* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download ETHYLENE
DNA vaccination wikipedia , lookup
Designer baby wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
Polycomb Group Proteins and Cancer wikipedia , lookup
Epigenetics of human development wikipedia , lookup
History of genetic engineering wikipedia , lookup
Gene expression profiling wikipedia , lookup
Mir-92 microRNA precursor family wikipedia , lookup
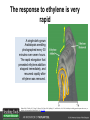
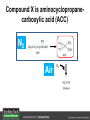
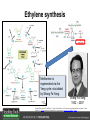
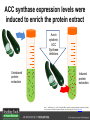
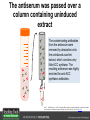
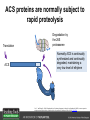
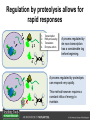
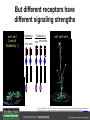
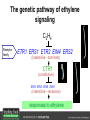
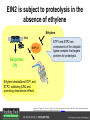
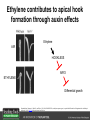
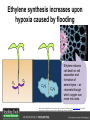
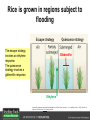
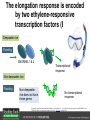
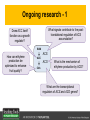
Ethylene Cover image from Science Vol. 241, no. 4869, 26 August 1988, reprinted with permission from AAAS; photo by Kurt Stepnitz, Michigan State University Ethylene (C2H4) is a gaseous hormone with diverse actions Air (control) 7 days ethylene Ethylene regulates: •fruit ripening •organ expansion •senescence •gene expression •stress responses Air Ethylene Cotton plants Arabidopsis Beyer, Jr., E.M. (1976) A potent inhibitor of ethylene action in plants. Plant Physiol. 58: 268-271. Early fruit-ripening practices Ethylene in smoke has long been used to ripen fruit; this practice has included ripening pears in the smoke from incense. Gashing of unpollinated figs has also been practiced; the ethylene produced upon wounding induces ripening. Image sources: British Museum; Kurt Stüber Ethylene responses in Arabidopsis Ethylene-induced gene expression Inhibition of root elongation Inhibition of leaf cell expansion Acceleration of leaf senescence Lorenzo, O., Piqueras, R., Sanchez-Serrano, J.J., and Solano, R. (2003). ETHYLENE RESPONSE FACTOR1 integrates signals from ethylene and jasmonate pathways in plant defense. Plant Cell 15: 165-178; Rüžička, K., Ljung, K., Vanneste, S., Podhorská, R., Beeckman, T., Friml, J., and Benková, E. (2007). Ethylene regulates root growth through effects on auxin biosynthesis and transport-dependent auxin eistribution. Plant Cell 19: 2197-2212. When germinating in the dark, impeded seedlings produce ethylene which confers a characteristic “triple response” Ethylene induces the triple response: •reduced elongation, •hypocotyl swelling, •apical hook exaggeration. C2H4 C2H4 It’s thought that this response helps the seedling push past the impediment. By treating dark-grown seedlings with exogenous ethylene, ethylene-response mutants could be identified quickly and easily based on the triple response phenotype. The response to ethylene is very rapid A single dark-grown Arabidopsis seedling photographed every 30 minutes over seven hours. The rapid elongation that preceded ethylene addition stopped immediately, and resumed rapidly after ethylene was removed. Binder, B.M., O’Malley, R.C., Wang, W., Moore, J.M., Parks, B.M., Spalding, E.P., and Bleecker, A.B. (2004). Arabidopsis seedling growth response and recovery to ethylene. A kinetic analysis. Plant Physiol. 136: 2913–2920. Ethylene synthesis and homeostasis In 1901, ethylene was identified as a compound that affects plant growth Increasing ethylene Illuminating gas distilled from tar contains very high levels of ethylene. In 1901, Dimitry Neljubow traced the source of the strange growth patterns of his dark-grown pea seedlings to the ethylene produced by gas-burning lamps. Neljubov, D.N. (1901) Uber die horizontale Nutation der Stengel von Pisum sativum und einiger anderen Pflanzen. Beih. Bot. Centralbh. 10: 129–139. In 1934, Gane purified ethylene from ripening apples, demonstrating that it is an endogenous hormone How to measure ethylene circa 1943 The relative insensitivity of the early methods made it difficult to detect small changes in ethylene production. Avocados were an early model for studying fruit ripening Pratt, H.K., Young, R.E., and Biale, J.B. (1948). The identification of ethylene as a volatile product of ripening avocados. Plant Physiol. 23: 526-531. In 1959 gas chromatography (GC) was used to measure ethylene levels This new method was a million-fold more sensitive than earlier methods. Using GC, Burg and Thimann showed that ethylene production is temperature dependent. Burg, S.P., and Thimann, K.V. (1959). The physiology of ethylene formation in apples. Proc. Natl. Acad. Sci. USA 45: 335-344. GC revealed that ethylene is a cause, not consequence, of ripening Ethylene production precedes ripening and its associated CO2 production. Burg, S.P., and Burg, E.A. (1962). Role of ethylene in fruit ripening. Plant Physiol. 