* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download DORV DR KSHITIJ
Electrocardiography wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
History of invasive and interventional cardiology wikipedia , lookup
Heart failure wikipedia , lookup
Cardiothoracic surgery wikipedia , lookup
Artificial heart valve wikipedia , lookup
Coronary artery disease wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Cardiac surgery wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Aortic stenosis wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
Mitral insufficiency wikipedia , lookup
Atrial septal defect wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
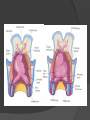
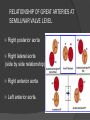
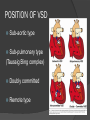
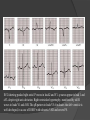
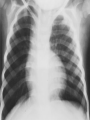
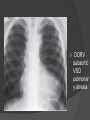
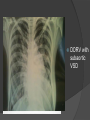
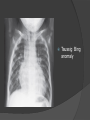
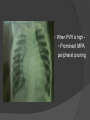
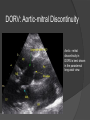
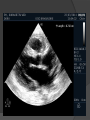
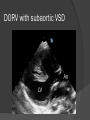
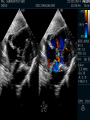
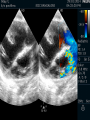
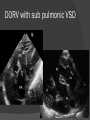
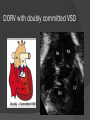
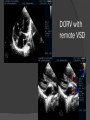
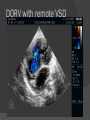
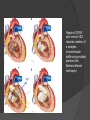
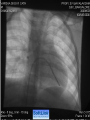
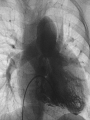
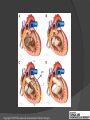
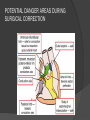
Dr. Kshitij Mavade Introduction Newfield et al – Both great arteries and arterial trunks arise exclusively from RV, neither semilunar valve is in fibrous continuity with either AV valve, and usually, a VSD is present and represents the only outlet from the left ventricle. Pulmonary valve or subpulmonary stenosis may be present or absent. Introduction Lev et al. One complete arterial trunk and at least half of the other arterial trunk emerge from RV, and there may or may not be mitral-aortic or mitral-pulmonary continuity Van Praagh et al – Relied on subaortic and subpulmonary conus to produce mitral discontinuity as diagnostic marker. • Kirklin- Malformation in which the whole of one great artery and more than half of the other originates from the right ventricle Anderson and his colleagues(Tynan et al) DORV- abnormal ventriculoarterial connection characterised by more than half of each great artery originating above the morphologic right ventricle HISTORY 1703 • The earliest report in French • John Abernathy, an assistant surgeon at St Bartholomew’s 1793 Hospital in London, described “partial transposition” • Karl von Vierordt called double outlet right ventricle partial transposition to signify that the aorta was transposed but 1898 the pulmonary trunk was normally aligned • Witham introduced double outlet right ventricle as a diagnostic term for a partial transposition complex 1957 HISTORY 1957 - Kirklin, first repair of DORV with subaortic VSD, at the mayo clinic 1961- Neufeld et al – physiological classification based on presence or absence of PS & position of VSD 1968- Patrick & Mcgoon- surgical classification based on relationship of GA & VSD Incidence <1% of CHD 0.06 case per 1000 live birth No sexual or racial predilection No associated Genetic defect identified Mostly sporadic, 22q11 deletion associated with some cases DORV EMBRYOLOGY Goor & Edwards: - DORV appears to represent a primitive embryologic condition because of failure to achieve conotruncal rotation and left shift of the conus. CONUS: A circular tube of muscle, upon which semilunar valve sits up. The more conal muscle present beneath a semilunar valve, the more that valve is pushed superiorly and anteriorly. Van praagh theory of conal underdevelopment “ the distal or semilunar part of infundibulum or conus arteriosus performs an arterial switch during cardiogenesis” DORV represent a part of the whole spectrum of conotruncal anomaly Spectrum of conotruncal