* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download ORGANIC CHEMISTRY NOTES , 2s , 2px , 2py , 2pz , 2s , 2px , 2py
Electron transport chain wikipedia , lookup
Microbial metabolism wikipedia , lookup
Isotopic labeling wikipedia , lookup
Radical (chemistry) wikipedia , lookup
Photosynthesis wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Biosynthesis wikipedia , lookup
Biochemistry wikipedia , lookup
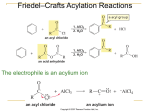
www.fahadsacademy.com and 2py change shape and form 3 sigma bonds as shown in the diagram. Where as the 2pz ORGANIC CHEMISTRY NOTES orbital remains unchanged. These half-filled 2pz orbitals overlap sideways and form a ring of delocalized electrons above and below the Interpret and Use the Nomenclature, General hexagonal structure as shown. Formula, Displayed Formula of the following Arenes HalagenoArenes Phenols AcylChlorides Amides and Amino Acids ARENES Arenes are derivatives of Benzene which are Cyclic unsaturated hydrocarbons . Physical Properties Arenes have relatively high melting/boiling points due to greater van der waals dispersion forces due to delocalized electrons Insoluble in water IUPAC Priority Rules Electronic configuration of Carbon in ground 2 2 1 1 0 2 1 1 1 1 state 1s , 2s , 2px , 2py , 2pz . Carbon only has 2 half-filled outermost orbitals for bonding. By promoting one electron from 2s orbital to 2pz orbital and going into excited state 1s , 2s , 2px , 2py , 2pz it can now form 4 bonds which is more exothermic and leads to more stability. 1- Carboxylic Acids and Derivatives (esters, acylchlorides, amides) 2- Nitriles (prefix: cyano, suffix: nitrile) 3- Aldehyde(suffix: -al) 4- Ketone (suffix: -one) 5- Alcohols (prefix: hydroxyl, suffix:-ol) 6- Ammines (prefix:amino, suffix:amines) 7Naming Aromatic Hydrocarbons which are derivatives of Alkenes Multiple substituents should be named by giving the smallest number to each substituent in either direction of the ring Carbon undergoes sp2 hybridization in benzene, where 3 half-filled outermost orbitals 2s, 2px, www.fahadsacademy.com Bromine and Chlorine are not electronegative enough to attack benzene. FeCl3 and FeBr3 form metal complexes with Cl2 and Br2. The lone pair of Cl2/Br2 is then used for dative bonding with Fe. This increases electronegativity of Cl2/Br2. Benzene group as a side chain is known as Phenyl. Halagen Substituents Florine, Chlorine, Bromine, Iodine have exactly the same priority as alkyl chains. Floro-, Chloro-, Bromo-, Iodo- prefixes are used. Alkyl chains and halogen substituents are written in alphabetical order. 2-4-6 Directing Groups and 3-5 Directing Groups FeBr3 and AlCl3 act as Lewis acids (electron pair acceptor) -CH3, -OH, -NH2 are 2-4-6 directing Nucleophilic Substitution Reaction in Methyl Benzene and Chlorine They increase electron cloud density on the 2nd, 4th and 6th C atom Boiling MethylBenzene or Benzene with Cl2 in the absence of catalyst and presence of UV light -NO2, -COOH are 3-5 directing The N and C atom are attached to an electronegative O atom which pull electrons away from the benzene cloud. Electrophilic Substitution Reaction of Benzene and Methyl Benzene with Chlorine/Bromine Benzene is highly attracted to electrophiles due to delocalized electrons but the delocalized electron cloud has extra stability and the reaction is more difficult compared to alkenes. Catalysts are therefore required for this reaction. Nitration of Benzene (electrophilic substitution) To substitute NO2 in the Benzene ring, you would need NO2+1 (nitronium ion). HNO3 is mixed with H2SO4 to get this electrophile. HNO3 + 2 H2SO4 NO2+1 + HSO4-1 www.fahadsacademy.com benzene ring to provide extra strength to the + H3O+1 bond. Nucleophilic substitution (like those in halogenoalkanes) only occurs under very