* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Optimal ventricular rate slowing during atrial fibrillation - AJP
Coronary artery disease wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Heart failure wikipedia , lookup
Cardiac surgery wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Myocardial infarction wikipedia , lookup
Jatene procedure wikipedia , lookup
Electrocardiography wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Ventricular fibrillation wikipedia , lookup
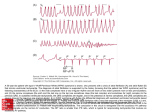
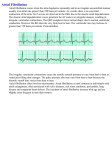
Am J Physiol Heart Circ Physiol 282: H1102–H1110, 2002. First published November 23, 2001; 10.1152/ajpheart.00738.2001. Optimal ventricular rate slowing during atrial fibrillation by feedback AV nodal-selective vagal stimulation YOUHUA ZHANG, KENT A. MOWREY, SHAOWEI ZHUANG, DON W. WALLICK, ZORAN B. POPOVIĆ, AND TODOR N. MAZGALEV Department of Cardiovascular Medicine, The Cleveland Clinic Foundation, Cleveland, Ohio 44195 Received 17 August 2001; accepted in final form 19 November 2001 heart rhythm; arrhythmia; hemodynamics; autonomic control; atrioventricular node ATRIAL FIBRILLATION (AF) has been long recognized as one of the most frequent chronic arrhythmias. Although the restoration and maintenance of normal sinus rhythm is the ultimate goal, it is frequently not achievable. Therefore, ventricular rate (VR) control during AF remains the only realistic long-term solution in a majority of patients (27). Although a VR from 60 to 90 beats/min has been recommended for patients with AF (45, 47), no specific experimental or clinical data are available to support this recommendation (22). The VR-hemodynamics relationship could be determined in several experimental models. In choosing the one implemented in this study, we rejected those that are either associated with marked changes of the nor- Address for reprint requests and other correspondence: T. N. Mazgalev, Research Institute FF1-02, Cleveland Clinic Foundation, 9500 Euclid Ave., Cleveland, OH 44195 (E-mail: [email protected]). H1102 mal physiology, are inherently imprecise, or are difficult to implement. First, the atrioventricular node (AVN) can be destroyed (ablated), followed by implantation of ventricular pacemaker. This approach results in retrograde ventricular activation starting with depolarization of the apex. On the positive side, the pacemaker permits maintenance of any desired constant retrograde VR. Because our scope was limited to physiologically normal anterograde conduction, this method was not investigated in the present study. Second, the AVN conduction function could be depressed by various drugs. This approach preserves the normal anterograde propagation. However, the use of drugs to determine an optimal VR has limitations because the decrease in the VR during treating of AF occurs gradually and unpredictably (22). In addition, most drugs have direct effect on cardiac contractility (31). Third, partial ablation (modification) of the slow AVN pathway has been used to slow the VR. However, a progressive attenuation of the rate by this method is virtually impossible and the procedure frequently results in complete AVN block (10, 25). A fourth, novel, approach has emerged recently. It is well established that stimulation of parasympathetic nerves, which selectively innervate the AVN, can slow AV conduction (2, 6, 12, 13, 21, 28–30). In our recent in vitro study, we found that vagally induced slowing of the VR during AF is feasible by using postganglionic endocardial nerve stimulation (23), whereas similar effects were also obtained by utilizing epicardial (44) or endocardial (36–39) nerve stimulation in vivo. This approach is nondestructive and associated with maintenance of physiological anterograde propagation of the cardiac beat. The vagal effects can be turned on and off almost instantaneously and can be repeated multiple times. We hypothesized that selective atrioventricular node (AVN) vagal stimulation (AVN-VS), by varying the nerve stimulation intensity, could achieve precise graded VR slowing and thus permit evaluation of the optimal VR during AF. Accordingly, the aims of the The costs of publication of this article were defrayed in part by the payment of page charges. The article must therefore be hereby marked ‘‘advertisement’’ in accordance with 18 U.S.C. Section 1734 solely to indicate this fact. 0363-6135/02 $5.00 Copyright © 2002 the American Physiological Society http://www.ajpheart.org Downloaded from http://ajpheart.physiology.org/ by 10.220.32.246 on May 3, 2017 Zhang, Youhua, Kent A. Mowrey, Shaowei Zhuang, Don W. Wallick, Zoran B. Popović, and Todor N. Mazgalev. Optimal ventricular rate slowing during atrial fibrillation by AV nodal-selective vagal stimulation. Am J Physiol Heart Circ Physiol 282: H1102–H1110, 2002. First published November 23, 2001; 10.1152/ajpheart.00738. 2001.