anomalies P P A VSD A TOF A A P P DORV TGA (double coni) PATHOLOGIC CLASSIFICATION Based on •Location of VSD to great arteries •Great artery relationship Ventricular septal defect The only outflow tract of the left ventricle. Mostly conoventricular. Accordingly however DORV can be classified with respect to VSD location. Subaortic Subpulmonary Doubly committed Non committed. RELATIONSHIP OF GREAT ARTERIES AT SEMILUNAR VALVE LEVEL Right posterior aorta Right lateral aorta (side by side relationship) Right anterior aorta Left anterior aorta. POSITION OF VSD Sub-aortic type Sub-pulmonary type (Taussig Bing complex) Doubly committed Remote type GREAT ARTERY RELATIONSHIPS AT THE LEVEL OF SEMILUNAR VALVES Right posterior aorta The aortic valve and trunk originate from the right ventricle at a location posterior and to the right of the pulmonary valve and its arterial trunk Right lateral aorta (side-by-side relationship) The aorta is to the right of the pulmonary artery, and the semilunar valves lie approximately in the same transverse and coronal plane. This is the classically described great artery relationship in DORV Right anterior aorta (D-malposition)The aorta is to the right and anterior to the pulmonary artery. This grouping also may include some cases with the aorta directly anterior Left anterior aorta (L-malposition)The aorta is to the left and anterior to the pulmonary artery. Thihs is least common great artery position. 4 x 4 TYPES OF DORV, AND MORE… 16 possible variations of DORV based on the great artery relationships and the location of the VSD In addition, an intact ventricular septum allows four other possible types of DORV, depending on the great artery relationships However, these include only cases with situs solitus of the atrial and viscera, AV concordance, and two well-developed ventricles and AV valves Multiple other variations and combinations are possible if one also includes situs inversus and situs ambiguus as well as AV discordance RELATIONSHIP OF GREAT ARTERIES AND VSD Reationship Location of VSD Of great Sub Sub Non – remote artereis aortic pulmonary commited Total Normal 3% Side by side Right anterior aorta 0 0 0 3% 46% 8% 3% 7% 64% 16% 10% 0 0 26% Left anterior aorta 3% 0 0 7% TOTAL 68% 22% 3% 7% 4% DORV with Subaortic VSD Aorta VSD DORV with Subpulmonic VSD Aorta PA VSD PHYSIOLOGIC CLASSIFICATION Depends on: Presence or absence of associated pulmonary stenosis. Relationship of VSD to great arteries. DORV may simulate that of large VSD, TOF, TGA. ASSOCIATED ANOMALIES Pulmonary Stenosis - most common – 40-70%, - frequently co-exists with subaortic VSD (80%); - bicuspid pulmonary valve; - rarely seen in subpulmonary VSD type. ASSOCIATED ANOMALIES Subaortic stenosis – 3%; Frequently co-exist with sub-pulmonary VSD type (50%). Coarctation – 12%; frequent in subpulmonary VSD type 50%. Mitral valve anomalies – 10%; frequent in remote VSD ASD – 10% TAPVC – 2% AV canal – 5%, Common in remote VSD. CORONARY ANOMALIESNormal Similar to TOF- LAD from RCA Similar to TGA- RCA from the right posterior aortic cusp & LCA from the left posterior cusp Anomalous origin of the left Cx from RCA, single coronary ostium, origin of right Cx from LCA. DORV – MAJOR CLINICAL PATERNS Group 1 - Sub aortic VSD with PS (resembles TOF) Group 2-Sub pulmonary VSD, with or without PS (resembles TGA). Group 3-Sub aortic VSD, no PS, (resembles VSD) Group4- Sub aortic VSD with PVOD (Resembles Eisenmenger– complex SUB AORTIC VSD WITH PS 50 % of DORV with subaortic VSD have PS RV LV aorta aorta Aortic saturation decreased PBF decreased Resemble TOF SUBPULMONARY VSD LV pulmonary trunk RV aorta Pulmonary saturation > Aortic If PVR decreased good aortic saturation; LV volume overload If PVR increases Blood from LV and RV Aortic saturation decreases. Aorta SUBAORTIC VSD WITH DECREASE PVR WITH NO PS - Resembles VSD - PBF increased -Aortic saturation is normal. SUBAORTIC VSD – INCREASE PVR -Increased blood to aorta -Decreased flow to pulmonary trunk -Aortic saturation falls GROUP 1: SUBAORTIC VENTRICULAR SEPTAL DEFECT AND