* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Polymers - hrsbstaff.ednet.ns.ca
Survey
Document related concepts
Transcript
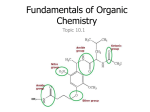
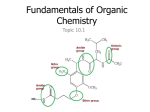
Functional Groups CHEMISTRY 11 MS. MCGRATH Functional Groups • A functional group is a portion of a molecule that is a recognizable / classified group of bound atoms • In organic chemistry it is very common to see molecules comprised mainly of a carbon backbone with functional groups attached to the chain • The functional group gives the molecule its properties, regardless of what molecule contains it; they are centers of chemical reactivity • A functional group can also be the reactive double bond in an alkene or the triple bond of an alkyne Functional Groups • The functional group within a molecule need to be identified when naming • The suffix of a compound’s name indicates the functional group of a molecule • For example, the suffix –ene indicates the presence of a double bond • For example, the suffix –ol indicates the presence of a hydroxyl group Functional Groups • Compounds with the same functional group often have similar physical properties • Compounds with the same functional group react chemically in very similar ways Functional Groups We will be looking at the following compounds which each have a single bonded functional group: Alcohols Aldehydes Ketones Carboxylic acid Esters Alcohols • Has a functional group written as –OH, or hydroxyl • Alcohols can be identified as: primary - the hydroxyl group is bonded to a carbon that is bonded to a carbon that is bonded to only one other carbon secondary - the hydroxyl group is bonded to a carbon that is bonded to a carbon that is bonded to two other carbons tertiary - the hydroxyl group is bonded to a carbon that is bonded to a carbon that is bonded to three other carbons Alcohols - Nomenclature Step 1: Locate the longest chain that contains the –OH group attached one carbon atom Step 2: Replace the –e at the end of the alkane with –OH Step 3: Add the position number before the root name to identify th position of the –OH group. If there is more than one –OH group, leave the –e in the name of the alkane, place the appropriate prefix (di-, tri-, etc) before the suffix -ol Step 4: Name and number other branches on the main chain. Add the name of the branches to the prefix.