* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Maxwell Relations
Conservation of energy wikipedia , lookup
Heat transfer wikipedia , lookup
Heat capacity wikipedia , lookup
Equipartition theorem wikipedia , lookup
State of matter wikipedia , lookup
Thermal conduction wikipedia , lookup
Calorimetry wikipedia , lookup
Temperature wikipedia , lookup
Heat equation wikipedia , lookup
Maximum entropy thermodynamics wikipedia , lookup
First law of thermodynamics wikipedia , lookup
Heat transfer physics wikipedia , lookup
Entropy in thermodynamics and information theory wikipedia , lookup
Extremal principles in non-equilibrium thermodynamics wikipedia , lookup
Non-equilibrium thermodynamics wikipedia , lookup
Internal energy wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Equation of state wikipedia , lookup
Second law of thermodynamics wikipedia , lookup
Van der Waals equation wikipedia , lookup
James Clerk Maxwell wikipedia , lookup
Adiabatic process wikipedia , lookup
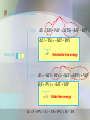
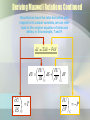
MAXWELL’S THERMODYNAMIC RELATIONSHIPS AND THEIR APPLICATIONS Submitted By Sarvpreet Kaur Associate Professor Department of Physics GCG-11, Chandigarh James Clerk Maxwell (1831-1879) http://en.wikipedia.org/wiki/James_Clerk _Maxwell • Born in Edinburgh, Scotland • Physicist well-known for his work in electromagnetism and field theory • Also known for his work in thermodynamics and kinetic theory of gases Why Use Maxwell Relations? • Certain variables in thermodynamics are hard to measure experimentally such as entropy • Some variables like Pressure, Temperature are easily measureable • Maxwell relations provide a way to exchange variables Maxwell relations derived by the method based on Thermodynamic Potentials Why are thermodynamic potentials useful Thermodynamic potentials give the complete knowledge about any thermodynamic system at equilibrium e.g. U=U(T,V) does not give complete knowledge of the system and requires in addition P=P(T,V) equation of state U=U(T,V) and P=P(T,V) complete knowledge of equilibrium properties U(T,V) is not a thermodynamic potential However We are going to show: U=U(S,V) complete knowledge of equilibrium properties U(S,V): thermodynamic potential The thermodynamic potential U=U(S,V) Now Consider first law in differential notation dU dQ dW dQ TdS dQ inexact differentials expressed by dW dU TdS PdV So dU is an exact potental. 2nd law exact differentials dW PdV Note: exact refers here to the coordinate differentials dS and dV. TdS and PdV are inexact By Legendre transformation (T,V): dU TdS PdV d(TS) SdT PdV d(U TS) SdT PdV from (S,V) : F to (T,P): Helmholtz free energy dF SdT PdV SdT d(PV ) VdP d(F PV ) SdT VdP : G Gibbs free energy G F PV U TS PV H TS dU TdS dPV VdP easy check: Product rule dPV VdP VdP PdV VdP PdV dU dPV TdS VdP dU PV TdS VdP =:H (enthalpy) Enthalpy H=H(S,P) is a thermodynamic potential dH TdS VdP Now dU, dF, dG and dH are exact differentials e.g Internal Energy dU TdS PdV Helmholtz Free Energy dF d (U TS ) SdT PdV Gibb' s Function dG d ( F PV ) SdT VdP Enthalpy dH TdS VdP Using these exact differentials we derive maxwell’s relations . Maxwell’s Thermodynamic Relations T P V S S V S P V T T V T V P S S P S V P T T P Deriving Maxwell Relations Using thermodynamic Potentials First, start with a known equation of state such as that of internal energy Next, take the total derivative of with respect to the natural variables. For example, the natural of internal energy are entropy and volume. U U dU dS dV S V V S Deriving Maxwell Relations Continued Now that we have the total derivative with respect to its natural variables, we can refer back to the original equation of state and define, in this example, T and P. U U dU dS dV S V V S U T S V U P V S Deriving Maxwell Relations Continued We must now take into account a rule in partial derivatives When taking the partial derivative again, we can set both sides equal and thus, we have derived a Maxwell Relation Similarily using dF,dG and dH other Maxwell Relations are Mnemonic Device for Obtaining Maxwell Relations T P V S Write T,V,S,P in a clockwise manner by Remembering the line TV Special Programme.. •Four relations are obtained by starting either from T or S Clockwise or anticlockwise direction. A negative Sign must appear in the resulting equation Using Maxwell Relations Maxwell Relations can be derived from basic equations of state, and by using Maxwell Relations, working equations can be derived and used when dealing with experimental data. Application of Maxwell’s Relations The Four Maxwell relations have a very wide range of applications . They apply to all kind of substances (solids,liquids,gases)under all type of conditions of Pressure, volume and temperature. Before Discussing applications We define some thermodynamic terms e.g i) Specific Heat at Constant Volume ii) Specific Heat at Constant Pressure U Cv T v U CP T p iii) Pressure and Volume Coffecient of Expansion 1. Cooling Produced By Adiabatic Expansion of Any Substance 1. Cooling Produced By Adiabatic Expansion of Any Substance In adiabatic process entropy S remains constant. Therefore by considering the Thermodynamic relation T V We can prove p S S V T Tp mCv Most of the substances expand on heating , they have +ve beta value. T Will be –ve i.e all the substances will cool down. A few substances like rubber have –ve beta value. They will get heat up.. 2. Adiabatic Compression of A Substance By considering the Thermodynamic relation T V S p S We can prove p TV T C p Above result shows that if is +ve, then adiabatic increase in pressure causes the temprature to rise. ** Similarily using other maxwell’s equations we can explain the stretching of wires and thin films 3. Change of internal energy with Volume, Using the third Maxwell’s relation s P v T T v u P T P v T T v Since 19 For Ideal Gas This result helps to show that the internal energy of an ideal gas does not depend upon specific volume. This is known as Joule’s Law. For Vander Waal’s / real gases ( p a )(V b ) RT 2 V U V T a V 2 V Thus Vander Waal’s gas expands isothermally as its internal energy increases. 20 4. Cp – Cv = R for ideal gases. Other relations for the specific heats are given below. where is the volume expansivity and is the isothermal compressibility, defined as 21 The difference Cp – Cv is equal to R for ideal gases and to zero for incompressible substances (v = constant). 5. Variation of Cv with specific volume. 5. Variation of Cv of an ideal gas does not depend upon specific volume. For an ideal gas 22 Therefore, the specific heat at constant volume of an ideal gas is independent of specific volume. For Vander Waal’s gas also it is independent of volume. 23 6. Change of state and clapeyron’s equation In ordinary phase transition of matter(solid phase to liquid phase, liquid to vapour, and solid to vapour) take place under constant Temperature and pressure. During the transition a certain amount of heat, known as latent heat must be supplied to the substance for a change Of phase. During this change temperature remains constant. Therefore using maxwell relation S p V T T V using ΔU L, the latent heat, Wehave p T V L T (V f Vi ) This equation is known as Clausius-Clapeyron’s latent heat Thank You