PULMONARY STENOSIS Features similar to TOF PS is severe- early cyanosis, failure to thrive, exertional dyspnea, squatting, polycythemia Cyanosis clubbing may be evident. GROUP 1: SUBAORTIC VENTRICULAR SEPTAL DEFECT AND PULMONARY STENOSIS.. The precordium show evidence of a right ventricular impulse at the left sternal border, and a prominent systolic thrill upper left sternal border. Grade 4 to 5/6 systolic ejection murmur, which radiates into the lung fields The first heart sound is normal, and the second heart sound is usually single. GROUP 2: SUBPULMONARY VENTRICULAR SEPTAL DEFECT Features resembling those in TGA with VSD. These patients present with cyanosis and heart failure in early infancy. When PS is also present, the cyanosis and polycythemia may be more severe GROUP 2: SUBPULMONARY VENTRICULAR SEPTAL DEFECT.. A precordial bulge and right ventricular impulse are present at the left sternal border. A grade 2 to 3/6 high-pitched systolic murmur may be present at the upper left sternal border. . GROUP 2: SUBPULMONARY VENTRICULAR SEPTAL DEFECT.. When PS is present, a systolic thrill may be present, and the murmur is loud (grade 3 to 4/6). The second heart sound is loud and single because of the proximity of the aorta to the chest wall . With increased pulmonary flow, an apical diastolic rumble may be present. GROUP 3: SUBAORTIC VENTRICULAR SEPTAL DEFECT WITHOUT PULMONARY STENOSIS Patients present features typical of those with a large VSD and pulmonary hypertension. Usually, little cyanosis is evident, but failure to thrive and heart failure are dominant features. With increased pulmonary flow, respiratory tract infections are frequent. GROUP 3: SUBAORTIC VENTRICULAR SEPTAL DEFECT WITHOUT PULMONARY STENOSIS.. A systolic thrill may be present at the upper left sternal border, and a grade 3 to 4/6 holosystolic murmur may be evident at the left sternal border. An apical diastolic rumble and a third heart sound are audible at the cardiac apex. GROUP 4: SUBAORTIC VENTRICULAR SEPTAL DEFECT WITH PULMONARY VASCULAR OBSTRUCTIVE DISEASE Pulmonary flow is reduced, and heart failure and frequent respiratory infections are less evident. Cyanosis and clubbing may be present. GROUP 4: SUBAORTIC VENTRICULAR SEPTAL DEFECT WITH PULMONARY VASCULAR OBSTRUCTIVE DISEASE On examination, The systolic murmur may be diminished or absent The second sound is very loud and single. A decrescendo diastolic murmur of pulmonary valve insufficiency may be present. ELECTROCARDIOGRAPHIC FEATURES Right ventricular hypertrophy and right-axis deviation are the most common features Combined ventricular hypertrophy - markedly increased pulmonary flow (observed in patients with subpulmonary VSD. First-degree AV conduction delay is a common feature; however, it is not uniformly observed. ELECTROCARDIOGRAPHIC FEATURES Right atrial enlargement -patients with PS left atrial enlargement may be observed in instances of increased pulmonary flow with intact atrial septum. Patients with complete AV septal defect associated with DORV also typically have left axis deviation, combined ventricular hypertrophy, atrial enlargement, and firstdegree AV conduction delay . ECG showing deep S wave in V5-6 indicating right ventricular hypertrophy. Biventricular hypertrophy is manifested by large RS complexes in leads V3-6, in a case of DORV, subaortic VSD. ECG showing peaked right atrial P waves in leads2 and V1. q waves appear in lead 1 and aVL despite right axis deviation. Right ventricular hypertrophy manifested by tall R waves in leads V1 and aVR. The qR pattern in leads V5-6 indicates that left ventricle is well developed, in a case of DORV with subaortic VSD and severe PS. ECG Distinguishing points from TOF Counter clockwise initial force with q waves in leads I & aVL, even when the axis is vertical or rightward. Deep & prolonged terminal force with broad, slurred S waves in leads I, aVL & V5-V6 and broad R wave in lead aVR. CHEST X RAY In patients with PS Features may resemble TOF Mild degree of cardiomegaly Pulmonary vascularity is diminished MPA segment is