37: 179-189. Burg and Thimann made a key discovery about ethylene production Return to air after 4 hours oxygen deprivation Controls When an apple deprived of oxygen for four hours is returned to an aerobic environment, there is a dramatic burst of ethylene production. This suggests that an ethylene precursor accumulates in oxygen-deprived cells! Burg, S.P., and Thimann, K.V. (1959). The physiology of ethylene formation in apples. Proc. Natl. Acad. Sci. USA 45: 335-344. This ethylene precursor was called “Compound X” N2 Compound X Air O2 Radiolabeled methionine was used to identify Compound X Adams and Yang incubated apple slices in 14C-Met to see what compound accumulated when oxygen was withheld. Air 14C-Ethylene N2 to Air N2 Adams, D.O., and Yang, S.F. (1979). Ethylene biosynthesis: Identification of 1-aminocyclopropane-1-carboxylic acid as an intermediate in the conversion of methionine to ethylene. Proc. Natl. Acad. Sci. USA 76: 170-174. They identified Compound X! Compound X 14C-Met 14C-Ethylene Air N2 to Air Air N2 N2 N2 to Air Adams, D.O., and Yang, S.F. (1979). Ethylene biosynthesis: Identification of 1-aminocyclopropane-1-carboxylic acid as an intermediate in the conversion of methionine to ethylene. Proc. Natl. Acad. Sci. USA 76: 170-174. Compound X is aminocyclopropanecarboxylic acid (ACC) N2 Air O2 Ethylene synthesis Ethylene is produced from methionine (Met) via S-adenosylmethionine (AdoMet) by the action of ACC synthase (ACS) and ACC oxidase (ACO). Reprinted from Chae, H.S., and Kieber, J.J. (2005). Eto Brute? Role of ACS turnover in regulating ethylene biosynthesis. Trends Plant Sci.10: 291-296 with permission from Elsevier. Ethylene synthesis Methionine is regenerated via the Yang cycle, elucidated by Shang Fa Yang. Shang Fa Yang 1932 – 2007 Reprinted from Chae, H.S., and Kieber, J.J. (2005). Eto Brute? Role of ACS turnover in regulating ethylene biosynthesis. Trends Plant Sci.10: 291-296 with permission from Elsevier.; Image sources: University of California; Crenim The two key enzymes, ACS and ACO, are rare and unstable ACS is ACC synthase ACO is ACC oxidase Isolating these proteins and the genes that encode them was a significant effort. Tony Bleecker and Hans Kende made major contributions to the study of ethylene synthesis and responses. Tony Bleecker (1950 – 2005) Hans Kende (1937 - 2006) Reprinted from Chae, H.S., and Kieber, J.J. (2005). Eto Brute? Role of ACS turnover in regulating ethylene biosynthesis. Trends Plant Sci.10: 291-296 with permission from Elsevier.; Photos courtesy of Alan Jones (University of North Carolina) and Kurt Stepnitz (Michigan State University). Characterization of ACC synthase Proteins extracted from ripening tomatoes were used to make monoclonal antibodies. Bleecker, A.B., Kenyon, W.H., Somerville, S.C., and Kende, H. (1986). Use of monoclonal antibodies in the purification and characterization of 1-aminocyclopropane-1carboxylate synthase, an enzyme in ethylene biosynthesis. Proc. Natl. Acad. Sci. USA 83: 7755-7759. Characterization of ACC synthase The antibodies were screened for selectivity to ACC synthase and then used to immunoprecipitate the enzyme. ACC synthase The other two proteins are derived from the antibody. Bleecker, A.B., Kenyon, W.H., Somerville, S.C., and Kende, H. (1986). Use of monoclonal antibodies in the purification and characterization of 1-aminocyclopropane-1carboxylate synthase, an enzyme in ethylene biosynthesis. Proc. Natl. Acad. Sci. USA 83: 7755-7759. An antibody purification scheme was used to clone an ACC synthase cDNA Proteins were purified from ripening zucchini ACC synthase expression levels were induced to enrich the protein extract Auxin cytokinin, ACC Synthase inhibitors Uninduced protein extraction Induced protein extraction Sato, T., and Theologis, A. (1989). Cloning the mRNA encoding 1-aminocyclopropane-1-carboxylate synthase, the key enzyme for ethylene biosynthesis in plants. Proc.Natl. Acad. Sci. USA 86: 6621-6625. Y The partially purified inducedprotein extract was used to produce antiserum Induced protein extract Rabbit by Danko Y The antiserum was passed over a column containing uninduced extract The contaminating antibodies from the antiserum were removed by absorption onto the uninduced zucchini extract, which contains very little ACC synthase. The resulting antiserum was highly enriched for anti-ACC synthase antibodies. Sato, T., and Theologis, A. (1989). Cloning the mRNA encoding 1-aminocyclopropane-1-carboxylate synthase, the key enzyme for ethylene biosynthesis in