* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download presented - EPP Group
Survey
Document related concepts
Transcript
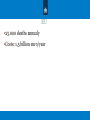
Antimicrobial resistance “One health fits all” Anja Schreijer, MD, PhD, MPH Senior policy advisor AMR Centre for Infectious Disease Control RIVM EU 25.000 deaths annualy Costs: 1,5 billion euro/year WHO draft global action plan Five strategic objectives: 1. to improve awareness and understanding of antimicrobial resistance; 2. to strengthen knowledge through surveillance and research; 3. to reduce the incidence of infection; 4. to optimize the use of antimicrobial agents; 5. to develop the economic case for sustainable investment that takes account of the needs of all countries, and increase investment in new medicines, diagnostic tools, vaccines and other interventions. (to be presented at WHA May 2015) Identifies 7 areas in which actions are most necessary: - making sure antimicrobials are used appropriately in both humans and animals - preventing microbial infections and their spread - developing new effective antimicrobials or alternatives for treatment - cooperating with international partners to contain the risks of AMR - improving monitoring and surveillance in human and animal medicine - promoting research and innovation - improving communication, education and training. Transmission of resistant microorganisms One health approach: 8 intersectoral surveillance Specific objectives of AMR surveillance & AB use Monitor – – – – Antibiotic-susceptibility patterns common pathogens Magnitude and trends of AMR Emergence of new AMR Use/overuse/misuse of antibiotics In order to – Improve quality, safety and costs of health care • Guidance adequate antibiotic therapy – appropriate use • Support infection prevention & control – Awareness & advocacy – Benchmarking – Improve laboratory capacity, expertise & quality 9 No surveillance no control.. 1 Use of antibiotics in Europe 1 (humans) Use of antibiotics in Europe (animals) Intersectoral Coordination: Ministry of Health, Welfare and Sports & Ministry of Economic Affairs (Agriculture and Livestock) - Health Council of the Netherlands report (2011): Antibiotics in food animal production and resistant bacteria in humans. - Preventive use was prohibited - Restricted use of all critically important antibiotics (3rd choice) - Last resort antibiotics excluded from veterinary use - Mandatory sensibility testing 3rd choice antibiotics - Independent Veterinary Drug Authority (2011): Monitor trends + benchmarking + transparency - Amendment to the law (2013): Antibiotics only with prescription from veterinarian - Guidelines AMR control Infection control Appropriate use of antibiotics Control of AMR: infection control Infection prevention & hygiene Professional guidelines for each setting Search and destroy or control/contain Awareness and sense of urgency Outbreakmanagement Enforcement by the national health inspectorate Problem ownership/responsibilities Control of AMR -> Appropriate use of antibiotics ● Right indication, right dose, adequate period, at the lowest cost, good quality ● Treatment is correctly followed by the patient. ● Bacteria causing the infection need to be susceptible. ● ● ● Inappropriate use includes over-prescription, under prescription, and prescription and dispensing of unnecessary antibiotic combination (From: The evolving threat of antimicrobial resistance - Options for action, WHO 2012) Control: surveillance, awareness, problem ownership, responsibilities, professional guidelines, antibiotic stewardship, enforcement Control of AMR: raising awareness TAP: when knowledge is not enough Guide to Tailoring AMR Programmes Design tailored interventions instead of “spray campaigns” Describes an step by step approach and provides tools to assist national AMR programmes to increase prudent antibiotic use Based on TIP (Guide to Tailoring Immunization programmes) TAP is a conceptual model that combines several behavioural change theories, social marketing concepts with previous experience. TAP brings together several stakeholders from the start to gain ownership and to recognize the work that has to be done together in order to work towards the intervention together Current state: in development/pilot fase → Sweden, England, Netherlands AMR Dutch EU presidency ● ● ● ● NL involvement in WHO GAP and GHSA (one of the leading countries) Minister of Health announced at the 3rd International One Health Congress in Amsterdam (March 2015): AMR will be one of my priorities during the Dutch EU presidency first half 2016 Focus on One Health approach to AMR; measures needed in human health sector, animals, food and environment Further recommendations for the next EU AMR Action plan Conclusions: - - No action today no cure tomorrow No surveillance no control of AMR and use of antibiotics AMR control consists of infection prevention and appropriate us of antibiotics Knowledge is not enough for behavior change One health fits all (better safe then sorry) Questions?