absent, which results in a concave upper left border of the heart DORV subaortic VSD pulmonar y atresia In cases of subaortic VSD without PS Generalized cardiomegaly Prominent main pulmonary artery segment Increased pulmonary vascularity DORV with subaortic VSD Taussig Bing anomaly When PVR is high Prominent MPA peripheral pruning ECHO 4 OBSERVATIONS NOTED FOR DIAGNOSIS Origin of one great vessel from the RV and overlie of at least 50% or more of the other great artery over the RV Mitral- semilunar discontinuity with conus Absence of LVOT other than VSD Spatial relationship of great arteries determined by bifurcation of pulmonary artery and branching of aorta. DORV: Aortic-mitral Discontinuity RV Ao LV LA Aortic - mitral discontinuity in DORV is best shown in the parasternal long-axial view. DORV with subaortic VSD DORV with sub pulmonic VSD DORV with doubly committed VSD DORV with remote VSD DORV with remote VSD Echo Evaluation of DORV: Role of Echocardiography in Planning Surgical Strategy: The crucial question: IS TWO VENTRICLE REPAIR FEASIBLE? Two ventricle repair is preferred over the single ventricle (Fontan) option whenever feasible: Better long term survival Less arrhythmias Better functional capacity Role of Echocardiography in Planning Surgical Strategy: Criteria for Two Ventricle Repair ○ Two good-sized ventricles ○ No straddling of either of the AV valves ○ The VSD should be suitably located for intra-ventricular re-routing ○ No significant AV valve tissue in the way between VSD and the aorta Echo Evaluation of DORV: Suitability for two-ventricle repair Good separation between Pulm. and Tricusp. Valves: Separation between Pulm. and Tricusp. Valves < Ao annulus Minimum separation between Pulm. and Tricusp. Valves Repair of DORV with remote VSD requires creation of a complex intraventricular baffle using multiple patches (the Barbero-Marciel technique) Echo Evaluation of DORV: Two Ventricle Repair Candidates- Additional Issues - Branch PAs - Origin of LAD in the TOF type of DORV - The origin of coronary arteries in the Taussig-Bing anomaly prior to an arterial switch operation - Additional muscular VSDs which may require to be closed at the time of surgery - Other associated anomalies Echo Evaluation of DORV Prior to Single Ventricle Repair Echo complements cardiac catheterization Ventricular function Presence and severity of AV valve regurgitation Size of the branch pulmonary arteries Peripheral pulmonic stenosis if any Severity of pulmonic stenosis (indirect estimate of PA pressures) CARDIAC CATHETERISATION OBJECTIVES: to evaluate • Routability of VSD to aorta • Branch PA anatomy Pulmonary vascular resistance Coronary artery and aortic arch anatomy. Natural History 1 -Infant without PS may develop severe CHF -Later PVOD -Spontaneous closure of VSD –fatal,rare 2 –When PS present ,complications of CCHD (Polycythemia,CVA) Natural history 3 -Taussig Bing –severe PVOD develop early as in TGA 4 -Associated anomalies –COA - LV hypoplasia - poor prognosis Management Includes Medical care and surgical management Optimize medical treatment before surgical intervention Surgical – palliative or definitive Thorough evaluation important before any plan is made MEDICAL MANAGEMENT Inadequate pulmonary blood flow – maintain ductal patency. Prostaglandin E1(ie, alprostadil) is the standard of care until repair can take place. MEDICAL MANAGEMENT Clinical picture of congestive heart failure – diuresis, inotropic support When double outlet right ventricle repair is planned – review : PS or PAH Single or Two ventricle repair Relationship of great vessels Location of the ventriculoseptal defect (VSD) and its size Associated lesions Accurately determine surgically relevant features : Separation of the pulmonary valve from the tricuspid valve relative to the diameter of the aortic valve annulus Location of the VSD, including degree of involvement of the conal septum Chordal attachments to the conal septum, VSD ridge, and presence of straddling chordae Degree of subpulmonary stenosis and whether it is fixed or dynamic Degree