plants. Proc.Natl. Acad. Sci. USA 86: 6621-6625. The anti-ACC antibody was used to screen a cDNA expression library Induced extract A cDNA expression library made from induced zucchini mRNA was screened using the purified antiserum to obtain an ACC synthase cDNA. Y Uninduced extract Y Blot probed with purified antiserum Blot probed with crude antiserum Sato, T., and Theologis, A. (1989). Cloning the mRNA encoding 1-aminocyclopropane-1-carboxylate synthase, the key enzyme for ethylene biosynthesis in plants. Proc.Natl. Acad. Sci. USA 86: 6621-6625. Yeast or E. coli cells expressing ACS cDNA make ACC This study provided proof that the cloned cDNA encodes ACS Cloning an ACO cDNA was similarly challenging…. Control ACO antisense Ethylene production in ripening fruit •A cDNA whose kinetics matched that of ethylene accumulation was cloned •introduction of an antisense construct into tomato reduced or eliminated ethylene production after wounding and during fruit ripening Reprinted by permission from Macmillan Publishers Ltd (Nature) Hamilton, A.J., Lycett, G.W., and Grierson, D. (1990). Antisense gene that inhibits synthesis of the hormone ethylene in transgenic plants. Nature 346: 284-287 Copyright 1990. Yeast expressing the ACO cDNA can make ethylene C2H4 After these key genes were cloned, it was possible to examine how their expression was regulated. Ethylene production is primarily regulated by ACS accumulation •ACS is encoded by 9 genes with diverse functions and expression patterns •Some ACS proteins are strongly regulated post-translationally ACS is encoded by 9 genes and functions as a dimer Type I Type III The ACS gene family products can potentially form 45 homoand heterodimers of which 25 are functional. Type II Type I S SSS Type II S Type III Yamagami, T., Tsuchisaka, A., Yamada, K., Haddon, W.F., Harden, L.A., and Theologis, A. (2003). Biochemical diversity among the 1-aminocyclopropane-1-carboxylate synthase isozymes encoded by the arabidopsis gene family. J. Biol. Chem. 278: 49102-49112. Different ACS dimers have different catalytic properties The subset of genes are that are expressed in any cell determines the types of ACS dimers that can form, and affects the rate of ethylene synthesis. The ACC synthase genes are differentially regulated and induced Tsuchisaka, A., and Theologis, A. (2004). Unique and overlapping expression patterns among the Arabidopsis 1-amino-cyclopropane-1-carboxylate synthase gene family members. Plant Physiol. 136: 2982-3000. ACS genes have unique and common functions Single mutant analysis shows that each gene has a unique and specific function Higher order mutants show that there are common essential functions including effects on flowering time..... Higher order ACS mutants flower earlier: ethylene delays flowering The pentuple mutant lacks activity of 5 genes, hexuple lacks 6, etc. Tsuchisaka, A., Yu, G., Jin, H., Alonso, J.M., Ecker, J.R., Zhang, X., Gao, S., and Theologis, A. (2009). A combinatorial interplay among the 1aminocyclopropane-1-carboxylate isoforms regulates ethylene biosynthesis in Arabidopsis thaliana. Genetics 183: 979-1003. ACS genes have unique and common functions Higher order ACS mutants are more susceptible to pathogens; ethylene contributes to pathogen resistance A mutant lacking all 9 ACS genes is not viable – ethylene is necessary for plant survival. Tsuchisaka, A., Yu, G., Jin, H., Alonso, J.M., Ecker, J.R., Zhang, X., Gao, S., and Theologis, A. (2009). A combinatorial interplay among the 1aminocyclopropane-1-carboxylate isoforms regulates ethylene biosynthesis in Arabidopsis thaliana. Genetics 183: 979-1003. Post-translational control of ACS activity Genetic studies identified ethyleneoverproducer (eto) mutants Wild Type AIR eto1 ETHYLENE AIR eto mutants show a tripleresponse in air and overproduce ethylene. Guzman, P., and Ecker, J.R. (1990). Exploiting the triple response of Arabidopsis to identify ethylene-related mutants. Plant Cell 2: 513-523. ETO1 is a component of a ubiquitinligase complex ETO1 targets ACS proteins for ubiquitination and proteolysis by the 26S proteosome. ETO1 ACS CUL3 WT ACS5 is selectively stabilized in loss-ofACS5 function eto1 mutants. -tubulin eto1 26S proteasome Reprinted by permission from Macmillan Publishers Ltd: Wang, K.L.C., Yoshida, H., Lurin, C., and Ecker, J.R. (2004). Regulation of ethylene gas biosynthesis by the Arabidopsis ETO1 protein. Nature 428: 945-950, copyright 2004. The eto2 and eto3 mutations affect stability of ACS5 and ACS9 ACS5 eto2 ACS9 eto3 The mutations in eto2 and eto3 are due to changes in the C-terminal region of ACS5 or ACS9. The mutant proteins are stabilized, enhancing ethylene synthesis. Chae, H.S., Faure, F., and Kieber, J.J. (2003). The