* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download O 2 - physics.muni.cz
Survey
Document related concepts
Transcript
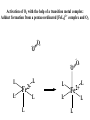
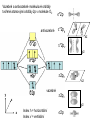
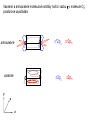
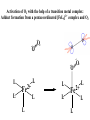
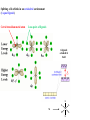
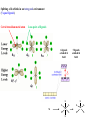
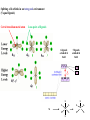
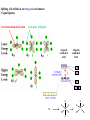
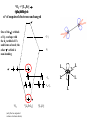
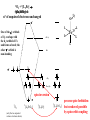
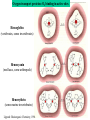
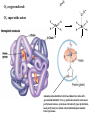
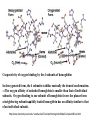
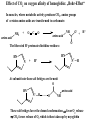
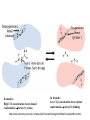
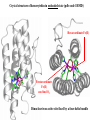
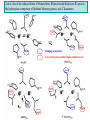
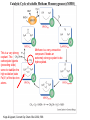
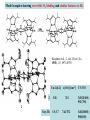
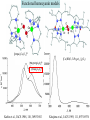
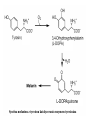
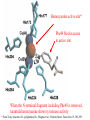
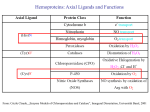
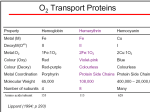
F5351 Metalloproteins reacting with oxygen 1. Why do aerobic organisms need metalloproteins? 2. Oxygen transport proteins & Oxygenases 2.1. Hemoglobin, Myoglobin & Cytochrome P450 2.2. Hemerythrin & Methane monooxygenase 2.3. Hemocyanin & Tyrosinase 3. Conclusion Jiří Kozelka 13.11. 2014 [email protected] 1. Why do aerobic organisms need metalloproteins? Cells of aerobic organisms need oxygen. First, oxygen is needed to gain energy from food (respiration) and for other processes. Second, toxic organic substances are eliminated from the body by oxidation, whereupon OH-groups are attached to the molecule (this specific process is called hydroxylation, in mammals it occurs mainly in the liver). This renders the toxic molecule water-soluble and it can be eliminated (through the urine in mammals). Cellular respiration C6H12O6 + 6 O2 6 CO2 + 6 H2O DG0 = -674 kcal/mol Elimination of xenobiotics. Example: hydroxalation of hexane by Cytochrome P450 OH n-Hexane 1-Hexanol minor OH + 1/2 O2 3-Hexanol minor 2-Hexanol major Cytochrome P450 OH Use of oxygen by aerobic organisms is hampered by two problems: 1. The solubility problem Water solubility of oxygen at 25oC and pressure = 1 bar is at 40 mg/L water. This is not enough to guarantee the oxygen supply to mitochondria by mere diffusion. Cells of aerobic organisms use therefore oxygen transporters. 2. The kinetic problem Oxygen has two unpaired electrons in its ground state and forms therefore a triplet state. The overwhelming majority of organic molecules (such as glucose or n-hexane) have all electrons paired and occur therefore in the singlet state. The products of oxidation of organic molecules, CO2 and H2O, are also in singlet states. According to the so-called Wigner-rule, processes in which the spin-state changes are « spin-forbidden », that is, they have a large kinetic barrier. The solution of the problem is binding of O2 to a transition metal complex. In transition metal complexes, spin-state changes are less inhibited due to the spin-orbit coupling. The oxygen-bound metal complex can therefore transit from a triplet state to a singlet state, and then react with an organic substrate which has also a singlet ground-state. Molecular orbital level diagram for O2: 3Sg- state 2p 2p 2s 2s O O2 O Activation of O2 with the help of a transition metal complex: Adduct formation from a pentacoordinated [FeL5]2+ complex and O2 _ _ O_ O_ _ _ O L L 2+ Fe L L L L L O_ 2+ Fe L L L Vazebné a antivazebné molekulové orbitály tvořené atomovými orbitály 2p v molekule O2 antivazebné s*2p p*2ph xz p*2pv yz p2ph vazebné y x xz p2pv yz z Index h = horizontální Index v´= vertikální s2p Vazebné a antivazebné molekulové orbitály tvořící vazbu p v molekule O2: prostorové uspořádání antivazebné vazebné y x z O O p*2pv p*2ph O O p2pv p2ph Activation of O2 with the help of a transition metal complex: Adduct formation from a pentacoordinated [FeL5]2+ complex and O2 _ _ O_ O_ _ _ O L L 2+ Fe L L L L L O_ 2+ Fe L L L Splitting of d orbitals in an octahedral environment (6 equal ligands) Cetral transition metal atom Lone-pairs of ligands 6 ligands octahedral field z2 x2-y2 xy xz yz L L L M M L L L Splitting of d orbitals in an tetragonal environment (5 equal ligands) Cetral transition metal atom Lone-pairs of ligands 6 ligands octahedral field 5 ligands octahedral field z2 x2-y2 xy xz yz L L L L L L L M M L L M L L Splitting of d orbitals in an tetragonal environment (5 equal ligands) Cetral transition metal atom Lone-pairs of ligands 6 ligands octahedral field 5 ligands octahedral field z2 x2-y2 x2-y2 z2 xy xz yz L L L L L L L M M L L M L L Splitting of d orbitals in an tetragonal environment (5 equal ligands) Cetral transition metal atom Lone-pairs of ligands 6 ligands octahedral field 5 ligands octahedral field z2 x2-y2 x2-y2 z2 xy xz yz xy xz xz L L L L L L L M M L L M L L + 1[L5Fe] 3[L FeO ] spin-allowed: 5 2 n° of unpaired electrons unchanged 3O 2 dx2-y2 dz 2 L p* dxy dxz,dyz 3 O2 (only the two unpaired valence electrons shown) 3 [L5FeO2] 1 [L5Fe] L Fe2+ L L L + 1[L5Fe] 3[L FeO ] spin-allowed: 5 2 n° of unpaired electrons unchanged 3O 2 s* One of the p* orbitals of O2 overlaps with the dz2 orbital of Fe and forms a bond; the other p* orbital is non-bonding dx2-y2 dz 2 L p* dxy dxz,dyz s 3 O2 (only the two unpaired valence electrons shown) 3 [L5FeO2] 1 [L5Fe] L Fe2+ L L L + 1[L5Fe] 3[L FeO ] spin-allowed: 5 2 n° of unpaired electrons unchanged 3O 2 _ _ O s* L One of the p* orbitals of O2 overlaps with the dz2 orbital of Fe and forms a bond; the other p* orbital is non-bonding L dx2-y2 O_ Fe2+ L L L d z2 p* dxy dxz,dyz spin inversion s 3 O2 (only the two unpaired valence electrons shown) 3 [L5FeO2] 1 [L5Fe] 1 [L5FeO2] process spin-forbidden but rendered possible by spin-orbit coupling In transition metal complexes, spin-orbit coupling renders spin-forbidden transitions possible. Metal complexes can therefore activate (triplet) oxygen for reactions with (singlet) organic molecules. [MLn]m+ + 3O2 1[ML m+ O ] n 2 + 1[Substrate] 1[Oxidation Metal-oxygen adducts can also be used as oxygen carriers! 2. Oxygen transport proteins & oxygenases products] Oxygen transport proteins: O2 binding in active sites Hemoglobin (vertebrates, some invertebrates) Hemocyanin (molluscs, some arthropods) Hemerythrin (some marine invertebrates) Lippard: Bioinorganic Chemistry, 1994 _ _ O_ O2 oxygen molecule O_ -_ _ O_ - O2 superoxide anion N N Fe 2+ N N N N N Fe . O_ 3+ N N N Aminokyselina histidin tvořící koordinativní vazbu k Fe „proximální histidin“. Toto je jediná kovalentní vazba mezi porfyrinem železa a proteinem. Ostatní síly jsou hydrofobní, mezi porfyrinovým cyklem a hydrofobními postranními řetězci proteinu. in vertebrates 2 2e- Reduction of O2 to H2O Catalyzed by the enzyme Cytochrome-oxidase 2.1. Hemoglobin, Myoglobin & Cytochrome P450 153 amino acids