* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download What about Artificial Organs?
Cell culture wikipedia , lookup
Embryonic stem cell wikipedia , lookup
Cell theory wikipedia , lookup
Somatic cell nuclear transfer wikipedia , lookup
Stem-cell therapy wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Hematopoietic stem cell transplantation wikipedia , lookup
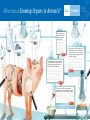
What about Artificial Organs? Path to the future... Mechanical Kidney Man-made Devices Researchers from the Kidney Project are creating a bioartificial kidney as a permanent solution to end stage renal disease. The implantable bioartificial kidney builds upon the existing extracorporeal Renal Assist Device (RAD), which is a bioartificial kidney that combines a membrane hemofilter and a bioreactor of human renal tubule cells to mimic many of the metabolic, endocrine, and immunological functions of a healthy kidney. The ultimate goal of The Kidney Project is to apply microelectromechanical systems (MEMS) and nanotechnology to miniaturize the extracorporeal RAD into a surgically implantable, self-monitoring, and self-regulating bioartificial kidney. For many years, reseachers have manufactured devices that are used to diagnose, prevent, or treat disease or other conditions in humans. Cochlear implants, pacemakers, ventricular assist devices, dialysis machines and even prosthetics, carried inside and outside the human body, have enabled major improvements in quality of life and even prevented imminent death while awaiting a transplant. Artificial Organs are specifically engineered from polymers, glasses and ceramics, nano-technology, metals, alloys and composites or a combination. These man made devices are to be implanted or integrated into a human body to replace a dysfunctional full organ. Source One solution for the shortage of transplantable organs is creating artificial ones that last. Already, researchers are developing bioartificial organs that can keep patients with serious organ failure alive and functioning for years. For now, the goal is to keep patients alive until they can receive a real organ, but one day, patients may be able to live for long periods of time with artificial hearts or kidneys. What if we could completely eliminate organ transplantation by deploying articifial hearts and kidneys which gift a full lifespan to the recipient? Source Source Total Artificial Heart For patients suffering from end-stage biventricular heart failure in which both ventricles can no longer pump enough blood for a person to survive, a donor heart transplant is the standard of care. However, not all patients waiting for a donor heart will have one available to them when they need it to save their lives. In the United States, approximately 16% of transplant-eligible patients on the list die or become too sick for a transplant while waiting for a donor heart. A manufactured full-functional artificial heart replaces both failing ventricles and all four heart valves. This artificial heart is deployed as either a bridge to a matching donor heart transplant or for permanent use for patiens who are not transplant-eligible, also known as destination therapy. Source HQ/TPTP/15/0010j Source What about Bioengineering Organ Preservation? Organs? Path to the future... Organ engineering is a new strategy to cope with the shortage of donor organs. The bioengineered organ will not be rejected and there will be no need for life-long immunosuppressant therapies. The scaffold material does not contribute to the rejection, and the cells that are used to repopulate the organ scaffold belong to the patient. Patients no longer face a lifetime filled with the need to take immunosuppressant drugs to prevent organ rejection, and will retain overall immune competence. The concept is based upon the isolation of a three-dimensional, biological scaffold material, which has no cells in it. This scaffold from explanted organs is prepared by removing all cellular components (decellularization) by washing it with a special detergent-like mixture. This acellular scaffold retains the native organ ultrastructure and can be seeded (repopulated)to generate a functional organ in vitro for transplantation. Source Source The patient’s own cells are converted into stem cells - those super-adaptable “pluripotent” cells that can transform into virtually any other kind of cell. The researchers take a patient’s cells, such as a skin cell or a muscle cell, and convert it into a stem cell that can be driven along a particular line of differentiation. This way, a stem cell can be coaxed into becoming a liver cell, or a kidney cell, a heart cell, or whatever is required. These cells are used to seed the 3D protein scaffold to grow a new organ. It is pivotal to also grow the appropriate blood vessels to provide the organ with oxygen and nutrients. Source Heart, liver, lung and kidneys. In recent years, organs such as heart, liver, lung and kidneys, have been reported to provide acellular extracellular matrix (ECM)-based scaffolds and demonstrated the potential of recellularization with selected cell populations, particularly with stem cells. The current scientific need for further studies, concerning the source of donor organs, optimization of the decellularization process, the cell type for the reseeding process and the establishment of the optimal conditions for the repopulation of the scaffold is still tremendous. Source HQ/TPTP/15/0010j What about Organ Preservation? Path to the future... Organ preservation is used to maintain an organ’s viability outside the human body, allowing more time for doctors to assess its condition prior to transplantation. Doctors are able to assess the quality of the organ and investigate its anatomy to confirm whether or not an organ meets the criteria to be transplanted. Source Source Will perfusion technology ultimately ensure that all donated organs are used? Dr. Shaf Keshavjee, a thoracic surgeon and director of the lung transplant program at Toronto General Hospital, who successfully developed a lung perfusion machine says: “In a donation after cardiac death, only 2% of lungs are typically used.” “I think this could easily be moved to 50%. We can re-use many of the lungs we don’t use today. Now we can see we are not only going to preserve an organ, but we are going to make it better. I think that this strategy is applicable to all organs, but they have specific needs. I think mistakes in the past have been assuming all organs need the same thing. So systems need to be developed to meet the needs of each organ.” Source Organ Perfusion Systems will keep organs alive outside the body. Instead of cooling the organ, which slows down the process of death, Organ Perfusion Systems keep the organs at normal body temperature to allow recovery from the trauma associated with donation or as a result of chronic disease. The Perfusion System feeds the organ with nutrients, removes waste products, provides oxygen and removes carbon dioxide according to the specific organ’s needs. Source Source The machine not only keeps the donor organ alive, it may even enhance the organ by improving its condition. By keeping the organ alive outside the body for a couple of days, we will be able to diagnose infections or abnormalities, and drugs can be delivered when needed. Recovery of the organ begins before the actual transplant starts. Source HQ/TPTP/15/0010j What about Printing Organs? Path to the future... 