eto1, eto2, and eto3 mutations and cytokinin treatment increase ethylene biosynthesis in Arabidopsis by increasing the stability of ACS protein. Plant Cell 15: 545-559. ACS proteins are normally subject to rapid proteolysis Degradation by the 26S proteasome Translation Normally ACS is continually synthesized and continually degraded, maintaining a very low level of ethylene ACS ETO1 CUL3 Liu, Y., and Zhang, S. (2004). Phosphorylation of 1-aminocyclopropane-1-carboxylic acid synthase by MPK6, a stress-responsive mitogen-activated protein kinase, induces ethylene biosynthesis in Arabidopsis. Plant Cell 16: 3386-3399. C-terminal phosphorylation stabilizes ACSs by interfering with ETO1 action C-terminal serines are targets for regulated phosphorylation Target of MAP Kinase P P P Type I S SSS P Type II Type III S Target of CDP Kinase The kinase activities are regulated by wounding and other hormones WOUNDING, PATHOGEN ATTACK ATP MAP kinase P P P Type I S SSS P Type II Type III S CDP kinase ATP ABIOTIC STRESS, OTHER HORMONES Regulation by proteolysis allows for rapid responses 2 1 3 1. 2. 3. 4. Transcription RNA processing Translation Enzyme action A process regulated by de novo transcription has a considerable lag before beginning. 4 A process regulated by proteolysis can respond very rapidly. X This method however requires a constant influx of energy to maintain. Ethylene synthesis and homeostasis - summary SAM Ethylene Biosynthesis ACS ACC ACO ACS proteins stabilized by wounding, other hormones C2H4 •Simple biosynthetic pathway regulated by expression and stability of ACS and ACO •ACS and ACO activities are tightly regulated transcriptionally and post-transcriptionally and sensitive to developmental cues, wounding and pathogen attack Ethylene response – receptors and downstream signaling In the 1980s, a genetic screen was carried out by Tony Bleecker, Hans Kende and colleagues to dissect the ethylene signaling pathway at the molecular level. Normal triple response Bleecker, A.B., Estelle, M.A., Somerville, C., and Kende, H. (1988). Insensitivity to ethylene conferred by a dominant mutation in Arabidopsis thaliana. Science 241: 1086-1089 reprinted with permission from AAAS; photo by Kurt Stepnitz, Michigan State University. Many signaling components were identified genetically Ethylene-insensitive mutants Ethylene-insensitive – no triple response in ethylene etr1 etr2 ein4 ein2 ein3 ein5 ein6 C2H4 Constitutive response – triple response in air Constitutive-response mutants ctr1 air ETHYLENE RESPONSE1 (ETR1) encodes an ethylene receptor ETR1 was the first protein to be unambiguously identified as a phytohormone receptor (1993) •ETR1 binds ethylene •ETR1 is similar in sequence to known-receptors in animal cells •ETR1 is membrane localized ethylene binding ETR1 GAF histidine kinase receiver The etr1-1 mutation is dominant WT WT WT WT ETR1 etr1-1 etr1-1 etr1-1 Introduction of the mutant etr1-1 allele into a wild-type plant causes an ethylene insensitive phenotype. From Chang, C., Kwok, S., Bleecker, A., and Meyerowitz, E. (1993). Arabidopsis ethylene-response gene ETR1: similarity of product to two-component regulators. Science 262: 539 – 544; reprinted with permission from AAAS. How can a mutant receptor have a dominant phenotype??? The receptors negatively regulate the responses No Ethylene Ethylene When bound to ethylene, the receptor does not shut off the ethylene response. When not bound to ethylene, the receptor shuts off the ethylene response. Responses OFF Responses ON A receptor that always shuts off signaling is dominant Ethylene Responses OFF Responses ON Responses OFF The dominant negative effect of etr1-1 and some other receptor mutants is because they always shut off responses, whether or not ethylene is present. Arabidopsis ethylene receptors resemble hybrid histidine kinases ethylene binding GAF histidine kinase receiver ETR1 The ethylene receptors structurally resemble the cytokinin receptors. However, unlike the cytokinin receptors, the histidine kinase domain has little role in signaling in vivo. Cytokinin receptor AHK4 CHASE domain histidine kinase receiver Arabidopsis ethylene receptor family ethylene binding GAF histidine kinase receiver ETR1 Subfamily I 83% 64% 64% ERS1 44-54% 38-41% 16-29% 32% EIN4 Subfamily II 58% 54% 38% 52% 55% 40% ETR2 ERS2 53% Loss-of-function mutations in ethylene receptors show constitutive ethylene responses Wild-type Responses OFF ers1 etr1 double loss-offunction mutant Responses ON Wang, W., Hall, A.E., O'Malley, R., and Bleecker, A.B. (2003). Canonical histidine kinase activity of the transmitter domain of the ETR1 ethylene receptor from Arabidopsis is not required for signal transmission. Proc. Natl. Acad. Sci. USA 