* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download The Development of a New Atomic Model
Quantum electrodynamics wikipedia , lookup
Atomic orbital wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Bremsstrahlung wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Hydrogen atom wikipedia , lookup
Electron configuration wikipedia , lookup
Planck's law wikipedia , lookup
Tight binding wikipedia , lookup
Particle in a box wikipedia , lookup
Magnetic circular dichroism wikipedia , lookup
Ultraviolet–visible spectroscopy wikipedia , lookup
Ultrafast laser spectroscopy wikipedia , lookup
Elementary particle wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Double-slit experiment wikipedia , lookup
Astronomical spectroscopy wikipedia , lookup
Atomic theory wikipedia , lookup
Matter wave wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
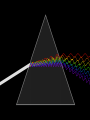
The Development of a New Atomic Model Rutherford- did not explain how the atom’s negatively charged particles are distributed in the space surrounding it’s positively charged nucleus. The New Atomic Model • Researcher used investigations into the absorption and emission of light • Studies revealed a relationship beteen light an atom’s electrons • New understanding about the nature of energy, matter, and atomic structure. Properties of Light • Before 1900, light was thought to behave solely as a wave – Wavelike description • It is type of electromagnetic radiation • Forms part of the electromagnetic spectrum • Significant wavelike motion characterized by wavelength and frequency – Wavelength (λ) is the distance between corresponding points on adjacent waves. – Frequency (v) is the number of waves that pass a specific point in a given time, usually one minute. Frequency and wavelength are mathematically related to each other • c=λv c is the speed of light, a constant • There for λ, wavelength in m, is inversely proportional to v, frequency in s-1. • As wavelength decreases frequency increases and vice versa The Photoelectric Effect • The photoelectric effect refers to the emission of electron when light is shines on a metal. The Photoelectric Effect • The mystery was….. • No electron would be emmited if the lights frequency (v) was below a certain minimum. • No matter how intense (W) the light was. • Wave theory state that light of any frequency could supply enough energy to eject an electron. The Particle Description of Light • Max Planck (1900 German Physicist) – Studied emission of light by hot objects – He proposed the hot objects do not emit electromagnetic energy continuously (as would be expected if all we were dealing with was a wave) The Particle Description of Light • Quantum- a measure of energy that is the minimum quantity of energy that can be lost or gain by an atom • Planck proposed a relationship between a quantum of energy and the frequency of radiation • E=hv • E= energy in joules, v is frequency (s-1), • h= a fundamental physical constant now know as Planck’s constant = 6.626x 10 -34 The Duality of particles and waves • Albert Einstein expanded on Planck’s work and proposed a radical idea • Electromagnetic radiation has a dual waveparticle nature • While like light act like a wave • It is a stream of particles • Each particle carries a quantum of energy • These particles are called photons – These particles have zero mass but carry a quantum of energy This explained the photoelectric effect • Einstein explained – Electromagnetic radiation is only absorbed by matter in whole numbers – In order for an electron to be rejected from the surface of the metal it must be struck by a single photon possessing at least a minimum energy required to knock the electron off. – Every metal has a different frequency Home work • Pages 124-125 – #1-25 – Complete sentences – If it is a multiple choice problem, select the letter choice them write a true complete sence out of the question and the choice: DO NOT COPY THE QUESTIONS – If it is a calculation, show equation used and ALL work – Have a great Thanksgiving!!!