* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Light Sources and Interaction of Light With Matter
Survey
Document related concepts
Transcript
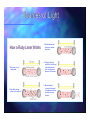
“Light Sources and Interaction of Light With Matter” IST 8A Lecture #3 - Jan. 12, 2005 M. Molinaro, F. Chuang IST 8A (Shedding Light on Life) - W05 Lawrence Livermore National Laboratory Sources of Light What do you think are some of the sources of light? Sources of Light Incandescence: Incandescence: light emission by thermal radiation of a temperature high enough to render the source of radiation visible. Electroluminescence: Electroluminescence: light-emitting diode (LED) Laser: Laser: light amplification through the stimulated emission of radiation Fluorescence: Fluorescence: light emission resulting from absorption of shorter-wavelength electromagnetic radiation. Persists for less than 10-8 sec after excitation. Phosphorescence: Phosphorescence: similar to fluorescence, but persisting longer than 10-8 sec following excitation. ChemiChemi- and Bio-luminescence: Bio-luminescence: e.g., the firefly Blackbody radiation: radiation: “pure” pure” form of incandescence Sources of Light - Incandescence What is it and how does it work? Incandescence Incandescence is the release of electromagnetic radiation from a hot body due to its high temperature. The release of radiation is usually in the infrared (heat (heat)) and the visible region of the electromagnetic spectrum (we perceive as light.) Incandescence occurs in both light bulbs where the electrical resistance to electron flow excites electrons in the material to jump to a higher orbit, and thus subsequently release a photon; and when something is on fire or during an explosive or a combustion reaction. X-rays can also be produced by incandescence, but often most energy is wasted in the form of heat. Tungsten bulb glows at 2700K. Range of wavelengths produced IR and visible Energy efficiency: about 90% given off as heat http://en.wikipedia.org/wiki/Incandescence http://en.wikipedia.org/wiki/Incandescent_light_bulb http://www.gwebdesign.de/company-en.html Sources of Light - Electroluminescence What is it and how does it work? Electroluminescence is an optical phenomenon and electrical phenomenon where a material such as a natural blue diamond emits light when an electric current is passed through it.Electroluminescence (EL) is mainly observed in semiconductors. semiconductors. The electric field excites electrons in the material which then emit the excess energy as photons. LEDs are the most well known example of electroluminescence. Range of wavelengths produced (LED) is a semiconductor device that emits incoherent monochromatic light. light. The wavelength of the light emitted depends on the bandgap energy of the materials forming the pn junction. The materials used for an LED have bandgap energies corresponding to near-infrared, visible or near-ultraviolet light. Energy efficiency: 2 to 4 times more efficient than incandescent - less heat! Demo: RGB LED http://www.kolej.pl/modele/JTZ/obwody/swiatlo.html http://en.wikipedia.org/wiki/Electroluminescence http://en.wikipedia.org/wiki/LED http://www.fortrantraffic.com/signals/led/LED.htm Sources of Light LASER = Light Amplification through Stimulated Emission of Radiation Monochromatic Coherent http://science.howstuffworks .com/laser.htm htm http://science.howstuffworks.com/laser. Directional Can focus to few hundred nm, high power Range from UV to Visible to IR + some are tunable Can pulse down to femtoseconds http://en.wikipedia.org/wiki/Laser Sources of Light How a Ruby Laser Works 1. Ruby laser in nonlasing state 2. Light flash excites atoms in the ruby rod 3. Excited atoms emit photons in random directions 4. Photons traveling parallel to rod axis get reflected back and forth, exciting more atoms in the process 5. Monochromatic, coherent, directional laser beam exits from partially mirrored surface Sources of Light - Fluorescence What is it and how does it work? Fluorescence is the property of some atoms and molecules to absorb light at a particular wavelength and to subsequently emit light of longer wavelength after a brief interval, termed the fluorescence lifetime. The process of phosphorescence occurs in a manner similar to fluorescence, but with a much longer excited state lifetime Range of wavelengths produced Usually the fluorescent materials are “excited” excited” by shining UV light and they re-emit in the visible and infra-red. Energy efficiency: 4 times more efficient than incandescent. Low heat! Inside the glass tube is a partial vacuum and a small amount of mercury. An electric discharge in the tube causes the mercury atoms to emit light. The emitted light is in the ultraviolet range and is invisible, and also harmful to living organisms, so the tube is lined with a coating of a fluorescent material, called the phosphor, which absorbs the UV and re-emits visible light. http://en.wikipedia.org/wiki/Fluorescence http://www.geology.wisc.edu/~museum/old/whatis3.html Sources of Light - Fluorescence http://micro.magnet.fsu.edu/primer/lightandcolor/fluorointroduction.html http://www.bris.ac.uk/Depts/Synaptic/info/imaging/imaging_1.htm Sources of Light - Fluorescence Fluorescence: Fluorescence: light emission resulting from absorption of shorter-wavelength electromagnetic radiation. Persists for less than 10-8 sec after excitation. http://micro.magnet.fsu http://micro.magnet.fsu..edu/primer/techniques/fluorescence/excitation.html edu/primer/techniques/fluorescence/excitation.html Sources of Light - Fluorescence Quantum Dots Dr. D. Talapin, University of Hamburg, http://www.chemie.uni-hamburg.de/pc/Weller/ http://www.qdots.com/live/render/content.asp?id=84 Sources of Light - Phosphorescence How it works