100: 352-357, copyright National Academy of Sciences USA. But different receptors have different signaling strengths ers1 etr1 (Loss of Subfamily 1) Subfamily I ETR1 ERS1 Subfamily II etr1 etr2 ein4 EIN4 ETR2 ERS2 Hall, A.E., and Bleecker, A.B. (2003). Analysis of combinatorial loss-of-function mutants in the Arabidopsis ethylene receptors reveals that the ers1 etr1 double mutant has severe developmental defects that are EIN2 dependent. Plant Cell 15: 2032-2041. Ethylene receptor mutants have also been identified in other plants The tomato Never ripe mutant has a dominant, ethylene-insensitive phenotype, like etr1-1. Wild type Wild type Never ripe Never ripe From Wilkinson, J.Q., Lanahan, M.B., Yen, H.-C., Giovannoni, J.J., and Klee, H.J. (1995). An ethylene-inducible component of signal transduction encoded by Neverripe. Science 270: 1807-1809, reprinted with permisison from AAAS; Lanahan, M.B., Yen, H.C., Giovannoni, J.J., and Klee, H.J. (1994). The Never ripe mutation blocks ethylene perception in tomato. Plant Cell 6: 521-530. The ethylene-binding domain ethylene binding GAF histidine kinase receiver ETR1 NH2 There are three transmembrane segments in the ethylene binding domain of ETR1 (four in subfamily II receptors) From Rodríguez, F.I., Esch, J.J., Hall, A.E., Binder, B.M., Schaller, G.E., and Bleecker, A.B. (1999). A copper cofactor for the ethylene receptor ETR1 from Arabidopsis. Science 283: 996-998, Reprinted with permssion from AAAS Mutations in the transmembrane domain abolish ethylene binding NH2 Abolishing ethylene binding causes a dominant ethyleneinsensitive phenotype. From Rodríguez, F.I., Esch, J.J., Hall, A.E., Binder, B.M., Schaller, G.E., and Bleecker, A.B. (1999). A copper cofactor for the ethylene receptor ETR1 from Arabidopsis. Science 283: 996-998, Reprinted with permssion from AAAS; Hall, A.E., Grace Chen, Q., Findell, J.L., Eric Schaller, G., and Bleecker, A.B. (1999). The relationship between ethylene binding and dominant insensitivity conferred by mutant forms of the ETR1 ethylene receptor. Plant Physiol. 121: 291-300. Control of receptor activity by interaction with RTE/GR RTE/GR WT etr1-2 ETR1 rte REVERSION-TOETHYLENE SENSITIVITY Loss-of-function of RTE suppresses ethylene insensitive etr1-2 phenotype. These studies suggest that RTE/GR is a negative regulator of ethylene signaling. Green ripe gain-offunction alleles confer a dominant, ethyleneinsensitive phenotype in tomato fruit. Resnick, J.S., Wen, C.-K., Shockey, J.A., and Chang, C. (2006). REVERSION-TO-ETHYLENE SENSITIVITY1, a conserved gene that regulates ethylene receptor function in Arabidopsis. Proc. Natl. Acad. Sci. USA 103: 7917-7922; Barry, C.S. and Giovannoni, J.J. (2006) Ripening in the tomato Green-ripe mutant is inhibited by ectopic expression of a protein that disrupts ethylene signaling. Proc. Natl. Acad. Sci. USA 103: 7923-7928; copyright National Academy of Sciences USA. Signaling downstream of the receptors Genetic epistasis studies determined the order of action of the genes ethylene + etr1 = ctr1 etr1 ctr1 The double mutant has the same phenotype as ctr1, indicating that it acts downstream from ETR1. ETR1 CTR1 responses The genetic pathway of ethylene signaling C 2H 4 Receptor family ETR1 ERS1 ETR2 EIN4 ERS2 (insensitive - dominant) CTR1 (constitutive) EIN2 EIN3 EIN5 EIN6 (insensitive - recessive) responses to ethylene CTR1 is a negative regulator of ethylene signaling ctr1 Wild type The ctr1 mutant has a constitutive triple response. Air CTR1 is a serine/threonine protein kinase that resembles animal Raf kinases and is predicted to act in a MAPK cascade Ethylene No substrates have been identified yet Reprinted from Kieber, J.J., Rothenberg, M., Roman, G., Feldmann, K.A., and Ecker, J.R. (1993). CTR1, a negative regulator of the ethylene response pathway in arabidopsis, encodes a member of the Raf family of protein kinases. Cell 72: 427-441 with permission from Elsevier. The receptors directly interact with CTR1 and affect its activity Ethylene Ethylene In the presence of ethylene, CTR1 is inactive. CTR1 (active) Responses OFF In the absence of ethylene, CTR1 is active and inhibits the ethylene responses. CTR1 (inactive) Responses ON Reprinted from Kendrick, M.D., and Chang, C. (2008). Ethylene signaling: new levels of complexity and regulation. Curr. Opin. Plant Biol. 11: 479-485 with permission from Elsevier. The ethylene receptors directly interact with CTR1 ETR1 ERS A yeast two-hybrid assay revealed a specific interaction between the Cterminal region of the ethylene receptors and the N-terminal region of CTR1. CTR1 Colony growth and lacZ expression means the two proteins interact. Clark, K.L., Larsen, P.B., Wang, X., and