of pulmonary valvar stenosis Coronary anatomy Relative size of the great vessels and their relationship The aortic arch and the presence of coarctation Surgical Care - Palliative As with medical treatment, this approach helps improve the patient's clinical condition, allowing the baby to gain weight to achieve optimal conditions for definitive surgical repair With pulmonary stenosis, a systemic-topulmonary shunt, such as a modified BlalockTaussig shunt, may be required if SpO2< 70% Surgical Care - Palliative Variations with increased PBF and CHF– may first require palliation with pulmonary artery banding. NOT RECOMMENDED for infants with Sub Aortic VSD or Doubly Committed VSD. Primary repair is a better choice Palliative Procedures Taussig-Bing type – Balloon /blade atrial septostomy, - for better mixing - for decompression the LA, which causes PV congestion. SURGICAL TREATMENT Analysis of the results of surgery requires consideration of 1. Morphologic subset of this defect 2. Associated congenital heart defect In this analysis the commitment of the VSD to great arteries appears to determine the outcome of repair. SURGERY GOALS OF REPAIR Establish connection from LV to aorta To connect RV to PA To close the VSD Repair of double outlet right ventricle with subaortic ventriculoseptal defect Repair of double outlet right ventricle with a subaortic VSD - intraventricular tunnel. If the VSD is suspected to be smaller than the aorta before or during surgery, the VSD is enlarged. Mortality rate is <5% (subaortic VSD) Repair of double outlet right ventricle with subaortic ventriculoseptal defect and pulmonary stenosis Similar to those with TOF Intraventricular tunnel repair of the VSD similar to that used for patients who have double outlet right ventricle and subaortic VSD without pulmonary stenosis. During intracardiac repair PS may require: - Pulmonary valvotomy - Infundibular resection - Patch enlargement of RVOT Carried out between 6 months and 2 yrs of age. If, coronary artery crosses the right ventricular outflow tract - conduit may be added as an additional outflow path from the right ventricle. Carried out at 3-4 years of age Anatomic repair of double outlet right ventricle with subpulmonary ventriculoseptal defect Taussig-Bing Anomaly: - 3 possible approaches. Early surgery is recommended because of rapid development of Pul vascular obstructive disease. 1. An Intraventricular tunnel between the VSD and PA with the arterial switch operation. Preferred surgical method Mortality 10-15%. Anatomic repair of double outlet right ventricle with subpulmonary ventriculoseptal defect The second method – Consists of construction of a long intraventricular tunnel to establish continuity between the left ventricle and the aorta and between the right ventricle and pulmonary artery. IMPORTANCE OF TRICUSPID TO PULMONARY VALVE DISTANCE 99 A. B. C. Dist > diameter of AV – tunnel can be created post to PV Dist is extremely short – tunnel created to keep PV on LV side Dist < diameter of AV – if tunnel is created post to PV, subaortic stenosis will develop. Tunnel created ant to PV The third method - involves closure of the VSD with baffling of the left ventricular outflow to the pulmonary artery with a subsequent atrial baffle (eg, Senning procedure, Mustard procedure). This method is associated with high operative and late mortality rates. Because coarctation of the aorta is commonly observed in this situation, patients may have undergone coarctation repair with a pulmonary artery band The subsequent procedure is a single stage complete repair with VSD enlargement if restrictive, repair of the VSD to direct the left ventricular blood to the pulmonary artery, followed by an arterial switch procedure. Aortic arch obstruction - repaired at the same time under hypothermic circulatory arrest Repair of double outlet right ventricle with doubly committed ventriculoseptal defect Surgical correction - similar to that described for double outlet right ventricle with subaortic VSD. The VSD, which is typically large, usually does not create