severe condition. Physical properties: They are liquids and insoluble in water. PHENOLS Position of Substituent in the product Nitro substituent is 3-5 directing Methyl substituent is 2-4 directing Phenols are way more acidic than alcohols, and its delocalized ring is more reactive than benzene. The Lone pair on the oxygen atom, starts to overlap with the delocalized electron cloud on the benzene. The makes the oxygen-benzene bond even stronger and more stable. Hence nucleophilic substitution, similar to aliphatic alcohols is not possible in Phenol It also reduces the strength of the O-H bond which make it easier for H+1 to ionize. Oxidation of Arenes ARYL HALIDES Chloro benzene is highly unreactive. The extra lone pairs on the Cl atom overlap with the delocalized electrons in the The phenoxide ion formed above, when phenol loses an H+1 is very stable as the negative charge on “O” gets delocalized over the entire benzene ring. The extra stability of the phenoxide ion shifts the equilibrium towards right and makes phenol more acidic -OH group is 2-4-6 directing www.fahadsacademy.com a) Reactions with Dilute Nitric Acid at Phenols are stronger acids compared to R.T.P gives 2-nitrophenol and 4aliphatic alcohols nitrophenol (Phenols>water>aliphatic alcohols) Phenols Neutralize Strong Bases Due to their weak acidity. b) Reactions with Concentration Nitric Acid leads to more than one electrophilic substitutions due to activated benzene ring Addition of a strong acid to sodium phenoxide recovers phenol c) Aqueous Bromine gets decolorized at room temperature when mixed with aqueous Phenol. White precipitate of 2,4,6-tribromophenol is produced. Reactions with Sodium Phenol reacts vigorously with Sodium, greater reactivity compared to reactions with Aliphatic alcohols. IodoForm Reaction and Identification of CH3CHOH- Group or CH3CO- Group Reagents: Iodine mixed in NaOH and gentle or no heat Electrophilic Substitution Reactions for Phenol (Nitration and Bromination of Phenol) Delocalized Electron cloud in Phenols is way more active compared to benzene due to increased electron charge density. www.fahadsacademy.com A pale yellow precipitate of tri-iodo methane, –COOH group would therefore make the acid CHI3, is obtained which gives evidence of the even less acidic, and would not favor presence of CH3CHOH- group dissociation. In the above diagram, Notice the formation of an Aldehyde in Step (2). The “H” present in (2) can also be an Alkyl Chain. The same test would also be useful in identifying CH3CO- group as aldehydes/ketones are already being produced as intermediates in the reaction with alcohols. If an “H” atom in the alkyl chain is substituted by an electronegative specie like “Cl” then a negative inductive effect would take place which would pull the electron density away from the –COO-1 atoms and making the ion more stable ACYL CHLORIDES Strength of Organic Acids CH3ClCOOH ↔ CH3ClCOO-1 + H+1 (higher pKa value means lower dissociation/weaker acid) CH3COOH ↔ CH3COO-1 + H+1 pKa=2.86 Hence a stronger acid is formed when compared its counterpart CH3COOH. pKa = 3.75 CH3CH2COOH ↔ CH3CH2COO-1 + H+1 pKa=4.76 The strength of an acid depends on the stability of the ethanoate ion R-COO-1. When –COOH dissociates, the negative charge on –COO-1 spreads over the two oxygen atoms. More negative charge on these two oxygen atoms makes this ion unstable and this favors the reverse reaction making the acid less acidic. Alky groups have a positive inductive effect and increase the electron density on the two oxygen atoms which increases the negative charge on these atoms. Bigger alkyl chains attached to the FORMATION OF ACYL CHLORIDES Carboxylic acids undergo nucleophilic substitution reactions to produce the corresponding acyl chloride. PCl3 and SOCl2 are