* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Internal Osmotic Pressure as a Mechanism of Retinal Attachment in
Survey
Document related concepts
Transcript
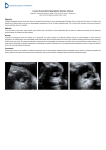
Internal Osmotic Pressure as a Mechanism of Retinal Attachment in a Vitreous Substitute WILLIAM J. FOSTER, HYDER A. ALIYAR, PAUL HAMILTON AND NATHAN RAVI* Surgical Services, St. Louis VA Medical Center, St. Louis, MO, 63106, USA and Department of Ophthalmology and Visual Sciences, Washington University, St. Louis, MO 63110, USA ABSTRACT: In this study, the possibility of using the internal osmotic pressure of intraocular polymeric hydrogel materials to attach the retina in the repair of a retinal tear or hole was investigated. This is in contrast to the conventional methods of retinal detachment repair (intraocular gas, polydimethylsiloxane, or n-perfluorooctane), which rely on surface tension and have recognized limits. The system selected for implementation of this scheme was based on an acrylamide copolymer that was crosslinked in an aqueous solution to provide a transparent hydrogel which allowed control of the swelling pressure. Synthetic hydrogels, such as those selected here, provide an alternative to materials currently used as vitreous prostheses. KEY WORDS: vitreous substitute, osmotic pressure, retinal detachment, retinal tamponade, hydrogel, vitreous prothesis, macular holes, retinal tears, retinal detachments, vitreous hemorrhage. INTRODUCTION Vitreous Biochemistry and Physiology he eye is conventionally divided into two anatomic regions, the anterior segment and the posterior segment. The anterior segment includes the cornea, iris, and lens (Figure 1). The posterior segment is composed of the vitreous body (also called the vitreous), the ciliary T *Author to whom correspondence should be addressed. E-mail: [email protected] Figures 1, 5, 6, and 7 appear in color online: http://ibe.sagepub.com Journal of BIOACTIVE AND COMPATIBLE POLYMERS, Vol. 21 — May 2006 0883-9115/06/03 0221–15 $10.00/0 DOI: 10.1177/0883911506064368 © 2006 SAGE Publications 221 222 W. J. FOSTER ET AL. Figure 1. Schematic diagram of the eye. body, retina, choroid, and sclera. The anterior segment structures are designed to admit light into the eye and to focus that light on the retina. The posterior segment structures are designed to detect light and transmit the resulting signal to the brain. The vitreous body is simply the clear “jelly-like” substance in the middle of the eye, posterior to the lens. It is a paucicellular natural hydrogel that is made up primarily of non-crosslinked Type II collagen fibrils and hyaluronic acid [1] and is approximately 99% water by weight. The non-crosslinked collagen forms a random polymer network that runs from one end of the vitreous cavity to the other and is responsible for the gelatinous vitreous. Intermeshed in the collagen polymer network, and also present in the region of liquid vitreous, is hyaluronic acid. This glycosaminoglycan, made up of two alternating monosaccharides (N-acetylglucosamine and glucuronic acid linked by glycoside bonds), forms a 1,000–10,000 Mw unbranched, coiled polyanion that has a high hydrated specific volume (2,000–3,000 cc/g). The concentration of the hyaluronic acid varies from 0.03–0.10% in humans [1]. The vitreous is non-uniform in density, with a 100–200 m-thick layer of solid cortical gel adjacent to the retina, ciliary body, and lens [1]. In addition, Cloquet’s canal, the central portion of the vitreous anterior to the optic nerve, frequently contains thin, multilayered, fenestrated sheaths of basal lamina tissue [1]. The vitreous is firmly attached to the anterior retina, where collagen fibers penetrate the retina and attach to the basement membrane of the ciliary epithelium and peripheral retinal glial cells [2]. The retina, the tissue that detects light, lines the inside of the eye and is in contact with the vitreous. The choroid is a highly vascular structure Internal Osmotic Pressure 223 that surrounds the retina (i.e., lies outside the retina) and provides nutrients to the retina. The sclera, which surrounds the choroid, is made of collagen fibrils with varying cross-sectional diameters and a haphazard arrangement [3]. This results in a lack of