Phosphorescence is a radiative transition involving a change in the spin multiplicity of a molecule. Because of this change, the radiative transition is delayed, and the phosphorescent material glows a while after the incident illumination stops. Because of this, phosphorescent materials are most commonly called "glow-in-the-dark". http://images.iwoot.com/large/glofri_lg.jpg http://www.valhallaarms.com/technology/lighting_fx.htm http://en.wikipedia.org/wiki/Phosphorescence http://www.olympusfluoview.com/theory/fluoroexciteemit.html Sources of Light - Chemi and Bioluminescence How it works: Chemoluminescence (sometimes "chemiluminescence") is the emission of light (luminescence) as the result of a chemical reaction. Most simply, given A and B, with an excited intermediate ◊, we have, [A] + [B] --> [◊] --> [Products] + light The decay of the excited state[◊] to a lower energy level is responsible for the emission of light. When chemoluminescence takes place in living organisms, the phenomenon is called bioluminescence http://en.wikipedia.org/wiki/Chemoluminescence and Bioluminescence http://www.omniglow.co.uk/inpublisher/ uploaded_images/snaplight_pk_ass.JPG http://www.mbari.org/itd/ retrospective/bioluminescence.html Sources of Light - Chemi and Bioluminescence Luminol : Luminol is used by investigators to locate blood, even if it has been cleaned or removed. Using its natural property of chemiluminesence, it glows if blood is present. The glow lasts for about 30 seconds and is blue. Luminol reacts with the iron in hemoglobin, a pigment present in blood. http://curiouslee.typepad.com/photos/best_of_curiouslee/firefly.html http://en.wikipedia.org/wiki/Luminol Sources of Light - Blackbody http://en.wikipedia.org/wiki/Blackbody How it works: A blackbody is an object that emits a well defined spectrum of radiation solely based on its temperature. Blackbody temperature is the temperature of an object if it is reradiating all the thermal energy that has been added to it; if an object is not a blackbody radiator, it will not reradiate all the excess heat and the leftover will go toward increasing its temperature. Stefan-Boltzmann Stefan-Boltzmann law - overall power emitted per unit area. Wien’ Wien’s law - Defines strongest emission Real objects never behave as full-ideal black bodies, and instead the emitted radiation at a given frequency is a fraction of what the ideal emission would be. http://www.astronomycafe.net/qadir/q72.html Colder Even as the peak wavelength moves into the ultra-violet enough radiation continues to be emitted in the blue wavelenths that the body will continue to appear blue. It will never become invisible. Hotter Sources of Light - IR emission Examples http://coolcosmos.ipac.caltech.edu/image_galleries Demo: Thermometer Sources of Light - Blackbody Interactive Applet Blackbody Interaction of Light with Matter Target material Incoming photon ? ? Q: What happens when photons hit matter..? A: Both light and matter can be affected. Consider both Fates of Light, and Fates of Material..! Interaction of Light with Matter Incoming photon Materials with high electron density, such as metals (i.e., silver, mercury, aluminum) Reflected photon Reflection: The return of radiation by a surface, without change in wavelength. Demo: Laser reflect, measure angles Interaction of Light with Matter Incoming photon Transmitted photon Materials like water, glass, plastic, etc… Transmission: The conduction of radiant energy through a medium. Interaction of Light with Matter Materials like water, glass, plastic, etc… Transmitted photon ray in the denser medium is always closer to the normal Incoming photon Refraction: Change in direction of a wave due to a change in velocity.. The refractive index of a material is the factor by which electromagnetic radiation is slowed down (relative to vacuum) when it travels inside the material. Glass 1.5-1.9, diamond 2.41 http://en.wikipedia.org/wiki/Refraction Snell’s Law: n1sin(θ1) = n2sin(θ2) Demo: Vapor chamber + plexiglas Interaction of Light with Matter Interactive Applet Reflection and Refraction Interaction of Light with Matter Side scatter Incoming photon Forward scatter Back scatter Scattering: The change in spatial distribution of a beam of radiation when it interacts with a surface or heterogeneous medium. The sky blue because molecules in the air preferentially scatter blue light. Scattering varies as a function of the ratio of the particle diameter to the wavelength of the radiation. http://en.wikipedia.org/wiki/Scattering Interaction of Light with Matter Incoming photon ? Diffraction: The T apparent bending and spreading of waves when they meet an obstruction. Diffraction also occurs when any group of waves of a finite size is propagating; for example, a narrow beam of light waves from a laser must, because of diffraction of the beam, eventually diverge into a wider beam at a sufficient distance from the laser. Interaction of Light with Matter Scattering Demo - tape and plastic cup Diffraction Demo - Light through a slit or on a hair Interaction of Light with Matter Interaction of Light with Matter http://www.olympusmicro.com/primer/lightandcolor/diffraction.htm and http://www.olympusmicro.com/primer/java/diffraction/index.htmll Interaction of Light with Matter Incoming photon Energy Absorption: The loss of light as it passes through a material, generally due to its conversion to other energy forms. Transforming Energy Thermal Heat Electrical Electricity Chemical Chemistry Photonic Light Kinetic Motion Interaction of Light with Biomaterials Incoming photon Reflection Absorption Transmission Scattering In complex materials, any combination of interactions are possible. The exact nature of each process depends on the physical and chemical structure of the biomaterial.