—Although the beneficial effects of ventricular rate (VR) slowing during atrial fibrillation (AF) are axiomatic, the precise relationship between VR and hemodynamics has not been determined. We hypothesized that selective atrioventricular node (AVN) vagal stimulation (AVN-VS) by varying the nerve stimulation intensity could achieve precise graded slowing and permit evaluation of an optimal VR during AF. The aims of the present study were the following: 1) to develop a method for computerized vagally controlled VR slowing during AF, 2) to determine the hemodynamic changes at each level of VR slowing, and 3) to establish the optimal anterograde VR during AF. AVN-VS was delivered to the epicardial fat pad that projects parasympathetic nerve fibers to the AVN in 14 dogs. Four target average VR levels, corresponding to 75%, 100%, 125%, and 150% of the sinus cycle length (SCL), were achieved by computer feedback algorithm. VR slowing resulted in improved hemodynamics and polynomial fit analysis found an optimum for the cardiac output at VR slowing of 87% SCL. We conclude that this novel method can be used to maintain slow anterograde conduction with best hemodynamics during AF. H1103 OPTIMAL VENTRICULAR RATE IN AF present study were the following: 1) to develop a method for computerized vagally controlled VR slowing during AF, 2) to determine the hemodynamic changes at each level of VR slowing, and 3) to establish the optimal anterograde VR during AF. METHODS AJP-Heart Circ Physiol • VOL feedback ⫽ pgain 䡠 error ⫹ igain 䡠 兰 error 䡠 dt ⫹ dgain 䡠 d共error兲 dt where pgain is the proportional correction, igain is the integral correction, and dgain is the derivative correction. The first component determined the feedback amplification based on the absolute value of the difference between TRR and each current ARR. The second component adjusted the feedback amplification based on the cumulative value of the error over all preceding beats. The third component was included to account for the specific dynamics of R-R changes. The “igain” was the major source of control. In most cases, we used igain ⫽ 10,000, with the “pgain” and the “dgain” being set to zero. This PID1 feedback voltage (range ⫾ 10 V) was used to determine the instantaneous amplitude of the impulses applied for VS. Specifically, bursts containing 20 pulses (6 ms apart, 1 ms in duration) were synchronized with the right ventricular apex electrogram (Fig. 1). The polarity of each consecutive pulse in a burst was alternated to diminish any polarization effects. The impulses were delivered to the animal via optically isolated constant-current devices (Isolator10, Axon Instrument). Study protocol. The experiments consisted of three steps. First, after the surgical preparation and at least 30 min of stabilization, all electrical and hemodynamic data (surface ECG, right atrial and right ventricular epicardial electrograms, blood pressures, and AoF) were collected during sinus rhythm as the baseline values. During the data collection period, the sinus cycle length (SCL) was determined by averaging 100 consecutive cycles by a computer. Second, AF was induced by rapid right atrial pacing (20 Hz, 1 ms) and thereafter maintained by subthreshold stimulation of the right pulmonary vein-atrial junction fat pad (selective VS to the sinus node and surrounding atrium). The latter stimula- Fig. 1. A right ventricular apex (RVA) complex (top trace) triggers the delivery of 20 impulses (bottom trace) of vagal stimulation (VS) stimuli (6 ms apart, 1 ms in duration, and the polarity is alternated to diminish any polarization effect). The amplitude of atrioventricular node (AVN)-VS impulses is determined with the use of computer feedback control program (see METHODS). 282 • MARCH 2002 • www.ajpheart.org Downloaded from http://ajpheart.physiology.org/ by 10.220.32.246 on May 3, 2017 The study was approved by the Institutional Animal Research Committee and is in compliance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Surgical preparation. Fourteen adult mongrel dogs (body wt 21–30 kg) were premedicated with thiopental sodium (20 mg/kg iv) and intubated and ventilated with room air supplemented with oxygen as needed to maintain normal arterial blood gases by a respirator (NARKOMED 2, North American Drager; Telford, PA). Anesthesia was then maintained with 1–2% isoflurane throughout the experiment. The left external jugular vein was cannulated to infuse normal saline at 100–200 ml/h to replace spontaneous fluid loses. Standard surface electrocardiogram (ECG) leads (I, II, and III) were continuously monitored. Intermittent arterial blood gas measurements were taken and ventilator adjustments were made to correct any metabolic abnormalities. Body temperature was monitored with a rectal probe (model TM-2400, Electromedics), and an electrical heating pad under the animal and operating-room lamps were used to maintain a body temperature of 36°-37° C. The right femoral artery was cannulated, and a micromanometer-tipped catheter pressure transducer (Millar; Houston, TX) was inserted and advanced to the thoracic aorta to monitor the systemic