transparency for this tough outer coat of the eye. In young children, the vitreous, which is secreted by hyalocytes, is firm and helps to maintain the retina and keep it pressed against the back of the eye. It acts as a viscoelastic damper, thought to be due to the presence of hyaluronic acid [1] and the uniform distribution of the osmotic pressure ensures retinal attachment to choroid. As a person ages, the vitreous develops liquefied pockets, characterized by a paucity of collagen [1], and eventually separates from the retina completely, except for the most anterior retina. This has been ascribed to aggregation of the collagen fibrils into thicker cables [4–7] as well as changes and thickening of the basement membrane of the retina that result in loss of collagen fibril adherence [1] except in the anterior eye, where collagen fibrils remain firmly attached. During the acute phase of this separation (known as posterior vitreous detachment or PVD), the vitreous may be abnormally attached to the retina at some focal point. The vitreous will naturally move within the eye during normal eye movement and in the process can create a tear or hole in the retina at the point of adhesion. This retinal tear should be repaired as it can lead to retinal detachment (fluid under the retina) with the risk of blindness. Retinal Detachments and Treatments The vitreous body undergoes a nonuniform transition from a gellike substance in a young child to a more fluid-like substance in an older adult. Associated with this transition are a number of visionthreatening phenomena such as macular holes, retinal tears, retinal detachments, and vitreous hemorrhage. The mechanism for these phenomena is considered [8] to involve traction of the liquefying vitreous gel on the retina when currents are created in the vitreous by eye movements. The creation of a hole or tear in the retina by vitreous traction is a surgical emergency. Treatment of this hole or tear involves relieving the traction and blocking egress of fluid through the hole by reapproximation of the retina to the underlying tissues. Details of treatment options for this condition have been thoroughly reviewed by Wilkinson and Rice [8]. An increasingly common technique to achieve these results is vitrectomy surgery. When a patient undergoes a vitrectomy, 224 W. J. FOSTER ET AL. much of the vitreous is removed by using aspiration and cutting techniques to relieve the traction on the retina. Frequently a gas (air, sulfur hexafluoride, or perfluoropropane) is introduced into the eye to hold the retina against the back of the eye (the retinal pigment epithelial layer) until a scar is formed between the retina and the underlying tissue. This process can take weeks, and during this time, the installed compound must remain in contact with the retinal hole. For this reason, patients who have received intraocular gas are usually positioned for a week or more in a face-down position that many find difficult to maintain. Obviously, holes located in the inferior retina are not easily amenable to closure by intraocular gas. Silicone oil (polydimethylsiloxane) has been instilled in the eye for complex retinal tears, for inferior tears, or in patients unable to position themselves. Given its lower surface tension at the water interface (30 dynes/cm as opposed to 70 dynes/cm at the gas/water interface), the success rate for hole closure is frequently less [9]. In addition, the silicone oil must be removed later, which is difficult to remove completely, since it is toxic to intraocular structures and is associated with glaucoma and corneal decompensation [10] (both of which can lead to blindness). Vitreous Substitutes, Perfluoron and Silicone Oils Over the years, attempts to find vitreous substitutes has been mostly trial-and-error [11]. The materials currently used include the following: • • • • air; perfluorocarbon gasses (especially n-perfluoropropane); sulfur hexafluoride; saturated perfluoronated oils (particularly n-perfluorooctane [12] and n-perfluorodecalin [13]); • polydimethylsiloxane [14]. Materials that have been experimentally investigated include the following: • • • • • • perfluorophenanthrene [15]; perfluorotri-n-propylamine [16]; polymethyl-3,3,3-trifluoropropylsiloxane-co-dimethylsiloxane [17]; poly(2-hydroxyethyl acrylate) [18]; semifluorinated alkanes [19]; silicone gel [20]; 225 Internal Osmotic Pressure • • • • • • methylated collagen [21]; collagen/hyaluronic acid mixtures [22]; hydroxypropylmethyl cellulose [23]; crosslinked poly(vynyl) alcohol [24]; polymethylacrylamidoglycolate methyl ester [25]; crosslinked poly(1-vinyl-2-pyrrolidinone) [26]. All of these compounds either depend on surface tension [27] or are structurally similar to silicone oils and presumably depend on surface tension and the (as yet incompletely determined) other mechanisms through which silicone oils may act. In this paper, our efforts to develop alternatives to silicone oil and perfluorocarbon liquids for repair of complex retinal detachments are described. Unique to our system is the ability to form a hydrogel under physiologic conditions and then generate internal osmotic pressure, thus closing the retinal tear. We are trying to create an artificial version of the of the juvenile vitreous by developing new polymeric solgel systems. This would avoid issues related to patient positioning and known toxicity of current materials as well as provide better treatment of inferior tears. By selecting such systems, we also can avoid exposure of the intraocular tissues to toxic monomers and the thermal energy present in exothermic in vivo polymerization reactions. Injection, using a small-gauge needle, is the only practical procedure for the delivery of a vitreous substitute. The injection process induces some loss of elasticity when preformed hydrogel materials were injected [26] due to breakage of some of the crosslinks in the gel network. Such loss of elasticity does not occur in the hydrogel system investigated in this work because gelation occurs only in the vitreous cavity after the polymer is injected. For retinal tamponade with osmotic swelling to be achieved, the swelling pressure of the polymer in the eye should be positive but tailored in such a manner as to avoid excessive pressure on the retinal vasculature (possibly causing vascular occlusion) and retina. According to the Flory–Rehner theory [28], the swelling pressure can be expressed as: [ln(1 ) w ] G RT 2 3 (1) where is the molar volume of the solvent, is the volume fraction of the polymer, and w are the second- and third-order interaction parameters, and G is the elastic modulus of the swollen network. This relationship can be used to select an appropriate polymer composition. 226 W. J. FOSTER ET AL. METHODS The chemicals were purchased from Aldrich-Sigma Chemical Co. St. Louis, MO, USA and used without further purification. All reagents used were analytical grade. Preparation of poly(acrylamide-co-2-thiol ethyl acrylamide) hydrogel Acrylamide and bisacryloylcystamine (BAC) were copolymerized at acrylic mole ratios of 96/4 at 5% (w/w) monomer concentration in 25% ethanol (25:75 ethanol:water v/v). The hydrogel was then reductively liquefied followed by precipitation in methanol to obtain water-soluble thiol-containing polymer. These procedures have been previously reported [29,30]. The resulting soluble polymer obtained was labeled “the copolymer”. Characterization of the Copolymer The thiol (—SH) content present in the copolymer was determined by Ellman’s analysis [31]. The molecular weight of the reduced copolymer was determined with an HPLC-GPC system equipped with a triple detector (refractive index, light scattering, and viscosity; Viscotek, Houston, TX, USA), with a G4000PWXL column (Tosoh Biosep, Montgomeryville, PA, USA). The mobile phase was 20 mM Bis-Tris buffer (pH 6.0, 0.1% sodium azide). Samples were prepared in water (pH 4 and N2 saturated) at a concentration of 0.5% (w/v). Poly(ethylene glycol) standards (Viscotek, Houston, TX, USA) of molecular weight (Mw) 1,000 to 950,000 were used for calibration. Regelation in the Vitreous Cavity A stock solution of the copolymer (20 mL, 8% w/v, pH 5 in water) was prepared. Four milliliter aliquots with a final copolymer concentration of 7% w/v were prepared from this stock solution. The pH of each 4 mL aliquot was adjusted to 7.0 with a small amount of 1 M NaOH. This thiol copolymer solution was regelled in situ in the vitreous cavity by two methods: first, an equivalent amount of dithiodipropionic acid (DTDP) was added to a 1:1 molar ratio of the —SH content contained in the copolymer. The DTDP copolymer mixture was well stirred just before injection into the vitreous cavity. The copolymer rapidly regelled within minutes. A second method involved bubbling Internal Osmotic Pressure 227 air through the dissolved 8% polymer solution at pH 4 for 30 min before raising the pH to 7.0. The regellation process usually took several hours. Toxicity Testing of Materials Viability and toxicity testing were carried out on Chinese hamster ovary (CHO) cells in tissue culture using the methylthiazole tetrazolium (MTT) assay [32]. In this assay, 3-(4,5-dimethyl-2-thiazolyl)2,5-diphenyl-2H-tetrazolium bromide (thiazolyl blue) is converted to formazan, which forms a water-insoluble precipitate by an enzymatic reaction that occurs only in living cells. The experiment was performed as follows: CHO cells were plated at a density of 10,000 cells per a 16mm well in 24 well clusters. The media used was 1 mL per well of minimal essential medium (MEM). All tissue culture reagents were from Sigma Chemical Co (St. Louis, MO) with 10% fetal calf serum with gentamycin and penicillin/streptomycin. Twenty-four hours after plating, the media was aspirated, and fresh media containing the material to be tested was added. Cells were then incubated for 72 h. After incubation, 0.5 mL of M199 medium without phenol red, containing thiazolyl blue (MTT) (Sigma/Aldrich) (0.65 mg/mL) and 10% fetal calf serum was added. The cells were then incubated an additional 4 h. The medium was aspirated, and the precipitated formazan in the cell monolayer was dissolved in 200 L of DMSO. Samples were transferred to a microtiter plate and the absorbance at 540 nm was measured. Samples were performed in quadruplicate. Results obtained from the cells treated with the test compounds were expressed as a percent of results obtained from control, or untreated, cells. The IC50 was derived from a plot of the data as the concentration where the value obtained would equal 50% of control and was used as a measure of toxicity. To test for toxicity, a specific amount of the methanol precipitated, lyophilized copolymer (300 mg) was added to acidified (pH 6.0) MEM media (9.7 mL). This copolymer solution was allowed to dissolve for 24 h. The pH was then adjusted to pH 7.4 and the solution mixed with 10 mL of MEM with 20% fetal calf serum to give a final concentration of 15 mg/mL polymer in MEM with 10% fetal calf serum. The material was allowed to mix in the presence of air to allow the formation of disulfide bonds. The resultant media was added to the CHO cells for testing. In addition to the copolymer, linear acrylamide, acrylamide monomer, and DTDP were also tested under identical conditions. 228 W. J. FOSTER ET AL. Surgery Fresh human cadaver globes were obtained from the hospital morgue. A pneumatic vitrectomy was performed with a modified version of the standard technique [33]. The cadaver eyes were tilted so that the visual axis was horizontal. In 1 mL increments, air was injected into the vitreous cavity of the eyes, in 10 min intervals, through a pars plana approach (4 mm posterior to the limbus) with a 30-gauge needle. A 27-gauge needle was then placed through the pars plana inferiorly to allow vitreous fluid to drain. A total of 5 mL of air was injected. Ten minutes after the final injection of air, 4 mL of the copolymer solution was injected into the vitreous cavity of each eye through the pars plana with a 27-gauge needle. The eyes were wrapped in saline-saturated gauze and were kept overnight at room temperature. The globes were then formalin fixed, sectioned with a Surgitron cutting device (Ellman International Inc. Oceanside, NY), and photographed. RESULTS Preparation and Characterization of the Copolymers Copolymerization of acrylamide and BAC provided a unique hydrogel. The critical step in obtaining the desired water-soluble copolymers from crosslinked gels involves the complete reduction of all the disulfide bonds (—S—S—) in the gels into —SH groups, as shown in Figure 2. More details on the preparation and characterization of a similar thiol polymer can be obtained from previous reports [29,30]. The disulfide bonds of the gels were almost completely reduced by dithiothreitol (DTT). Ellman’s analysis shows a —SH content of (CH2 Ch2 CH CONH2 A AAm CH CO NH CH2CH2S)2 BAC s s s s s s s SH s B C HS s s s s s s s s s s s SH HS s Figure 2. Schematic representation for preparation, liquefaction of the crosslinked gel to give the copolymer. (a) Copolymerization with APS and TEMED. (b) Swelling and washing of the gel to remove unreacted acrylamide () and BAC () monomers. (c) Reductive liquefaction of the gel with DTT followed by precipitation in methanol, filtration, and vacuum drying to again get the copolymer. 