Chang, C. (1998). Association of the Arabidopsis CTR1 Raf-like kinase with the ETR1 and ERS ethylene receptors. Proc. Natl. Acad. Sci. USA 95: 5401-5406, copyright National Academy of Sciences USA. CTR1 acts (somehow) through EIN2, a positive regulator of ET signaling Genetic studies show that EIN2 acts downstream of CTR1, but how the signal is transduced remains a mystery! ? EIN2 Responses ON Reprinted from Kendrick, M.D., and Chang, C. (2008). Ethylene signaling: new levels of complexity and regulation. Curr. Opin. Plant Biol. 11: 479-485 with permission from Elsevier. CTR1 acts (somehow) through EIN2, a positive regulator of ET signaling EIN2 has 12 membrane spanning domains but its function is unknown. ? EIN2 Responses ON Loss-of-function mutants are ethylene insensitive –EIN2 has a positive role. From Alonso, J., Hirayama, T., Roman, G., Nourizadeh, S., and Ecker, J. (1999). EIN2, a bifunctional transducer of ethylene and stress responses in Arabidopsis. Science 284: 2148 – 2152 reprinted with permission from AAAS; Kendrick, M.D., and Chang, C. (2008). Ethylene signaling: new levels of complexity and regulation. Curr. Opin. Plant Biol. 11: 479-485 with permission from Elsevier. EIN2 is subject to proteolysis in the absence of ethylene Ethylene EIN2 ETP1, 2 Responses ON ETP1 and ETP2 are components of the ubiquitin ligase complex that targets proteins for proteolysis. Ethylene destabilizes ETP1 and ETP2, stabilizing EIN2 and promoting downstream effects. From Qiao, H., Chang, K.N., Yazaki, J., and Ecker, J.R. (2009). Interplay between ethylene, ETP1/ETP2 F-box proteins, and degradation of EIN2 triggers ethylene responses in Arabidopsis. Genes Devel. 23: 512-521. Downstream of EIN2 a transcriptional cascade controls gene expression EIN3 and EIL1 are transcription factors that bind an ethylene binding site (EBS) in the promoter of ERF1. ERF1 encodes another TF that targets ethyleneresponsive genes. EIN2 EIN3/EIL1 EBS ERF1 GCC C2H4 Responsive Gene Reprinted from Chao, Q., Rothenberg, M., Solano, R., Roman, G., Terzaghi, W., and Ecker, J. (1997). Activation of the ethylene gas response pathway in Arabidopsis by the nuclear protein ETHYLENE-INSENSITIVE3 and related proteins Cell 89: 1133 – 1144 with permission from Elsevier; Kendrick, M.D., and Chang, C. (2008). Ethylene signaling: new levels of complexity and regulation. Curr. Opin. Plant Biol. 11: 479-485 with permission from Elsevier. In the absence of ethylene, EIN3 and EIL1 are targeted for proteolysis EBF1 and EBF2 are F-box proteins that target EIN3 and EIL1 for proteolysis EBF1/2 Degradation by the 26S proteasome via SCFEBF1/2 EIN3/EIL1 EBS ERF1 GCC C2H4 Responsive Gene Reprinted from Kendrick, M.D., and Chang, C. (2008). Ethylene signaling: new levels of complexity and regulation. Curr. Opin. Plant Biol. 11: 479-485 with permission from Elsevier. Accumulation of EBF1 and EBF2 is regulated in part at the RNA level EIN5 encodes a RNA exoribonuclease that affects the stability of EBF mRNA and so affects ethylene signaling. EIN5/XRN4 EBF1/2 Degradation by the 26S proteasome via SCFEBF1/2 EIN3/EIL1 EBS ERF1 GCC C2H4 Responsive Gene Reprinted from Kendrick, M.D., and Chang, C. (2008). Ethylene signaling: new levels of complexity and regulation. Curr. Opin. Plant Biol. 11: 479-485 with permission from Elsevier. Summary of ethylene synthesis and signaling SAM Ethylene Biosynthesis ACS ACC ACO C2H4 ETR1 and others RTE/GR CTR1 Ethylene Signaling EIN2 EIN3, EILs ERF1 and ERFs ETP1 and ETP2 EBF1 and EBF2 Ethylene perception and signaling - summary Arabidopsis genetics, and especially the easy-to-score triple response, were instrumental in identifiying the genes encoding the signaling pathway The pathway has a novel combination of proteins acting in a mainly linear pathway Negative regulation plays an important role! Protein turnover is an important regulatory mechanism Ethylene’s role in whole-plant processes • Shoot and Root elongation • Reproductive development • Sex determination • Petal senescence • Fruit ripening • Flooding responses – • Aerenchyma formation, leaf epinasty • Deepwater rice • Pathogen responses Ethylene restricts elongation of the shoot and root in the dark C2H4 C2H4 C2H4 C2H4 Auxin is required for ethylene effects in the root Auxin-signaling is required for ethylene-induced gene expression in the elongating region of the root. EBS GUS A reporter construct for ethylene-induced gene expression Stepanova, A.N., Yun, J., Likhacheva, A.V., and Alonso, J.M. (2007). Multilevel interactions between ethylene and auxin in Arabidopsis roots. Plant Cell 19: 2169-2185. Ethylene’s effects are mediated by auxin in the root Stepanova, A.N., Yun, J., Likhacheva, A.V., and Alonso, J.M. (2007). Multilevel