aortic blood pressure (AoP). For left ventricular (LV) pressure (LVP) measurements, another Millar catheter was inserted through the left carotid artery and advanced so that the tip was in the left ventricle. Before the insertion, both Millar catheters were soaked in warm saline for 30 min and precalibrated. After the chest was opened through a median sternotomy, a pericardium cradle was created to support the heart. Custom-made Ag-AgCl quadripolar plate electrodes were sutured to the high right atrium and right ventricular apex for bipolar pacing and recording. Similar bipolar plate electrodes were also sutured to two epicardial fat pads that contain parasympathetic neural pathways selectively innervating the sinus node (SN) and the AVN, the right pulmonary vein-atrial junction fat pad, and the inferior vena cava-left atrium fat pad, respectively (44). The ascending aorta was isolated, and a flow probe was placed around it and connected to a flowmeter (16A/20A, HT 207, Transonic System; Ithaca, NY) to display and measure aortic flow (AoF). All signals (surface ECG, right atrial and right ventricular electrocardiogram, aortic blood pressure, LVP, and AoF signals) were amplified, filtered, and displayed on our recording system (GE Marquette Medical Systems). In addition these signals along with calibrations were simultaneously recorded on magnetic tape (model 400A; Vetter Digital) for later computer analysis with AxoScope (Axon Instruments) and a custom-written software program. Delivery of selective AV nodal vagal nerve stimulation. To achieve different levels of VR slowing, a computer-controlled feedback program was developed. The program was personal computer based using an analog-to-digital (A/D) board from Microstar Laboratories (Bellevue, WA). Data acquisition and stimulation output were controlled from Microsoft Excel via a Microsoft Visual Basic interface with the A/D board. All desired measurements were written to the spreadsheet in real time. Development was centered on an existing Mi- crostar command (PID1) that implemented a classic, proportional-integral derivative, closed loop process control. Specifically, a R-R value was defined as a target ventricular interval (TRR), such as the R-R interval during spontaneous sinus rhythm. The actual R-R interval (ARR) was measured in real time and compared with the TRR. The difference between the two was the control error. The PID1 command would then compute the feedback necessary to move the measured R-R toward the TRR. For example, if TRR ⬎ ARR in the current beat, the amplitude of AVN-VS was increased during the next beat, and vice versa. The following algorithm was used H1104 OPTIMAL VENTRICULAR RATE IN AF Y ⫽ Y avg ⫹ 共b1 ⫻ 1兲 ⫹ 共b2 ⫻ 2兲 ⫹ · · · ⫹ 共bn ⫻ n兲 where Yavg is the average value obtained with all levels of VR slowing, n is the order of fitted polynomial, bn is a fitting parameter, and n is determined as n ⫽ 关共%SCL ⫺ 100兲/25兴n ⫹ mn ⫺ 2 ⫻ 关共%SCL ⫺ 100兲/25兴n ⫺ 2 ⫹ · · · ⫹ mn ⫺ 2k ⫻ 关共%SCL ⫺ 100兲/25兴n ⫺ 2k where m is a scaling factor (42) and k is an integral value ranging from 1 to n/2. The maximum value of Y determined the optimal %SCL. A P value ⬍0.05 was required for statistical significance. RESULTS VR during sinus rhythm, AF alone, and graded VR slowing by AVN-VS. AF normally resulted in irregular and rapid ventricular responses that rendered an average VR substantially faster than the spontaneous sinus rate (the average R-R during AF was 61 ⫾ 10% of the SCL, n ⫽ 14). In 2 of the 14 dogs, however, the average R-R intervals during AF were 78% and 81% of the SCL, respectively. In these two cases, the target of 75% SCL could not be achieved because AVN-VS always prolongs the R-R intervals. In two other dogs, a 150% SCL slowing was not achievable at the maximal available intensity of AVN-VS. These four dogs were excluded from the analysis, and the reported results are based on 10 dogs, in which the average R-R during AF was 58 ⫾ 7% of the SCL and all four target VR levels (faster and slower than the sinus rate) were achieved. The combined averaged VR achieved and maintained in all dogs are shown in Table 1. As expected, compared with sinus rate the VR was significantly faster during AF (125 ⫾ 22 vs. 214 ⫾ 33 beats/min). Computer feedback-assisted AVN-VS resulted in a graded predetermined VR slowing. The feedback control was highly effective, as evident from the closeness of the target and achieved levels (numbers in parentheses in Table 1). Even in the case of the 150% SCL target, the VR was slowed to 86 beats/min, which equals 145% SCL. Figure 2 shows a representative experiment, in which the four different levels of VR are illustrated. In this figure, episodes of 500 R-R intervals at sinus rhythm, AF, and four different levels of AVN-VS were generated as an uninterrupted sequence for illustration purpose. In the real experiment, as previously stated, each episode contained up to 1,000 beats, was followed by a 5-min pause and the order was randomized. It can be seen in Fig. 2 that although