229 Internal Osmotic Pressure 5.1 104 moles/g. The predicted value was 5.4 104 moles/g. The molecular weight distribution analysis of the copolymer showed a broad distribution with a polydispersity of 3.4 and weight average molecular weight (Mw) of 3.8 105. In Situ Regelation of the Copolymer Solution Regelation of copolymer can be achieved by air oxidation of the thiol groups or by a thiol-disulfide exchange reaction. The air oxidation took several hours to form gels at pH 7.5, but the exchange reaction yielded gelation in 5 min at pH 7.0. The gels obtained were optically transparent. The suitability of the water-soluble copolymer solution for in vivo regelation was established in the human cadaver vitreous cavities in vitro. Regelation was performed with 7% w/v (final concentration) solution of the copolymer. The thiol-disulfide exchange reaction, leading to the formation of gel, is schematically represented in Figure 3. As the copolymer was prepared monomer free and injected at the desired concentration, monomer toxicity was avoided. The swelling pressure of the polymer in the eye should be positive but tailored in such a manner as to avoid excessive pressure on the retinal vasculature (possibly causing vascular occlusion) and retina. The hydrated gel formed from the 7% w/v copolymer was noted to have a slightly higher elastic modulus compared with the consistency of natural vitreous. Gels of similar composition, formed in vitro, have an elastic modulus of 100 to 150 Pa and contain more than 95% water. The mechanical properties and swelling characteristics of the regelled hydrogel can be tuned through appropriate modification of the properties SH HS S SH S HS S SH HS S SH HS S S S S Figure 3. Schematic representation of regelation of copolymer solution through thioldisulfide exchange reaction with DTDP at pH 7.0 in the vitreous cavity. 230 W. J. FOSTER ET AL. Linear polyacrylamide AB4 MTT Abs, % of cont 100.0 50.0 0.0 0.0 1.0 2.0 3.0 4.0 5.0 6.0 7.0 8.0 9.0 10.0 MG/ML Figure 4. Toxicity of linear polyacrylamide, polymerized in the absence of a crosslinker compared with polyacrylamide polymerized with BAC thiol as a crosslinker (labeled AB4) (both polymers were methanol precipitated and the lyophilized material was used for toxicity testing as described). of the thiol polymer, such as molecular weight, thiol content, and hydrophobicity. Intravitreal Gel This protocol in cadaveric eyes was successfully implemented as depicted in Figures 5 to 7. The hydrated gels were consistently formed and were observed to have vitreous-like consistency. In performing this series of experiments, we have developed critical experience that will guide us in animal studies and trials of other sol-gel systems. Toxicity Results From the toxicity curves (Figure 4), linear polyacrylamide has an IC50 of 3.2 mg/mL, whereas the copolymer has an IC50 of 7.25 mg/mL. Toxicity can be reduced by further purification of the copolymer by Soxhlet extraction and dialysis, or multiple methanol reprecipitations [34]. Consistent with other observed work [35], when the acrylamide monomer was tested under identical conditions to those of the copolymer, it showed a high level of toxicity in CHO cells, with an IC50 of Internal Osmotic Pressure 231 Figure 5. The formed polymeric gel is visible after the sclera has been cut away. Figure 6. The eye, with gel in situ, has been coarsely cut to show the anatomically correct position of the gel within the globe. 