3-D Printing also allows other body parts and tissues to be generated such as muscle, bone and outer-tissue body parts like ears. We are still many years away from 3-D Printing on a routine basis, but there is hope that organ printing could one day supplement the shortage of live organs. Skin, blood cells, heart valves, blood vessels and bladder are under investigation and there are high hopes that one day we will be printing complete organs like kidney, heart and liver. All would be printed with the patient’s own cells, so the organ works for life. Source Researchers create a blueprint from a patient’s organ by computerising a CT scan of the particular organ. All data from the CT scan is used to build a 3-D reconstruction of both the inside and outside of the organ, including its blood supply vessels. Dr. Anthony Atala, a practising surgeon and director of the Wake Forest Institute for Regenerative Medicine:“This model is used to guide the printer as it layer-by-layer prints a three-dimensional structure made up of cells and the biomaterials to hold the cells together.” Source An inkjet made my bladder. Printers are modified in such way that the printer sprays cells instead of ink. By spraying layers of cells in a biodegradable mould, both tissues and organs can be printed for safe and effective long term use. Future developments will allow 3-D printing directly onto the patient, for instance by scanning a wound. Source HQ/TPTP/15/0010j Will 3-D printing of replacement tissues and organs finally put an end to organ shortages? Regenerative Medicine is investigating how to replace old and poorly-functioning tissues and organs with new and healthy tissues and organs originating from the patient’s own cells. Some cell types like skin or blood cells are easily obtained from the patient’s as they are constantly growing. Other cell types need to be grown from stem cells recovered from the patient’s bone marrow. Stem cells may also be derived from amniotic fluid, placenta or umbilical cord. Source Source What about Growing Organs in Animals? Path to the future... Will growing organs in animals provide a plentiful supply of donor organs? An exciting vision for the future would be realised if Organ Engineering became a substitute for transplantation, overcoming problems such as organ donor shortages thus reducing or obviating the need for immunosuppressive therapy. Source When fully grown, the adult host animal is put down, and the human organ will be harvested and transplanted into the human patient with organ failure. Using a patient’s own stem cells could help to reduce the risk of the transplanted organ being rejected while also providing a plentiful supply of donor organs. Professor Chris Mason, chair of regenerative medicine at University College London, said: “For something like a kidney transplant where it is not urgent, it would be highly attractive to be able to take cells from a patient, grow them in this way and deliver a personalised kidney.” Researchers injected stem cells from rats into the embryos of mice that had been genetically altered so they could not produce their own organs, thereby creating mice with rat organs. Professor Hiromitsu Nakauchi, director of the centre for stem cell biology and regenerative medicine at the University of Tokyo in Japan: “The technique, called blastocyst complementation, provides us with a novel approach for organ supply. We have successfully tried it between mice and rats. We are now rather confident in generating functional human organs using this approach.” Source Source This technique could be used to introduce human stem cells into the embryo of an animal, most likely a pig, to create a chimeric embryo. By using a patient’s own stem cells it will help to reduce organ rejection. This chimeric embryo will be implanted into an animal’s womb where it will grow into a ‘normal’ animal. As the host animal matures into an adult, the human stem cells will grow to become a perfect human organ such as a pancreas, kidney or a heart. Source HQ/TPTP/15/0010j What about Self Artificial Monitoring? Organs? Path to the future... Dose Concentration versus Drug Response By improving the analytical methods and devices to measure the low concentrations of immunosuppressants in biological fluids, organ recipients can very easily monitor their individual drug levels at home. Measurement of the low drug concentrations offers the opportunity to reduce patient variability using real concentrations in the body rather than by dose alone. The dose of the drug can then be customized to the patient to maximise therapeutic effect while minimising the risk of irreversible kidney damage when given in too high a dose. Source The dose administered does not directly correlate to the available drug concentration in the body, for instance due to variability in absorption or metabolism of the drug. Therefore, a poor relationship exists between dose concentration and drug response. Because of this poor relationship, immunosuppressant drugs have significant inter individual variability. Even within the same one patient, drug concentration may vary, for example due to drug-nutrient interaction, renal insufficiency or inflammation. Source As some drugs have a narrow therapeutic index, the difference between therapeutic benefit and toxicity is small. Too much drug is associated with toxicity and too little with organ rejection. Source Easy Self Monitoring could help to increase patient survival. As the number of immunosupressant drugs will increase in the future, Self Monitoring will tailor immunosuppression to the specific characteristics of the individual patient changing dose and drugs as time progresses and conditions change. Source What if we could obtain the optimum balance between therapeutic efficacy and adverse and toxic events by Self Monitoring? How about that? Source HQ/TPTP/15/0010j