* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Bridging work * gcse to as level sciences. standard defintions and
Survey
Document related concepts
Transcript
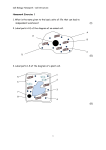
AS Biology (Bridging Work) Tasks To support your move from GCSE to AS Level Biology, there are 3 tasks for you to complete in preparation for your course. Please bring these to your first lesson in September 2016. The three tasks include: 1. Maths for Biology (Questions 1-9) 2. Researching cell structure 3. Learning standard definitions and mathematics 1 1. Maths for Biology 1 Rewrite the following using prefixes: 12000 g 0.005 m 0.000087 V 2 What do the following units represent? gcm-3 Jkg-1 mm3s-1 kgm-2y-1 3 Rewrite the following in standard form: 1942 kg 0.007 m 0.000002 A 0.034 s 14.5 MJ 178846 km 440 mm 4 How many nm are there in 450mm? 5 Write 84µm in mm. 6 Write 180µm as mm. 7 Write 0.2mm in µm. 8 A holly leaf was measured and had a diameter of 6.5cm. a) What is its diameter in mm? b) What is its diameter in nm? c) What is its diameter in nm? 9. 2 2. Cell Structure Cells are the building blocks of life. Research the 2 main types of cells Eukayotes and Prokaryotes. Draw or print and learn a labelled diagram of these two types of cells and complete a structure to function table for the following organelles listed below. Nucleus Plasma membrane Mitochondria Centriole Lysosome Cytoplasm Golgi apparatus (body) Rough endoplasmic reticulum Smooth endoplasmic reticulum Microtubules Theses websites will help http://www.s-cool.co.uk/a-level/biology/cells-and-organelles/revise-it/organelles http://www.biologyguide.net/cells/ultrastructure.htm 3. Standard Definitions and Mathematics Revise and learn the following definitions and mathematical concepts in preparation for AS Level Biology. Units, measurements and standard form Units are very important part of biology. The common units you should know are: unit m kg A s o C M J name metres kilograms amps Seconds* Degrees celsius1 Molar Joule Measurement of Distance or length mass current time temperature concentration energy *Notice ‘s’ is the abbreviation for seconds, not ‘sec’. 1You may come across the unit K (Kelvin). To convert a K temperature to oC, take away 273. E.g. 373K = 100oC 3 However, there are several units derived from these basic units that you will come across commonly in biology. These are: Unit cm3 ml mm µm nm mV name centimetres cubed millilitres millimetres micrometres or microns nanometres millivolts Measurement of Volume, usually solids and gases* Volume, liquids* length length length voltage Notice that cm3 and ml are an equal measure i.e. 1cm3 = 1ml What happened to litres? Instead of using litres (l), at A level you will be expected to use dm 3 (decimetres cubed). This avoids confusing l for litres with a number 1. Millilitres are still represented as ml. ‘Per’ At GCSE, you would have written metres per second like this: m/s ms-1 A levels use a different notation: There is a mathematical reason for this, but you don’t need to know it (unless you are desperate to find out!). The minus sign when present in units tells you that it should be read as ‘per’, e.g. kg per second kgs-1 bubbles per minute bubbles min-1 per litre dm-3 4 Prefixes These go before a unit to alter its magnitude. You are familiar with some of them already. symbol M k m µ n prefix Mega kilo milli micro nano meaning x 1,000,000 (million) x 1,000 ÷ 1000 ÷ 1,000,000 (millionth) + 1,000,000,000 (billionth) Example MJ kg mV µm nm Millivolts are often used in measuring voltage in cells. µm are commonly used in measurements of cells and organelles. nm are used in measuring wavelengths of light. Standard form Biology often uses numbers that are too large to be written down conveniently. Standard form is a short hand way for writing large or small values. Instead of 1400 m standard form would be 1.4 x 103 m This is the same as saying 1.4 x 10 x 10 x 10. If you work this out, it is the same as 1400 m. You can use 1.4km which is the same thing, but as you will see below, it is good practice to get used to using standard form. Notice that the first value will be a number between 1 and 9, so that: 1450 m is 1.49x103 m Another way to think about it is by moving the digits along, so: 1.49x103m move the digits 3 places to the left of the decimal point: 1 1 4 9 . . 0 5 4 9 0 0 However, you will be much more likely to come across small values is biology. In standard form, a minus sign is used, so that: 0.003m is 3x10-3 m This time, you move the digits 3 places to the right of the decimal point: 3 0 . . 0 0 3 It gets easier when you start to recognise the relationship between standard form and the prefixes: Standard form x103 x10-3 x10-6 x10-9 Same as kilo milli micro nano x1000 ÷10000 ÷1,000,000 ÷1,000,000,000 Moles. In the equation for respiration, each symbol could represent a single atom or molecule, e.g. there are 6 oxygen molecules, or 1 glucose molecule. Scientists would read this equation as the symbols representing a mole (mol) of the substance, e.g. 1 mol of glucose reacts with 6 mol of oxygen to make 6 mol of carbon dioxide and 6 mol of water. 