* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Homeostasis - the ability or tendency of an organism or cell to
Survey
Document related concepts
Transcript
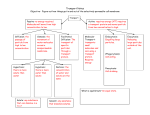
Background to Plasmolysis Lab Homeostasis – the ability of an organism to maintain chemical or physical equilibrium. Plasmolysis - shrinkage or contraction of the cytoplasm away from the wall of a living plant or bacterial cell, caused by loss of water through osmosis. This can cause the cell membrane to lyse. Lysis (Greek λύσις, lusis from luein = to separate) refers to the death of a cell by breaking of the cellular membrane, often by viral or osmotic mechanisms that compromise its integrity. A solution containing the contents of lysed cells is called a "lysate". Turgid – When the cytoplasm and water vacuoles of a cell are filled, the cell membrane will push against the cell and the cell is described as being turgid. A flaccid cell is created when the cell membrane shrinks or contracts during plasmolysis. Osmosis is the net movement of water across a selectively permeable membrane driven by a difference in solute concentrations on the two sides of the membrane. A selectively permeable membrane is one that allows unrestricted passage of water, but not solute molecules or ions. Different concentrations of solute molecules on either side of a semi-permeable membrane will lead to different concentrations of water molecules on either side of that membrane. On the side of the membrane with higher water concentration (i.e. a lower concentration of solute), more water molecules will strike the pores in the membrane in a given interval of time. More strikes equates to more molecules passing through the pores, which in turn results in net diffusion of water from the compartment with high concentration of free water to that with low concentration of free water. This net movement of water from an area of high water concentration/low solute concentration across a membrane to an area of lower water concentration/higher solute concentration is called osmosis. (High water to solute ratio Low water to solute ratio) Isotonic solution: The solutions being compared have equal concentration of solutes. Hypertonic solution: The solution with the higher concentration of solutes. Hypotonic solution: The solution with the lower concentration of solutes.