* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download PowerPoint
Survey
Document related concepts
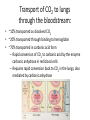
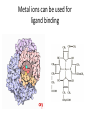
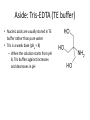
Transcript
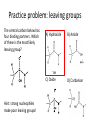
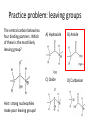
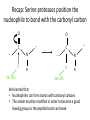
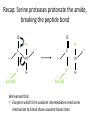
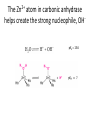
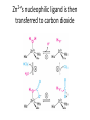
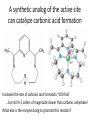
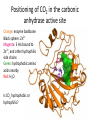
Practice problem: leaving groups The central carbon below has four binding partners. Which of these is the most likely leaving group? A) Hydroxide B) Amide C) Oxide D) Carbanion 2 Hint: strong nucleophiles make poor leaving groups! Practice problem: leaving groups The central carbon below has four binding partners. Which of these is the most likely leaving group? A) Hydroxide B) Amide C) Oxide D) Carbanion 2 Hint: strong nucleophiles make poor leaving groups! Recap: Enzymes increase reaction rates by several major mechanisms 1) 2) 3) 4) Positioning substrates to react with each other Donating or accepting a proton (acid-base catalysis) Positioning a metal ion to react with a substrate Reacting covalently with a substrate, then releasing it On Friday we studied a prime example of #2 and #4: the serine proteases Recap: Serine proteases cleave peptide bonds Peptide bond (to be broken) + H2O Amino acid residue n Amino acid residue n+1 Recap: Serine proteases create a strong nucleophile, RO- We learned: • A nucleophile donates two electrons to form a new bond • How to predict the strength of a nucleophile • How this nucleophile is created from a serine side chain Recap: Serine proteases position the nucleophile to bond with the carbonyl carbon O O… C … N O- Ser 195 … C … N O H H Ser 195 We learned that: • Nucleophiles can form bonds with carbonyl carbons • The amide must be modified in order to become a good leaving group so the peptide bond can break Recap: Serine proteases protonate the amide, breaking the peptide bond O- O H C … N+ O Ser 195 H … C … N O H H Ser 195 We learned that: • Enzymes which form covalent intermediates need some mechanism to break those covalent bonds later … Recap: Enzymes increase reaction rates by several major mechanisms 1) 2) 3) 4) Positioning substrates to react with each other Donating or accepting a proton (acid-base catalysis) Positioning a metal ion to react with a substrate Reacting covalently with a substrate, then releasing it Today, we’ll see an example of #3 – carbonic anhydrase – and learn some widely applicable principles along the way: • pH and osmo-regulation • acid and metal terminology • enzyme kinetics Lecture 55: Mechanisms of Enzyme Catalysis II Carbon dioxide dissolved in water can form carbonic acid This reaction is appreciable without catalysis, but exceptionally fast (diffusion-limited) when catalyzed by carbonic anhydrase. Three major reasons to control this reaction: • Enhance CO2 transport/storage (respiration, photosynthesis) • Regulate pH • Control water balance (eye, kidney) Transport of CO2 to lungs through the bloodstream: • ~10% transported as dissolved CO2 • ~20% transported through binding to hemoglobin Hemoglobin’s N-termini are nucleophilic & can bind to CO2: Bonus: the structural changes caused by CO2 binding favor release of O2. Transport of CO2 to lungs through the bloodstream: • ~10% transported as dissolved CO2 • ~20% transported through binding to hemoglobin • ~70% transported in carbonic acid form – Rapid conversion of CO2 to carbonic acid by the enzyme carbonic anhydrase in red blood cells – Requires rapid conversion back to CO2 in the lungs, also mediated by carbonic anhydrase Carbonic acid and pH regulation Carbonic acid has two protons to lose: pKa= 6.3 Carbonic acid Bicarbonate pKa= 10.4 Carbonate Bicarbonate can bind an extra H+ when pH is lowered, or release an extra H+ when pH is raised. As a result, it is more difficult to raise or lower the pH of solution containing bicarbonate than pure water. Titration curves exemplify the buffering effect of bicarbonate Start with a solution of carbonic acid at low pH, then record pH as a base is added pKa #2 pKa #1 Carbonic acid forms faster in basic solution kf Can you explain why? Carbonic acid forms faster in basic solution kf OH- is a stronger nucleophile than H2O. Carbonic anhydrases produce the strong nucleophile OH- in their active site Carbonic anhydrases are not all homologous (i.e., they are not related by descent from a single gene in a common ancestor) • Evolved at least three times independently Carbonic anhydrases produce the strong nucleophile OH- in their active site Carbonic anhydrases are not all homologous (i.e., they are not related by descent from a single gene in a common ancestor) • Evolved at least three times independently • One feature in common: they all bind a zinc ion (Zn2+) which coordinates the nucleophile, OH- Zinc ion binding by histidines/cysteines can also be used for stabilization “Zinc finger” motif Many transcription factors contain zinc fingers Metal ions can be used for ligand binding Many enzymes use metal ions as cofactors • A cofactor is a non-peptide molecule