http://www.ul.ie/~childsp/CinA/Issue64/TOC36_Haemoglobin.htm Vazba myoglobinu (Mb) na kyslík Mb + O2 MbO2 Cvičení 1: definujte rovnovážnou konstantu pro zpětnou reakci (tzv. disociační konstantu, Kd) Cvičení 2: definujte saturaci vazebných míst, Y, definovanou rovnicí dole, pomocí Kd a [O2] jako proměnných. Nahraďte ve vzorcích pro Kd a pro Y koncentraci [O2] parciálním tlakem p(O2). Cvičení 3 Vypočtěte křivku frakční saturace kyslíku na myoglobinu. Disociační konstanta komplexu MbO2 je, při 37 °C, pH = 7 a p = 760 Torr, Kd = 2.8 Torr. p(O2) [Torr] 0.5 1 2 3 5 10 20 30 40 50 60 70 80 90 Y [%] Y [%] 100 80 60 Cvičení 4: Jaký význam má směrnice saturační křivky v bodě p(O2) = 0? Znázorněte graficky závislost dY/dp(O2) na p(O2) 40 20 0 0 30 60 p(O2) [Torr] 90 Saturační křivka hemoglobinu neodpovídá jedné jediné rovnovážné reakci Vazba O2 na jednu hemovou skupinu hemoglobinu zvyšuje afinitu pro O2 dalších jednotek „Kooperativní efekt“ Cooperativity of oxygen binding by the 4 subunits of hemoglobin: In deoxygenated form, the 4 subunits stabilize mutually the domed conformation. The oxygen affinity of unloaded hemoglobin is smaller than that of individual subunits. Oxygen binding to one subunit of hemoglobin favors the planar form at neighboring subunits fully loaded hemoglobin has an affinity similar to that of an individual subunit. http://www.chemistry.wustl.edu/~edudev/LabTutorials/Hemoglobin/MetalComplexinBlood.html Effect of CO2 on oxygen afinity of hemoglobin: „Bohr-Effect“ In muscles, where metabolic activity produces CO2, amino groups of certains amino acids are transformed to carbamate: NH2 + O C O amino acid amino acid HN N + H+ + H+ O The liberated H+ protonates histidine residues: HN O- NH N+ H At subunit interfaces salt bridges are formed: O HN N+ amino acid H O- NH These salt bridges favor the domed conformation favor O2 release CO2 favors release of O2 which is then taken up by myoglobin In muscles: High CO2 concentration favors domed conformation favors O2 release In bronchi: Low CO2 concentration favors planar conformation favors O2 binding http://www.chemistry.wustl.edu/~edudev/LabTutorials/Hemoglobin/MetalComplexinBlood.html Fe(II)-O2, Fe(III)-O2-, or Fe(IV)-O22-? FeIV _ _ O Fe _ _ _ _ _O Fe(IV)-O22- . FeIII . Pauling dioxygen . _ _ FeIII _ _ O _O _ _O _ _O Fe(II)-O20 peroxide _ O _ O II Fe(III)-O2- _ . Weiss superoxide What experimental data can be used to determine whether oxygen in oxyhemoglobin resembles more to Fe(III)-O2- or to Fe(II)-O2? Stretching frequencies and bond lengths in dioxygen species Species [A] nO-O [cm-1] d O-O O2 + 1905 1.12 O2 1580 1.21 O2 - 1097 1.33 O22- 802 1.49 M-O2- 1100-1150 1.24-1.31 M- O22- 800-900 1.35-1.50 1105 1.22 Mb-O2 Oxymyoglobin resembles FeIII-O2- F5351 Metalloproteins reacting with oxygen 1. Why do aerobic organisms need metalloproteins? 2. Oxygen transport proteins & Oxygenases 2.1. Hemoglobin, Myoglobin & Cytochrome P450 2.2. Hemerythrin & Methane monooxygenase 2.3. Hemocyanin & Tyrosinase 3. Conclusion Jiří Kozelka 13.11. 