form of HCO3-. Respiratory Buffer Systems • The respiratory system regulation of acidbase balance is a physiological buffering system • There is a reversible equilibrium between: – Dissolved carbon dioxide and water – Carbonic acid and the hydrogen and bicarbonate ions CO2+ H2O ↔H2CO3↔H++ HCO3¯ Respiratory Buffer Systems • CO2 is produced by cellular respiration. • CO2 is converted to bicarbonate by carbonic anhydrase. • results in LOWER pH in respiring tissues. • CO2 is exhaled in lungs. Haemoglobin binds both CO2 and H+ and so is a powerful buffer. Deoxygenated haemoglobin has the strongest affinity for both CO2 and H+; thus, its buffering effect is strongest in the tissues. Little CO2 is produced in red cells and so the CO2 produced by the tissues passes easily into the cell down a concentration gradient. Carbon dioxide then either combines directly with haemoglobin or combines with water to form carbonic acid. The CO2 that binds directly with haemoglobin combines reversibly with terminal amine groups on the haemoglobin molecule to form carbaminohaemoglobin. In the lungs the CO2 is released and passes down its concentration gradient into the alveoli. Respiratory Acidosis and Alkalosis • Result from failure of the respiratory system to balance pH • PCO2is the single most important indicator of respiratory inadequacy PCO2 levels • Normal PCO2 fluctuates between 35 and 45 mm Hg • Values above 45 mm Hg signal respiratory acidosi • Values below 35 mm Hg indicate respiratory alkalosis Respiratory Acidosis • • Respiratory acidosis is the most common cause of acid-base imbalance Occurs when a person breathes shallowly, or gas exchange is hampered by diseases such as pneumonia, cystic fibrosis, or emphysema Respiratory alkalosis • A common result of hyperventilation • Excessive loss of CO2 & subsequent loss of carbonic acid • Caused by hyperventillation:too much CO2 lost (↓carbonic acid and H+ ions) • Anxiety, high altitudes (low O2 levels), musicians, • Symptoms:lightheadedness, agitation, dizziness, Metabolic Acidosis • All pH imbalances except those caused by abnormal blood carbon dioxide levels • Metabolic acidosis is the second most common cause of acid-base imbalance • Typical causes are ingestion of too much alcohol and excessive loss of bicarbonate ions • Other causes include accumulation of lactic acid, shock, ketosis in diabetic crisis, starvation, vomiting, and kidney failure Metabolic Alkalosis • • Rising blood pH and bicarbonate levels indicate metabolic alkalosis Typical causes are: – Vomiting of the acid contents of the stomach – Intake of excess base (e.g., from antacids) – Constipation, in which excessive bicarbonate is reabsorbed Clinical Application Acid-Base Imbalances If the pH of arterial blood drops to 6.8 or rises to 8.0 for more than a few hours, the person usually cannot survive acidosis versus alkalosis factors that lead to respiratory acidosis Clinical Application Metabolic acidosis Respiratory alkalosis Metabolic alkalosis Renal Mechanisms of Acid-Base Balance Chemical buffers can tie up excess acids or bases, but they cannot eliminate them from the body – The lungs can eliminate carbonic acid by eliminating carbon dioxide – Only the kidneys can rid the body of metabolic acids (phosphoric, uric, and lactic acids and ketones) and prevent metabolic acidosis – The ultimate acid-base regulatory organs are the kidneys Kidney buffering power Whatever the nature of the disturbance, the response of the kidney leads to the formation and extraction from the plasma of a fluid with an excess or a deficit of acid. The primary result is a return of the H ion concentration of the blood toward the normal level. Renal Mechanisms of Acid-Base Balance • The most important renal mechanisms for regulating acid-base balance are: – Conserving (reabsorbing) or generating new bicarbonate ions: decreases acidity of ECF – Excreting bicarbonate ions: increases acidity of ECF – Excreting excess H+ Respiratory and Renal Compensations • Acid-base imbalance due to inadequacy of a one system is compensated for by the other system • The respiratory system will attempt to correct metabolic acid-base imbalances • The kidneys will work to correct imbalances caused by respiratory disease