interactions between ethylene and auxin in Arabidopsis roots. Plant Cell 19: 2169-2185. Ethylene contributes to apical hook formation through auxin effects Ethylene AIR HOOKLESS ARF2 ETHYLENE Differential growth Reprinted from Lehman, A., Black, R., and Ecker, J.R. (1996). HOOKLESS1, an ethylene response gene, is required for differential cell elongation in the Arabidopsis hypocotyl. Cell 85: 183-194 with permission from Elsevier. Sex determination in Cucumis Hermaphrodite Male Female Imperfect (nonhermaphroditic) flowers can lead to increased outcrossing and increased fitness. Image courtesy of Abdelhafid Bendahmane, URGV - Plant Genomics Research INRA Female flowers arise when stamen primordia abort Pistil Stamen Petals Sepals Image courtesy of Abdelhafid Bendahmane, URGV - Plant Genomics Research INRA Genes affecting sex determination encode ACS genes Elevated levels of ethylene production are correlated with developmental arrest of the stamen primordia Another sex determination gene affects receptor expression Downregulation of the ethylene receptor in stamen primordia makes these tissue more sensitive to ethylene Cell in developing pistil Cell in developing stamen Klee, H.J. (2004). Ethylene signal transduction. Moving beyond Arabidopsis. Plant Physiol. 135: 660-667. Ethylene promotes petal senescence Azad, A.K., Ishikawa, T., Ishikawa, T., Sawa, Y., and Shibata, H. (2008). Intracellular energy depletion triggers programmed cell death during petal senescence in tulip. J. Exp. Bot. 59: 2085-2095, by permission of Oxford University Press. Chemical and genetic approaches can prolong petal longevity STS and CACP interfere with ethylene binding to receptor Wildtype Expression of etr1-1 mutant allele represses petal responses to ethylene etr1-1 0 3 8 DAYS AFTER POLLINATION Reprinted from Serek, M., Woltering, E.J., Sisler, E.C., Frello, S., and Sriskandarajah, S. (2006) Controlling ethylene responses in flowers at the receptor level. Biotech. Adv. 24: 368-381 with permission from Elsevier; Wilkinson, J.Q., Lanahan, M.B., Clark, D.G., Bleecker, A.B., Chang, C., Meyerowitz, E.M., and Klee, H.J. (1997). A dominant mutant receptor from Arabidopsis confers ethylene insensitivity in heterologous plants. Nat Biotech 15: 444-447. Fruit ripening is induced by ethylene Ethylene Ripening includes: Changes in cell wall structure Pigment accumulation Flavor and aromatic volatile production Conversions of starches to sugars Ethylene synthesis increases dramatically during fruit ripening Ethylene accumulation Giovannoni, J.J. (2004). Genetic regulation of fruit development and ripening. Plant Cell 16: S170-180. Ethylene induces expression of ACS genes during ripening ACS Positive regulation – steep increase in ethylene production ACO SAM ACC LEACS6 C2H4 Perception LEACS1A LEACS4 LEACS2 Developmentally regulated Adapted from Barry, C.S., Llop-Tous, M.I., and Grierson, D. (2000). The regulation of 1-aminocyclopropane-1-carboxylic acid synthase gene expression during the transition from system-1 to system-2 ethylene synthesis in tomato. Plant Physiol. 123: 979-986. Fruit ripening can be controlled by controlling ethylene synthesis ACC synthase ACC oxidase H S-adenosyl methionine H C ACC C H H Ethylene Antisense ACC synthase Control Theologis, A., Zarembinski, T.I., Oeller, P.W., Liang, X., and Abel, S. (1992). Modification of fruit ripening by suppressing gene expression. Plant Physiol. 100: 549-551. Ethylene synthesis increases upon hypoxia caused by flooding Normally, soil has air pockets from which plant roots can take up oxygen. O2 When flooded, roots cannot take up oxygen, and become hypoxic – oxygen deprived. O2 C2H4 C2H4 Hypoxia induces ACC synthase and ethylene production. Ethylene synthesis increases upon hypoxia caused by flooding O2 O2 C2H4 C2H4 Ethylene induces cell death or cell separation and formation of aerenchyma – air channels through which oxygen can move into roots. Photo Author: Gordon Beakes©University of Newcastle upon Tyne Image courtesy LTSN Bioscience. A darkfield micrograph of a transverse section of a stem of Hippuris spp., showing aerenchyma. ACC moving from root to shoot induces ethylene formation and epinasty C2H4 In some plants ACC moves through the xylem into the shoot where it is converted to ethylene by ACC oxidase. C2H4 ACC C2H4 Leaf epinasty, caused by differential growth of the petiole, reduces light absorption by the Cleaves. 