2014 [email protected] Hemoproteins: Axial Ligands and Functions From: Cécile Claude, „Enzyme Models of Chloroperoxidase and Catalase“, Inaugural Dissertation, Universität Basel, 2001 Modification of the FeII/FeIII redox potential by the protein environment Hemoprotein proximal ligand FeII (Red.) stable Strong oxidants Em for FeII/FeIII (mV) FeIII/FeII (aq.) FeIII/FeII - +770 Human hemoglobin FeIII/FeII His +150 Microperoxidase11-CO FeIII/FeII His +100 Chloroperoxidase FeIII/FeII Cys- -150 NO synthase neuronal FeIII/FeII Cys- -250 Horse-radish peroxidase FeIII/FeII His -280 Cytochrome P450 2C5 FeIII/FeII Cys- -330 Catalase FeIII/FeII Tyr- -460 FeIII (Ox.) stable Strong reductants Source: C. Capeillere-Blandin, D. Matthieu & D. Mansuy, Biochem. J. 2005, 392, 583-587 Different metalloproteins need different redox potential for their function. Cytochrome P450 needs to access the unusual oxidation state Fe(V) to be able to oxidize even unreactive substrates. Therefore, it uses the negatively charged cysteine ligand which donates electrons to Fe and stabilizes the high oxidation state. One of strategies that proteins employ to modify the redox potential is using different proximal ligands. Examples of Cytochrome P450 substrates Hydroxylation at: -aliphatic carbons -aromatic carbons -double bonds -heteroatoms local anesthetic steroid hormone carcinogen from fungi antibiotic Alkaloid from Taxus brevifolia, potent anti-cancer drug Cytochrome P450cam (Campher-5-monooxygenase; pdb-code 1T86) access for substrate and O2 Hlavní dva rozdíly mezi hemoproteiny myoglobin a cytochrom P450, důležité pro jejich různé funkce: 1. Přístupový kanál vedoucí ke kofaktoru (hemu) je u myoglobinu velmi úzký, nedovoluje přístup větším molekulám než O2. U cytochromu P450 je kanál širší a v blízkosti kofaktoru obsahuje místo s vysokou afinitou pro specifické substráty. 2. Distální cystein a okolí kofaktoru snižuje u cytochromu P450 oxidačněredukční potenciál Fe, takže tento metaloprotein může fungovat jako oxygenáza a Fe v katalytickém cyklu může krátkodobě existovat v oxidačním stupni Fe(V). Tento velmi reaktivní přechodný stav je schopen hydroxylovat i poměrně nereaktivní alifatické atomy uhlíku. F5351 Metalloproteins reacting with oxygen 1. Why do aerobic organisms need metalloproteins? 2. Oxygen transport proteins & Oxygenases 2.1. Hemoglobin, Myoglobin & Cytochrome P450 2.2. Hemerythrin & Methane monooxygenase 2.3. Hemocyanin & Tyrosinase 3. Conclusion Jiří Kozelka 13.11. 2014 [email protected] http://notes.chem.usyd.edu.au/course/codd/CHEM3105/Metalloproteins3.pdf (HOI2)- Crystal structure of hemerytrhin in unloaded state (pdb-code 1HMD) Hexacoordinate Fe(II) Pentacoordinate Fe(II) can bind O2 Dinuclear iron active site fixed by a four-helix bundle Amino acids/subunit 153 113 628 Sipuncula Priapulida Brachiopoda Hemerythrin je metaloprotein transportující kyslík u některých bezobratlých Magelona papillicornis Active sites of the reduced forms of Hemerythrin, Ribonucleotide Reductase R2 protein, the hydroxylase component of Methane Monooxygenase, and D9 desaturase Bridging carboxylates Extra carboxylates stabilize higher oxidation states Catalytic Cycle of soluble Methane Monooxygenase (sMMO) This is a very strong oxydant. The carboxylate ligands (preceding slide) serve to stabilize the high oxidation state Fe(IV) of the two iron atoms. Methane is a very unreactive compound. Needs an extremely strong oxydant to be hydroxylated. Kopp & Lippard, Current Op. Chem. Biol. 2002, 568 F5351 Metalloproteins reacting with oxygen 1. Why do aerobic organisms need metalloproteins? 2. Oxygen transport proteins & Oxygenases 2.1. Hemoglobin, Myoglobin & Cytochrome P450 2.2. Hemerythrin & Methane monooxygenase 2.3. Hemocyanin & Tyrosinase 3. Conclusion Jiří Kozelka 13.11. 