1 mol of a substance contains exactly the same number of atoms/molecules (6.02 x 1023). The relative atomic mass of an element (R.A.M.) can be used to determine the mass of 1 mol of an element, e.g. 12 g carbon = 1 mol of C 16 g oxygen = 1 mol of O 40 g calcium = 1 mol of Ca 6 For molecules, add the R.A.M. of all the atoms present. This is known as the Relative Molecular Mass (R.M.M) e.g. 32 g oxygen = 1 mol of O2 17 g of ammonia = 1 mol NH3 44 g of carbon dioxide = 1 mol CO2 Useful formulae Mean average = (Σ x) / n Add the values together, and then divide by the number of different values. Given the symbol x . % change mass Final mass – original mass x 100 original mass % error Experimental result – actual result x 100 Actual result 7 Glossary of terms The following subject specific vocabulary provides definitions of key terms used in AQA's AS and A-level Biology, Chemistry and Physics specifications. Accuracy A measurement result is considered accurate if it is judged to be close to the true value. Calibration Marking a scale on a measuring instrument. This involves establishing the relationship between indications of a measuring instrument and standard or reference quantity values, which must be applied. For example, placing a thermometer in melting ice to see whether it reads 0⁰C, in order to check if it has been calibrated correctly. Data Information, either qualitative or quantitative, that have been collected. Errors See also uncertainties. measurement error The difference between a measured value and the true value. anomalies These are values in a set of results which are judged not to be part of the variation caused by random uncertainty. random error These cause readings to be spread about the true value, due to results varying in an unpredictable way from one measurement to the next. Random errors are present when any measurement is made, and cannot be corrected. The effect of random errors can be reduced by making more measurements and calculating a new mean. 8 systematic error These cause readings to differ from the true value by a consistent amount each time a measurement is made. Sources of systematic error can include the environment, methods of observation or instruments used. Systematic errors cannot be dealt with by simple repeats. If a systematic error is suspected, the data collection should be repeated using a different technique or a different set of equipment, and the results compared. zero error Any indication that a measuring system gives a false reading when the true value of a measured quantity is zero, egg the needle on an ammeter failing to return to zero when no current flows. A zero error may result in a systematic uncertainty. Evidence Data that have been shown to be valid. Fair test A fair test is one in which only the independent variable has been allowed to affect the dependent variable. Hypothesis A proposal intended to explain certain facts or observations. Interval The quantity between readings egg a set of 11 readings equally spaced over a distance of 1 metre would give an interval of 10 centimetres. Precision Precise measurements are ones in which there is very little spread about the mean value. Precision depends only on the extent of random errors – it gives no indication of how close results are to the true value. 9 Prediction A prediction is a statement suggesting what will happen in the future, based on observation, experience or a hypothesis. Range The maximum and minimum values of the independent or dependent variables; For example a range of distances may be quoted as either: 'From 10cm to 50 cm' or 'From 50 cm to 10 cm' Repeatable A measurement is repeatable if the original experimenter repeats the investigation using same method and equipment and obtains the same results. Reproducible A measurement is reproducible if the investigation is repeated by another person, or by using different equipment or techniques, and the same results are obtained. Resolution This is the smallest change in the quantity being measured (input) of a measuring instrument that gives a perceptible change in the reading. Sketch graph A line graph, not necessarily on a grid, that shows the general shape of the relationship between two variables. It will not have any points plotted and although the axes should be labelled they may not be scaled. True value This is the value that would be obtained in an ideal measurement. Uncertainty The interval within which the true value can be expected to lie, with a given level of confidence or probability egg “the temperature is 20 °C ± 2 °C, at a level of confidence of 95 %”. 10 Validity Suitability of the investigative procedure to answer the question being asked. For example, an investigation to find out if the rate of a chemical reaction depended upon the concentration of one of the reactants would not be a valid procedure if the temperature of the reactants was not controlled. Valid conclusion A conclusion supported by valid data, obtained from an appropriate experimental design and based on sound reasoning. Variables These are physical, chemical or biological quantities or characteristics. categoric variables Categoric variables have values that are labels egg s of plants or types of material or reading at week 1, reading at week 2 etc. continuous variables Continuous variables can have values (called a quantity) that can be given a magnitude either by counting (as in the case of the number of shrimp) or by measurement (egg light intensity, flow rate etc.). control variables A control variable is one which may, in addition to the independent variable, affect the outcome of the investigation and therefore has to be kept constant or at least monitored. dependent variables The dependent variable is the variable of which the value is measured for each and every change in the independent variable. independent variables The independent variable is the variable for which values are changed or selected by the investigator. nominal variables A nominal variable is a type of categoric variable where there is no ordering of categories 11