2H4 Rice is grown in regions subject to flooding After prolonged flooding, many strains of rice die, but submergence tolerant lines survive using either an escape or quiescence strategy. Reprinted by permission from Macmillan Publishers Ltd. NATURE from Voesenek, L.A.C.J., and Bailey-Serres, J. (2009). Genetics of high-rise rice. Nature 460: 959-960 copyright 2009 Rice is grown in regions subject to flooding The escape strategy involves an ethylene response. The quiescence strategy involves a gibberellin response. Gibberellin Ethylene Reprinted by permission from Macmillan Publishers Ltd. NATURE from Voesenek, L.A.C.J., and Bailey-Serres, J. (2009). Genetics of high-rise rice. Nature 460: 959-960 copyright 2009 In deepwater rice, ethylene induces internode elongation Preserved deepwater rice specimen These plants can grow as much as 15m high when subjected to flooding. Deepwater Reprinted by permission from Macmillan Publishers Ltd. From Hattori, Y., et al. (2009). The ethylene response factors SNORKEL1 and SNORKEL2 allow rice to adapt to deep water. Nature 460: 1026-1030, copyright 2009. Photo credit Moto Ashikari, Nagoya University. The elongation response is encoded by two ethylene-responsive transcription factors (ERFs) Deepwater rice Flooding SNORKEL1 & 2 Transcriptional response Non-deepwater rice Flooding Non-deepwater rice does not have these genes No transcriptional response Reprinted by permission from Macmillan Publishers Ltd. From Hattori, Y., et al. (2009). The ethylene response factors SNORKEL1 and SNORKEL2 allow rice to adapt to deep water. Nature 460: 1026-1030, c opyright 2009. Ethylene-insensitive tobacco has an impaired immune system Plants expressing a dominant ETR1 mutant gene lack resistance to normally harmless soil-borne fungi. Higher order ACS mutants are more susceptible to pathogens Knoester, M., van Loon, L.C., van den Heuvel, J., Hennig, J., Bol, J.F., and Linthorst, H.J.M. (1998). Ethylene-insensitive tobacco lacks nonhost resistance against soil-borne fungi. Proc. Natl. Acad. Sci. USA 95: 1933–1937, copyright National Academy of Sciences USA.; Tsuchisaka, A., Yu, G., Jin, H., Alonso, J.M., Ecker, J.R., Zhang, X., Gao, S., and Theologis, A. (2009). A combinatorial interplay among the 1-aminocyclopropane-1-carboxylate isoforms regulates ethylene biosynthesis in Arabidopsis thaliana. Genetics 183: 979-1003. Ethylene is required for wound or pathogen responses Plants that do not produce or respond to ethylene fail to induce expression of proteinase inhibitor 2 (pin2). Wounding ACO antisense plant Hours after wounding No treatment Wounding + silver thiosulfate, an inhibitor of ethylene responses. From O'Donnell, P.J., Calvert, C., Atzorn, R., Wasternack, C., Leyser, H.M.O., and Bowles, D.J. (1996). Ethylene as a signal mediating the wound response of tomato plants. Science 274: 1914-1917. Reprinted with permission from AAAS. In fact, both ethylene and jasmonate are needed for the defense response PDF1.2 is a defense gene that requires BOTH ethylene and jasmonate for induction (coi1 is a jasmonateinsensitive mutant). Ethylene works with jasmonate in defense-related gene expression. Penninckx, I.A.M.A., Thomma, B.P.H.J., Buchala, A., Metraux, J.-P., and Broekaert, W.F. (1998). Concomitant activation of jasmonate and ethylene response pathways is required for induction of a plant defensin gene in Arabidopsis. Plant Cell 10: 2103-2114. Ethylene/ JA responses are mediated by ERF1 and other TFs Lorenzo, O., Piqueras, R., Sanchez-Serrano, J.J., and Solano, R. (2003). ETHYLENE RESPONSE FACTOR1 integrates signals from ethylene and jasmonate pathways in plant defense. Plant Cell 15: 165-178. ETHYLENE- SUMMARY SAM Ethylene Biosynthesis ACS ACC ACO C2H4 ETR1 and others CTR1 Ethylene Signaling EIN2 EIN3, EILs ERF1 and ERFs Ethylene Responses •Cell elongation •Auxin synthesis and transport •Fruit ripening •Senescence •Pathogen defense Ongoing research - 1 What signals contribute to the posttranslational regulation of ACS accumulation? Does ACC itself function as a growth regulator? SAM How can ethylene production be optimized to enhance fruit quality? ACS ACC ACO C2H4 What is the mechanism of ethylene production by ACO? What are the transcriptional regulators of ACS and ACO genes? Ongoing research - 2 What are the roles of MAP kinases in synthesis EIN3/EIL1 and signaling? CTR1 ACS P P P S SSS ETR1 Many other ethyleneresponse mutants are being characterized and integrated into the pathway – what do they do? How does EIN2 work? EIN2 What role if any is played by the histidine kinase domain in the receptors? What do the different receptor isoforms do? How can we best use this knowledge to improve access to fresh food? enhanced ethylene response 4 Robles, L.M., Wampole, J.S., Christians, M.J., and Larsen, P.B. (2007). Arabidopsis enhanced ethylene response 4 encodes an EIN3-interacting TFIID transcription factor required for proper ethylene response, including ERF1 induction. J. Exp. Bot. 58: 2627-2639, by permission of Oxford University Press.