difficulty in channeling left ventricular blood to the aorta with an intraventricular tunnel. Concurrent pulmonary stenosis or obstruction of the right ventricular outflow tract due to the tunnel may necessitate the creation of a right ventricle outflow patch or even a right ventricle–to–pulmonary artery conduit. . Repair of double outlet right ventricle with noncommitted ventriculoseptal defect Most difficult to correct. High-risk procedure that often involves univentricular repair. However, biventricular repair of double outlet right ventricle with noncommitted VSD has been described and done The major feature of this anomaly is a persistent subaortic conus and a double infundibulum. Repair of double outlet right ventricle with noncommitted ventriculoseptal defect The subaortic conus is in excess to essentially normal right ventricular structures. Represents malposition of the aorta, with a normally positioned pulmonary artery and with the great vessels usually side by side. The VSD, usually perimembranous, often has inlet and/or trabecular extension and can be restrictive. Copyright ©1999 The American Association for Thoracic Surgery Contraindications to performing a biventricular repair Significant left ventricular hypoplasia Straddling of the atrioventricular valve Timing for Univentricular repair • If the child is stable, mildly cyanosedDirect Fontan operation may be done at 3-4 years of age (Class I) Stable, mildly cyanosed: Glenn at 1 year. Fontan at 3-4 year (Class IIa) Timing for Univentricular repair • • Significant cyanosis (SpO2< 70%) < 6 monthSystemic to pulmonary artery shunt followed by Glenn at 9 months- 1 year, and Fontan at 3-4 years (ClassI). Significant cyanosis (SpO2 <70%) > 6 monthsBD Glenn followed by Fontan at 3-4 years (ClassI). POTENTIAL DANGER AREAS DURING SURGICAL CORRECTION STATUS AFTER REPAIR Most patient surviving repair are NYHA class I Early primary repair improve outcome MAJOR CONCERNS AFTER REPAIR PROGRESSIVE VENTRICULAR DYSFUNCTION. Associated factors like Residual VSD AV valve insufficiency Prolonged circulatory arrest at intracardiac repair. MAJOR CONCERNS AFTER REPAIR LV Outflow Obstruction due to proliferation of fibrous tissue at VSD, distortion of intraventricular baffle, or hypoplasia of the aortic annulus. Late post op Arrhythmia and Sudden Cardiac Death - Associated factors like repair at older age -Ventricular Dysfunction - Increased PA pressure. ANATOMIC FEATURES AND SURGICAL STRATEGIES IN DOUBLE-OUTLET RIGHT VENTRICLE Records were reviewed of all children with DORV undergoing surgery between 1978 and 1993 Biventricular repair can be achieved in most patients with DORV with low risk In complex DORV, a Fontan procedure is associated with a lower surgical mortality Circulation. 1997; 96: 1233-1239 doi: 10.1161/01.CIR.96.4.1233 FOLLOW UP Long term regular follow up 6-12 month interval Subaortic VSD without PS – excellent long term outcome FOLLOW UP -Ventricular arrythmia should be treated - may cause sudden death -20% require reoperation of intraventricular tunnel -Continue SBE prophylaxis Consultations Geneticist to discuss the possibility of subsequent children having this or other forms of CHD. When CHD is detected, a detailed investigation for extracardiac malformation should be performed and vice versa. Also, issues such as preterm birth and stillbirth should be taken into account in risk assessment and risk stratification in patients born with CHD. Most prevalent extracardiac anomalies associated with conotruncal heart defects are anomalies of the GI and genitourinary systems. Associations with DORV – Omphalocele , gastroschisis, facial clefting. Preterm infants have been shown to have more than twice as many cardiovascular malformations as do term infants 16% of all infants with cardiovascular malformations are preterm. Prevalence of CHD is high among late stillbirths. In particular, a greater incidence of coarctation of the AO , double-inlet left ventricle, hypoplastic left heart, truncus arteriosus, DORV, and AV septal defect is noted among stillbirths.