required for an enzyme’s activity • Metal ions are one type of cofactor; organic vitamins are another major class • Roughly one-third of enzymes contain tightly bound metals or require the presence of metals for their activity Some terminology: what is a metal? To an astronomer: For our purposes: Metal Metalloid Non-Metal Some terminology: what is a metal? Metals are: • Shiny • Good thermal/electrical conductors • Able to form alloys with other metals • Able to form basic oxides, e.g. Metal Metalloid Non-Metal Some terminology: what is an acid? Arrhenius definition: • When added to water, acids increase [H+] and bases increase [OH-] • An acid-base reaction produces a salt and H2O Acid Base Salt Brønsted-Lowry definition: • Acids donate protons, and bases accept them • An acid-base reaction doesn’t necessarily produce water Acid Base Conjugate Conjugate base acid Some terminology: what is an acid? Arrhenius definition: • When added to water, acids increase [H+] and bases increase [OH-] Brønsted-Lowry definition: • Acids donate protons, and bases accept them Lewis definition: • Acids accept electron pairs, and bases donate electron pairs Acid Base • Note that all nucleophiles are Lewis bases: they donate a pair of electrons when they react. Metals form coordination complexes through Lewis acid-base reactions • The metal atom (often a cation) is the Lewis acid, and is called the coordination center • Other molecules bound to the metal atom are Lewis bases, and are called the ligands • A ligand can join the complex if: – The central metal has an empty orbital – The ligand has a free electron pair • Ligands that interact through multiple functional groups are very stably bound: this is called chelation 2+ Aside: Tris-EDTA (TE buffer) • Nucleic acids are usually stored in TE buffer rather than pure water • Tris is a weak base (pKa = 8) – When the solution starts from pH 8, Tris buffers against increases and decreases in pH Aside: Tris-EDTA (TE buffer) • Nucleic acids are usually stored in TE buffer rather than pure water • Tris is a weak base (pKa = 8) – When the solution starts from pH 8, Tris buffers against increases and decreases in pH • EDTA – Chelates free metal ions in your sample – Most enzymes that cut or modify DNA have a coordinated metal ion Why is Zn2+ used so often in metalloenzymes? • Can easily have 4, 5, or 6 ligands – No marked preference for the latter – Won’t simply bind and hold onto ligands forever • Full 3d orbitals • Strong Lewis acid even at neutral pH – Useful for acid-base catalysis • Several side chains make good ligands – Histidines, cysteines, glutamates and aspartates The Zn2+ atom in carbonic anhydrase helps create the strong nucleophile, OHpKa = 15.6 Zn2+’s nucleophilic ligand is then transferred to carbon dioxide A synthetic analog of the active site can catalyze carbonic acid formation Increases the rate of carbonic acid formation ~100-fold …but still 4-5 orders of magnitude slower than carbonic anhydrase! What else is the enzyme doing to promote this reaction? Positioning of CO2 in the carbonic anhydrase active site Orange: enzyme backbone Black sphere: Zn2+ Magenta: 3 His bound to Zn2+, and other hydrophilic side chains Green: hydrophobic amino acids nearby Red: H2O Is CO2 hydrophobic or hydrophilic? Positioning of CO2 in the carbonic anhydrase active site After the reaction takes place, the hydrophobic pocket helps promote exit of carbonic acid (a polar molecule). In general, poor binding of products -> faster enzyme turnover Is this mechanism enough to explain carbonic anhydrase’s reaction rate? • The maximum turnover rate for the enzyme is 106 reactions per second – Every part of the catalysis mechanism must be at least this fast! • The equilibrium constant for proton dissociation from Zn-OH2 is Ka = 10-7 M (because pKa = 7) koff kon Ka = koff / kon Is this mechanism enough to explain carbonic anhydrase’s reaction rate? • The maximum turnover rate for the enzyme is 106 reactions per second – Every part of the catalysis mechanism must be at least this fast! • The equilibrium constant for proton dissociation from Zn-OH2 is Ka = 10-7 M (because pKa = 7) • By definition, Ka = koff / kon, so koff = Kakon • Protons diffuse very rapidly in water, but kon still cannot be more than 1011 M-1 s-1 • Implies max rate of proton dissociation (and thus the reaction) is 104 s-1 The carbonic anhydrase proton shuttle Carbonic anhydrase and fluid transport Ion transport is used to regulate the movement of water • When [solute] rises in one compartment, H2O is driven there by osmosis • Pumping of carbonic anhydrase products can affect [H+] and [HCO3-] enough to raise/lower overall [solute] in a compartment Inhibitors of carbonic anhydrase can be used to treat glaucoma, a buildup of fluid in the eye. Epithelial cell H2O + CO2 C.A. H+ + HCO3H2O Extracellular fluid HCO3- (active) H2O (passive) Sulfonamides: potent carbonic anhydrase inhibitors What we hope you learned from carbonic anhydrase • Metal ions are Lewis acids that can create and position nucleophiles to form new bonds • Diffusion limits on substrate availability can be surpassed using an energetic binding “funnel,” e.g. the proton shuttle • Ion production can be used for pH buffering and osmoregulation