heated with carboxylic acid: 3 RCOOH + PCl3 3 RCOCl + H3PO4 RCOOH + SOCl2 RCOCl + SO2 + HCl www.fahadsacademy.com HYDROLYSIS OF ACYL CHLORIDES Acyl chlorides are more susceptible to nucleophilic attacks due to the presences of partial positive C atom which is bonded to “O” and “Cl” which are extremely electronegative. This was not the case in –COOH, where the attached OH group had an electron pushing effect, unlike Cl. React vigorously with water Carbon in Acyl Chlorides is bonded to two electronegative elements, which gives it a large partial positive charge and susceptible to nucleophilic attack. Carbon in Alkyl Chlorides (or halagenoalkanes) is also partial positive but the charge is not as high compared to acyl chlorides. Carbon in Aryl Chlorides is surrounded by delocalized electrons which hide the partial positive charge making it the least susceptible to a nucleophilic attack React vigorously with Alcohols to form Esters Reaction mechanism given below(not required) Reaction mechanism given below (not required) Ease of Hydrolysis of Acyl Chlorides, Aryl Chlorides, Alkyl Chlorides Acyl Chloride > Alkyl Chloride > Aryl Chloride www.fahadsacademy.com React Readily with Phenols on Warming Basic Nature of Ammines Phenyl ethanoate is formed in the above reaction. Phenol is dissolved in NaOH (aq) and warmed with ethanoyl chloride. Nitrogen has one lone pair which can accept a proton and form a dative bond. Reaction with Ammonia and Primary Ammines Another example of a vigorous nucleophilic substitution Excess ammonia produced reacts with HCl, which produces NH4Cl Primary Ammines react in a similar manner; The ability of the nitrogen atom to accept a proton depends on the strength of the negative charge on the lone pair i) ii) NITROGEN COMPOUNDS Formation of Ethyl Ammine and Phenyl Ammine through reduction Where [H] is a reducing agent LiAlH4 (lithium tetra hydrido borate). or Where nitrile is heated with H2 gas and a nickel or palladium catalyst. Primary ammines are most basic as alkyl groups have an electron pushing effect (positive inductive effect) which increases the charge on Nitrogen atom’s lone pair Phenyl ammine is the least basic as the lone pair gets delocalized over the entire benzene ring and then negative charge gets spread out and less intense, which therefore doesn’t accept H+1 that easily. www.fahadsacademy.com First step: Dissolve Phenol in NaOH Reaction of Phenyl Ammine with Br2(aq) Ammine is 2,4,6 directing and increases the negative charge on the benzene ring, activating it and making it susceptible to electrophilic attack Then add the mixture to a diazonium salt, reaction takes place < 10C. A bright yellow precipitate is obtained in the following reaction. (diazonium ion acts as an electrophile) The “–N=N -“ group in the compound is known as the Azo group. This is one method of producing very intense and stable dyes which do not fade easily. Other examples of Azo Dyes: Methyl Orange: Bromine water gets decolorized and white ppt of 2,4,6-tribromophenylammine is obtained Phenylamine and Diazonium Salt Ionic Eq: Temperature has to be kept below 10 C. Reagents: Phenyl amine and HCl, below 10 C are mixed with NaNO3. AMINO ACIDS as ZWITTER IONS Formation of Azodyes An Amino Acid: www.fahadsacademy.com Proteins are one form of Polypeptides. The presence of N-H in polypeptides causes hydrogen bonds to be formed. Hydrolysis of Proteins A zwitter ion has no overall electrical charge but contains separate parts which are positively and negatively charged. Adding an Alkali: Proteins are heated with 6 mol/dm3 Hydrogen for 24 hours at 1100C. Adding an Acid: Formation of Peptides from Amino Acids Amides have this link (Peptide Linkage) Polypeptides will have multiple amino acids joined together through condensation polymerization