2014 [email protected] Amino acids/subunit 153 113 628 Hemocyanin je metaloprotein transportující kyslík u většiny měkkýšů a u některých korýšů Panulirus interruptus Linulus polyphemus Octopus dofleini Megathura crenulata Hemocyanin: History 1878 Leon Federicq: Sur l‘hemocyanine, substance nouvelle de sang de Poulpe (Octopus vulgaris) (Compt. Rend. Acad. Sci. 87, 996-998) Discovery 1901 M. Henze: Zur Kenntniss des Haemocyanins Z. Physiol. Chem. 33, 370 Hemocyanin contains copper 1940 W. A. Rawlinson, Australian J. Exp. Biol. Med. Sci. 18, 131 Oxy-hemocyanin is diamagnetic Známé a hypotetické (*) komplexy mědi s jednotkou O2 http://webdoc.sub.gwdg.de/diss/2003/ackermann/ackermann.pdf On the search for functional hemocyanin model compounds Karlin et al., JACS 1988, 110, 3690’3692 The first model complex showing reversible O2 binding by a dicopper unit However, this complex differs from oxy-Hc: Cu-Cu[Å] 1 1 υ(O-O)[cm-1] 4.36 Oxy-Hc 3.5-3.7 834 744-752 Karlin et al., J. Am. Chem. Soc. 1988, 110, 3690-3692 UV-VIS 440(2000) 525(11500) 590(7600) 1035(160) 340(20000) 580(100) Model complex showing reversible O2 binding and similar features to Hc Kitajima et al., J. Am. Chem. Soc. 1989, 111, 8975-8976 Cu-Cu[Å] 2 3.56 υ(O-O)[cm-1] 741 UV-VIS 349(21000) 551(790) 2 Oxy-Hc 3.5-3.7 744-752 340(20000) 580(100) Functional hemocyanin models [(tmpa)2Cu2O2]2+ [Cu{HB(3,5-iPr2pz)3}]2(O2) Karlin et al., JACS 1988, 110, 3690’3692 Kitajima et al., JACS 1989, 111, 8975-8976 UV-Vis absorption spectra of the oxy forms of hemocyanin and tyrosinase ps→d pv→d d→d 5-9 years later (1994, 1998): Active sites in hemocyanins determined by X-ray crystallography Magnus et al.,Proteins Struct. Funct. Gen.1994 Limulus polyphemus Cuff et al.,J.Mol.Biol.1998 Octopus dofleini An earlier model for hemocyanin... …turned out to be a model for the enzyme tyrosinase! Karlin et al., JACS 1984, 106, 2121-2128 L-DOPAquinone Syntéza melaninu z tyrosinu katalyzovaná enzymem tyrosináza http://pollux.chem.umn.edu/~kinsinge/new_homepage/research/gss_presentation_3/sld019.htm Tyrosinase versus Hemocyanin The coupled binuclear copper sites in tyrosinase and hemocyanin are very similar. Why is then tyrosinase capable of reacting with substrates while hemocyanin is not? Solomon (Angew. Chem. Int. Ed. Engl. 2001, 40, 4570-450): Difference in accessibility of the active site Rates of peroxide displacement by azide (measured using UV absorption) at 4°C: Hemocyanins: k 0.04 h-1 Tyrosinase: k =0.95 h-1 Hypothesis, 1980: Solomon et al., JACS 1980, 102, 7339-7344, p.7343 Angew. Chem. Int. Ed. 2001, 40, 4570-4590 Proof, 1998 (J. Biol. Chem. 273, 25889-25892): Hemocyanine active site* Phe49 blocks access to active site When the N-terminal fragment including Phe49 is removed, tarantula hemocyanine shows tyrosinase activity * From X-ray structure of L.polyphemus Hc., Magnus et al., Proteins Struct. Funct.Gen.19, 302-309 Conclusions In many cases, metalloproteins use the same or similar active site for different purposes. The strategies to confer a particular activity to a given site include - Allowing/disallowing access of substrates to the active site (including the dynamics of diffusion of substrate/product) -Modifying the electrostatic potential by mutating the amino acids coordinated to the metal or surrounding the binding pocket -Architecture of the binding pocket defines substrate selectivity and affects energy of transition states→governs reaction outcome














































![1 [L 5 FeO 2 ] - physics.muni.cz](http://s1.studyres.com/store/data/000263887_1-9a7fea8feae8a4c4c33cd53b2038de6b-150x150.png)