Respiratory Compensation • In metabolic acidosis: – The rate and depth of breathing are elevated Blood pH is below 7.35 and bicarbonate level is low – As carbon dioxide is eliminated by the respiratory system, PCO2falls below normal • In respiratory acidosis, the respiratory rate is often depressedand is the immediate cause of the acidosis • In metabolic alkalosis: – Compensation exhibits slow, shallow breathing, allowing carbon dioxide to accumulate in the blood – Correction is revealed by:High pH (over 7.45) and elevated bicarbonate ion levelsRising PCO2 Clinical Applications • Homeostatic mechanisms slow down with age – Elders may be unresponsive to thirst clues and are at risk of dehydration – The very young and the very old are the most frequent victims of fluid, acid-base, and electrolyte imbalances Buffering power of saliva 1.The pH of the mouth must be maintained near neutral for normal tooth maintenance. 2.Oral pH is buffered to a small extent by saliva proteins and phosphate.The major influence on saliva pH is bicarbonate ion which is a by-product of cell metabolism 3.Bicarbonate concentration increases in saliva as the flow rate rises and is due to the increased metabolic rate. This, in turn, raises the pH (more alkaline) of saliva. 4.Bicarbonate ions diffuse into dental plaque and neutralise acid produced by plaque bacteria when carbohydrate is fermented. 5.This reaction is driven by a unique type of carbonic anhydrase which is secreted into saliva by serous acinar cells of the parotid and submandibular glands. 6.Bicarbonate ions maintain the pH of saliva above 6.3. Role of Bone Buffering • Bone consists of matrix within which specialised cells are dispersed. The matrix is composed of organic [collagen and other proteins in ground substance] and inorganic [hydroxyapatite crystals: general formula Ca10(PO4)6(OH)2] components. • The hydroxyapatite crystals make up two-thirds of the total bone volume but they are extremely small and consequently have a huge total surface area. The crystals contain a large amount of carbonate (CO3-2) as this anion can be substituted for both phosphate and hydroxyl in the apatite crystals. Bone is the major CO2 reservoir in the body and contains carbonate and bicarbonate equivalent to 5 moles of CO2 out of a total body CO2 store of 6 moles. (Compare this with the basal daily CO2 production of 12 moles/day) • CO2 in bone is in two forms: bicarbonate (HCO3-) and carbonate (CO3-2). The bicarbonate makes up a readily exchangeable pool because it is present in the bone water which makes up the ‘hydration shell’ around each of the hydroxyapatite crystals. The carbonate is present in the crystals and its release requires dissolution of the crystals. This is a much slower process but the amounts of buffer involved are much larger. Role of Bone Buffering • Bone is an important source of buffer in chronic metabolic acidosis (ie renal tubular acidosis & uraemic acidosis) • Bone is probably involved in providing some buffering (mostly by ionic exchange) in most acute acid-base disorders but this has been little studied. • Release of calcium carbonate from bone is the most important buffering mechanism involved in chronic metabolic acidosis. • Loss of bone crystal in uraemic acidosis is multifactorial and acidosis is only a minor factor • BOTH the acidosis and the vitamin D3 changes are responsible for the osteomalacia that occurs with renal tubular acidosis. Ammonia is produced in renal tubular cells by the action of the enzyme glutaminase on the amino acid glutamine. This enzyme functions optimally at a lower (more acidic) than normal pH. Therefore, more ammonia is produced during acidosis improving the buffering capacity of the urine. Ammonia is unionised and so rapidly crosses into the renal tubule down its concentration gradient. The ammonia combines with H+ to form the ammonium ion, which being ionised does not pass back into the tubular cell. The ammonium ion is therefore lost in the urine, along with the hydrogen ion it contains


























































![ACID-BASE BALANCE Acid-base balance means regulation of [H + ]](http://s1.studyres.com/store/data/000604092_1-2059869358395bda26ef8b10d08c9fb9-150x150.png)