0.02 mg/mL. Based on a weight/weight ratio, linear polyacrylamide showed a decrease in toxicity of 160-fold, and the thiol polyacrylamide polymer showed a decrease of 360-fold when compared with acrylamide monomer. DTDP was assayed and an IC50 of 7.8 mg/mL or 37 mM was obtained. The final concentration of DTDP needed to regel the copolymer at 7% w/w was 32 mM. 232 W. J. FOSTER ET AL. Figure 7. The removal of the polymer from the globe shows that the polymer is transparent. CONCLUSION Spontaneously forming reversible sol-gel systems are a reasonable alternative to current vitreous substitutes. By changing the paradigm from a liquid or gas to an artificial vitreous gel that approximates the retina against the RPE, we may avoid the limitations of current substitutes, such as, the need for face-down positioning and the need for a second operation to remove oil to prevent glaucoma and toxicity. Further work in vivo is needed to evaluate the long-term toxicity and the risk of complications. For the first time, the use of internal osmotic pressure to reapproximate the retina has been observed. In a series of proof-of-principle experiments, a commonly available monomer, acrylamide, was used to form hydrogels. Poly(acrylamide-co-BAC) hydrogels were prepared and reduced to obtain a water-soluble copolymer with pendant thiol (—SH) groups. An aqueous solution of the thiol polymers was then regelled to form a hydrogel, through a thiol-disulfide exchange reaction. In vitro regelation of this polymer system inside the vitreous cavity of human cadaver eyes indicates that this is a reasonable and potentially advantageous alternative to current vitreous substitutes. By changing the paradigm from a liquid or gas to a formed artificial vitreous that reapproximates the retina against the RPE through a mechanism of internal osmotic pressure, the limitations of current substitutes such as the need for face-down positioning are potentially avoided. Prelimi- Internal Osmotic Pressure 233 nary toxicity testing shows that the scrupulous removal of the acrylamide monomers by multiple methanol reprecipitation greatly reduced the toxicity of the material. Also, the introduction of thiol groups in the polymer matrix appears to have decreased the toxicity by two-fold over linear polyacrylamide. Further work is in progress to evaluate toxicity, the risk of complications of this material, and synthesis of new and better gel formulations. REFERENCES 1. Balazs, E.A. (1973). Fine Structure and Function of Ocular Tissues. The Vitreous, Int. Ophthalmol. Clin., 13: 169–187. 2. Hogan, M.J. (1963). The Vitreous, Its Structure, and Relation to the Ciliary Body and Retina. Proctor Award Lecture, Invest. Ophthalmol., 51: 418–445. 3. Zinn, K.M. and Mockel-Pohl, S. (1973). Fine Structure and Function of Ocular Tissues. The Cornea and Sclera, Int. Ophthalmol. Clin., 13: 93–107. 4. Balazs, E.A. and Denlinger, J.L. (1982). Aging Changes in the Vitreous. In: Sekuler, R., Kline, D. and Dismukes, K. (eds) Aging and Human Visual Function, A.R. Liss, New York, p. 4557. 5. Sebag, J. and Balazs, E.A. (1989). Morphology and Ultrastructure of Human Vitreous Fibers, Invest. Ophthalmol. Vis. Sci., 30: 1867–1871. 6. Sebag, J. (1987). Age-Related Changes in Human Vitreous Structure, Graefes Arch. Clin. Exp. Ophthalmol., 225: 89–93. 7. Los, L.I., van der Worp, R.J., van Luyn, M.J. and Hooymans, J.M. (2003). Age-Related Liquefaction of the Human Vitreous Body: LM and TEM Evaluation of the Role of Proteoglycans and Collagen, Invest. Ophthalmol. Vis. Sci., 44: 2828–2833. 8. Wilkinson, C.P. and Rice, T.A. (1997). Michels Retinal Detachment, 2nd edn, Mosby, St. Louis. 9. Goldbaum, M.H., McCuen, B.W., Hanneken, A.M., Burgess, S.K. and Chen, H.H. (1998). Silicone Oil Tamponade to Seal Macular Holes without Position Restrictions, Ophthalmology, 105: 2140–2148. 10. Federman, J.A. and Schubert, H.D. (1988). Complications Associated with the Use of Silicone Oil in 150 Eyes after Retina-Vitreous Surgery, Ophthalmology, 95: 870–876. 11. Colthurst, M.J., Williams, R.L., Hiscott, P.S. and Grierson, I. (2000). Biomaterials Used in the Posterior Segment of the Eye, Biomaterials, 21: 649–665. 12. Chang, S., Sparrow, J.R., Iwamoto, T., Gershbein, A., Ross, R. and Ortiz, R. (1991). Experimental Studies of Tolerance to Intravitreal Perfluoro-noctane Liquid, Retina, 11: 367–474. 234 W. J. FOSTER ET AL. 13. Velikay, M., Wedrich, A., Stolba, U., Datlinger, P., Li, Y. and Binder, S. (1993). Experimental Long-term Vitreous Replacement With Purified and Nonpurified Perfluorodecalin, Am. J. Ophthalmol., 116: 565–570. 