* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Guidelines for the use of zuclopenthixol acetate
Neuropharmacology wikipedia , lookup
National Institute for Health and Care Excellence wikipedia , lookup
Psychedelic therapy wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Drug interaction wikipedia , lookup
Adherence (medicine) wikipedia , lookup
Pharmacogenomics wikipedia , lookup
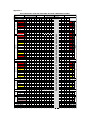
Guidelines for the use of zuclopenthixol acetate (Clopixol Acuphase®) injection Version 3.1 (Updated monitoring form dated November 2016) GUIDELINE REPLACED RATIFYING COMMITTEE DATE RATIFIED NEXT REVIEW DATE EXECUTIVE SPONSOR ORIGINAL AUTHOR REVIEWED / UPDATED BY Version 3 Drugs and Therapeutics Group August 2018 Executive Medical Director Jed Hewitt – Chief Pharmacist Helen Manuell – Lead Pharmacist (ESx) If you require this document in an alternative format, i.e. easy read, large text, audio or Braille please contact the pharmacy team on 01243 623349. Guidelines for the use of zuclopenthixol acetate (Clopixol Acuphase®) Injection Introduction In the past, Acuphase has often been too widely and possibly inappropriately used, sometimes without full regard being given to the fact that it is a potentially hazardous and toxic preparation with very little published information to support its use. Indeed, the Cochrane Library concludes that there is inadequate data on Acuphase and no convincing evidence to support its use in acute psychiatric emergency. So far as possible, it should therefore be reserved for the minority of patients who have a prior history of previous use and good response, with use defined in an advance directive. Similarly, as a general rule Clopixol Acuphase should not be used for Rapid Tranquillization unless such practice is also in accordance with an advance directive. Normally, Acuphase should never be considered as a first-line drug for Rapid Tranquillization as its onset of action will often not be rapid enough in these circumstances. In addition, the administration of an oil-based injection carries very high risk in a highly agitated patient. Acuphase should never be administered: In an attempt to hasten the antipsychotic effect of other therapy To a patient who is physically resistant, due to risk of intravasation Acuphase should never be used for patients: Who will accept oral antipsychotic medication Who are antipsychotic naïve Who are known to be sensitive to extrapyramidal side effects Who are unconscious Who are pregnant or are breast-feeding Who are known to have cardiac disease or renal or hepatic impairment Use outside the inpatient setting is not recommended, as the patient will require close monitoring over a full 24-hour period. Licensed Use. Acuphase is licensed as “initial treatment of acute psychoses, including mania and exacerbation of chronic psychoses, particularly where a duration of effect of 2-3 days is desirable”. It is important to recognise that onset of action is not as rapid as may be believed or required, particularly where rapid tranquillization is needed. Therefore, Acuphase should only be used after an acutely psychotic patient has required repeated injections of short-acting antipsychotics such as haloperidol or olanzapine, and/or sedative drugs such as lorazepam, and these have not been effective. (Unless there is an advance directive for its use). Further to this, Acuphase should be given only when enough time has elapsed to fully assess the response to previously injected drugs, ie. at least 15 minutes after IV injections and at least 60 minutes after those administered intramuscularly. Consideration should be given to zuclopenthixol plasma levels reaching a peak approximately 24-36 hours after administration of an Acuphase dose. Caution must therefore be applied if consideration is being given to the administration of a short-acting psychotropic IM injection during treatment with Acuphase, as excessive sedation and/or aggravated adverse events may occur if the patient is exposed to high plasma levels of multiple drugs. Dose Acuphase is licensed at a dose of 50mg to 150mg, (1-3ml), repeated if necessary after 2 or 3 days. (Some patients may need an additional injection between 1 and 2 days after the first, although at least 24 hours should be left between doses). The maximum dose per injection for an elderly patient is 100mg (2ml). For all patients the accumulated dosage must not exceed 400mg, (or 4 injections), within a 2-week period. Acuphase should not be viewed as a course of treatment and the patient should be carefully reviewed before each dose is prescribed / administered. The maximum licensed duration ensures that a treatment plan is put in place for the patient. More frequent administration or a more prolonged treatment period is not within the terms of the Product License and should only occur in very exceptional circumstances. The patient must be carefully monitored after each injection and a specific recording sheet is available for this purpose and should be retained in the patient’s notes following completion. The exact monitoring frequency is shown on the recording sheet. (See appendix). If used to good effect and the patient feels that they may benefit from its use in the future, then consideration should be given to the preparation of an advance directive. Onset and Duration of Action Sedative effects of Acuphase usually begin to appear within 2 hours of injection and may not reach a peak for another 24 to 36 hours. Significant effects may last for up to 72 hours although full elimination of the drug may not be complete for 7 days. Cross-Reference This guidance document should be read in conjunction with the Trust Rapid Tranquillisation Policy. References. Summary of Product Characteristics – Clopixol Acuphase. Lundbeck Ltd, August 2015 th The Maudsley Prescribing Guidelines, 12 Edition, 2015. The Psychotropic Drug Directory 2014. Stephen Bazire. Helen Manuell Lead Clinical Pharmacist August 2015 Appendix 1 ZUCLOPENTHIXOL ACETATE (ACUPHASE) PHYSICAL OBSERVATION CHART Patient name Ward Date of birth Tim e m onitoring initiated: Tim e m onitoring stopped: MEWS TIME (24 hour) 36 42 48 40 3 3 39 3 38.5 3 38.5 3 38 3 38 3 37.5 2 37.5 2 37 0 37 0 36 35 0 1 36 35 0 1 34 3 34 3 ≥200 3 190 2 180 2 170 2 160 1 150 1 140 1 130 0 120 0 110 0 100 0 90 1 80 1 70 3 60 3 50 3 40 3 140 > 3 130 3 120 3 110 2 100 1 90 0 80 0 70 0 60 0 50 40 1 3 ≥ 30 3 21-29 2 17-20 0 12-16 0 10-11 1 9 2 ≤8 Respirations 3 Temperature ≥200 3 190 3 180 3 170 3 160 3 150 3 140 3 130 3 120 3 110 3 3 2 0 0 0 2 3 100 90 80 70 60 50 40 Systolic BP Diastolic BP 140 3 130 3 120 3 110 2 100 1 90 0 80 0 70 0 60 0 50 40 1 3 Pulse ≥ 30 3 21-29 2 17-20 0 12-16 0 10-11 1 9 2 ≤8 3 Respirations 97-100 0 9 7 - 10 0 0 94-96 0 94-96 0 ≤ 93 3 ≤ 93 3 O xyge n S a t ura t io n Aw ake and active Aw ake and calm A s le e p but ro us a ble Asleep and U nr esp o nsive Temperature 30 Diastolic (Blood Pressure) (60-89) 24 Pulse 18 Respiratory Rate 12 SPO₂ SYSTOLIC Blood Pressure (100-139) Pulse (51 - 100) 6 3 Pulse Respirations (12 - 20) 2 39 Systolic BP Diastolic BP SPO₂ 1 40 Temperature Conscious Level 0 If patient is stable (total scores of 0 -1) consider discontinuing observations. Temperature (36.1 - 37.4 °C) Observations (HOURS) Oxygen Saturatio n A A R U 0 0 2 3 Concious Level DATE C o ns c io us Le v e l Co nscio us Level H ydra t e d- Y e s ( 0 ) o r N o ( 2 ) Hydrated TOTAL MEWS Initial TOTAL MEWS Initial Instructions: Zuclopenthixol acetate should ONLY be used in exceptional circumstances. It must not be routinely consided as an option in Rapid Tranquillization. Use this chart to record patient monitoring undertaken during a single episode of zuclopenthixol acetate (Acuphase) If patient consents to physical examinations, ALL observations on this chart should be recorded. If patient refuses observations, record as R. Respiration rate and consciousness should still be recorded. Signs of dehydration include dry mouth, UTIs, poor skin condition or reduced quantity of liquids comsumed Monitor at least hourly for the first 2 hours (especially if given with IM lorazepam) then every 6 hours. Continue monitoring for a minimum of 48 hours and longer if the patient remains unstable.