14. Cibis, P.A., Becker, B., Okun, E. and Canaan, S. (1962). The Use of Liquid Silicone in Retinal Detachment Surgery, Arch. Ophthalmol., 68: 590–599. 15. Peyman, G.A., Conway, M.D., Soike, K.F. and Clark, L.C. (1991). LongTerm Vitreous Replacement in Primates with Intravitreal Vitreon or Vitreon Plus Silicone, Ophthalmic Surg., 22: 657–664. 16. Bryan, J.S., Friedman, S.M., Mames, R.N. and Margo, C.E. (1994). Experimental Vitreous Replacement with Perfluorotri-n-propylamine, Arch Ophthalmol., 112: 1098–1102. 17. Doi, M. and Refojo, M.F. (1995). Histopathology of Rabbit Eyes with Silicone-Fluorosilicone Copolymer Oil as Six Months Internal Retinal Tamponade, Exp. Eye Res., 61: 469–478. 18. Chan, I.M., Tolentino, F.I., Refojo, M.G., Fournier, G. and Albert, D.M. (1984). Vitreous Substitute. Experimental Studies and Review, Retina, 4: 51–59. 19. Kuckelkorn, R., Becker, J., Zeana, D., Dahlke, C., Meinert, H. and Kirchhof, B. (1997). Long-term Tolerance of Intravitreal Semifluorinated Alkane in the Experimental Animal, Ophthalmic Res., 29: 125. 20. Peyman, G.A., Conway, M.D., Karacorlu, M., Soike, K.F., Bhatt, N., Clark, L.C. and Hoffmann, R.E. (1992). Evaluation of Silicone Gel as a Long-term Vitreous Substitute in Non-human Primates, Ophthalmic Surg., 23: 811–817. 21. Liang, C.P., Peyman, G.A., Serracarbassa, P., Calixto, N., Chow, A.A. and Rao, P. (1998). An Evaluation of Methylated Collagen as a Substitute for Vitreous and Aqueous Humor, Int. Ophthalmol., 22: 13–18. 22. Nakagawa, M., Tanaka, M. and Miyata, T. (1997). Evaluation of Collagen Gel and Hyaluronic Acid as Vitreous Substitutes, Ophthalmic Res., 29: 409–420. 23. Fernandez-Vigo, J., Refojo, M.F. and Verstraeten, T. (1990). Evaluation of a Viscoelastic Solution of Hydroxypropyl Methylcellulose as a Potential Vitreous Substitute, Retina, 10: 148–152. 24. Yamauchi, A. (1991). Synthetic Vitreous Body of PVA Hydrogel. In: DeRossi, D., Kajiwara, K., Osada, Y. and Yamauchi, A. (eds) Polymer Gels, Plenum Press, New York, pp. 127–134. 25. Chirila, T.V., Sharp, C., Moore, S.R., Davies, M.J., Constable, I.J., Hong, Y., Vijayasekaran, S., Humphrey, M.F., Dalton, P.D., Tahija, S.G., Maley, M.A.L. and Cuypers, M.J.H. (1995) Synthetic Hydrogel as an Artificial Vitreous Body. A One-Year Animal Study of Its Effects on the Retina, Cells Mater., 5: 83–96. 26. Hong, Y., Chirila, T.V., Vijayasekaran, S., Dalton, P.D., Tahija, S.G., Cuypers, M.J.H. and Constable, I.J. (1996). Crosslinked Poly(1-vinyl-2- Internal Osmotic Pressure 27. 28. 29. 30. 31. 32. 33. 34. 35. 235 pyrrolidinone) as a Vitreous Substitute, J. Biomed. Mat. Res., 30: 441–448. Foster, W.J. and Chou, T. (2004). Physical Mechanisms of Gas and Perfluoron Retinopexy and Sub-Retinal Fluid Displacement, Phys. Med. Biol., 49: 2989. Flory, P.J. and Rehner, J. (1943). Statistical Mechanics of Crosslinked Polymer Networks II. Swelling, J. Chem. Phys., 11: 521. Aliyar, H., Hamilton, P.D., Remsen, E.E. and Ravi, N. (2003). Bio-inspired Preparation of Nanoparticles by Intramolecular Disulfide Crosslinks, Polym Prepr., 44: 285. Foster, W.J., Ali, A.H. and Ravi, N. (2003). Sol-Gel Systems as Vitreous Substitutes, Association for Research in Vision Science and Ophthalmology, Fort Lauderdale, FL, Invest. Ophthalmol. Vis. Sci., E-abstract. Ellman, G.L. (1959). Tissue Sulfhydryl Groups, Arch. Biochem. Biophys., 82: 70–77. Bruining, M.J., Blaauwgeers, H.G., Kuijer, R., Pels, E., Nuijts, R. and Koole, L.H. (2000). Biodegradable Three-dimensional Networks of Poly(dimethylamino ethyl methacrylate). Synthesis, Characterization and In Vitro Studies of Structural Degradation and Cytotoxicity, Biomaterials, 21: 595–604. Thresher, R.M., Ehrenberg, M. and Machemer, R. (1984). Gas-Mediated Vitreous Compression: An Experimental Alternative to Mechanized Vitrectomy Graefes Arch. Clin. Exp. Ophthalmol., 221: 192–198. Hamilton, P.D., Aliyar, H. and Ravi, N. (2004). Biocompatibility of Thiolcontaining Polyacrylamide Polymers Suitable for Ophthalmic Applications, Polym. Prepr., 45: 495. Nordin-Andersson, M., Walum, E., Kjellstrand, P. and Forsby, A. (2003). Acrylamide-Induced Effects on General and Neurospecific Cellular Functions During Exposure and Recovery, Cell Biol. Toxicol., 19: 43–51.