the desired average levels of VR slowing were closely achieved, a substantial variability was observed on a beat-to-beat basis. Hemodynamic responses during sinus rhythm, AF alone, and AF with different levels of VR slowing. Figure 3 shows representative hemodynamic traces during AF (Fig. 3A), and VR slowing at 75%, 100%, Table 1. VR during spontaneous sinus rhythm, induced AF, and during AF with 4 target levels of feedbackcontrolled VR slowing by selective AVN-VS in 10 dogs Slowing of VR by AVN-VS during AF Parameter Sinus Rhythm AF 75% SCL 100% SCL 125% SCL 150% SCL 1 2 3 4 5 6 7 R-R interval, ms VR, beats/min 494 ⫾ 87 125 ⫾ 22 287 ⫾ 47 214 ⫾ 33 371 ⫾ 66 (75%) 166 ⫾ 29 488 ⫾ 86 (99%) 126 ⫾ 20 607 ⫾ 108 (123%) 102 ⫾ 18 715 ⫾ 117 (145%) 86 ⫾ 15 Values are means ⫾ SD. AF, atrial fibrillation; AVN, atrioventricular node; VS, vagal stimulation; VR, ventricular rate; SCL, spontaneous sinus cycle length. The achieved average R-R intervals during the AVN-VS slowing are shown in milliseconds as well as a percentage of the SCL (in parentheses) for 4 target levels. P ⬍ 0.001, statistically significant differences were observed between sinus rhythm and the corresponding values shown in columns 3, 4, 6, and 7, as well as between any pair of columns 3 to 7. AJP-Heart Circ Physiol • VOL 282 • MARCH 2002 • www.ajpheart.org Downloaded from http://ajpheart.physiology.org/ by 10.220.32.246 on May 3, 2017 tion utilized continuous sequence of very brief impulses with duration of 50 s, amplitude of 5–10 mA, and frequency of 20 Hz. In several cases, AF was maintained by continuing the rapid right atrial pacing. After the hemodynamics stabilized for at least 15 min, data were recorded during AF for at least 500 cardiac cycles. Third, while maintaining the AF, we initiated the feedback program to deliver the AVN-VS and to slow the VR. Four target levels were preprogrammed that were 75%, 100%, 125%, and 150% of the corresponding spontaneous SCL. The order of the four different levels was randomized. A rest period of 5 min was allowed after each of the AVN-VS periods, although the AF was maintained uninterrupted. Hemodynamic data were collected during up to 1,000 ventricular beats in each period, and beats numbered 200–700 were included in the analysis (this was done to exclude transients at the start of the AVN-VS). Data acquisition and analysis. Tape-recorded data were played back off-line and digitized at 1 kHz per channel by AxoScope (Axon Instrument). For each of the three steps of the protocol, the R-R intervals, systolic blood pressure (SBP) and diastolic blood pressure (DBP) from the AoP signal, LV systolic pressure (LVSP) and LV end-diastolic pressures (LVEDP) from the LVP signal, and stroke volume (SV) were calculated beat-by-beat during 500 beats and averaged by a custom-written software. The SV for individual beats was determined by integrating the AoF signal over the duration of the corresponding R-R interval. The ⫾d(LVP)/dt was derived from LVP, and cardiac output (CO) was calculated by multiplying the heart rate by the SV. The CO determined in this fashion represented the total LV outflow reduced by the coronary flow. Statistical analysis. Data are expressed as means ⫾ SD. Hemodynamic differences during sinus rhythm, AF alone, and AF plus different levels of AVN-VS were evaluated by single factor repeated measurements analysis of variance, followed by post hoc Tukey’s honestly significant difference test. To calculate the optimal average R-R intervals (expressed as %SCL), a contrast analysis was used to detect significant orthogonal polynomial components for each hemodynamic parameter Y (expressed as a function of %SCL) that were fitted to the regression equation (42) OPTIMAL VENTRICULAR RATE IN AF 125%, and 150% SCL (Fig. 3, B–E). Notably, rapid irregular electrical activation of the ventricles during AF resulted in many aborted beats (asterisks in Fig. 3A) along with deterioration of other parameters. The term “aborted beat” (sometimes referred to as “pulse deficit”) means that there was electrical activation but there was no subsequent SV. In such beats, LV contraction resulted in an insufficient LV pressure, less than the diastolic arterial blood pressure level. As a result, the aortic valve did not open and no blood was ejected from the left ventricle. In this study, we considered the beat to be aborted if the electrical activation was followed by ⬍1 ml of SV. With AVN-VS producing progressive VR slowing, these aborted beats were dramatically reduced (asterisks in Fig. 3, B–D) together with the improvement of other indexes. There were almost no aborted beats when VR was slowed to 150% SCL level (Fig. 3E). The composite hemodynamic responses in all 10 dogs during sinus rhythm, AF alone, and AF accompanied with different levels of VR slowing are summarized in Table 2. AF resulted in a fast VR with deterioration of all hemodynamic parameters compared with sinus rhythm (P ⬍ 0.001). Selective AVN-VS, by prolonging the R-R intervals, improved CO, SBP, DBP, LVSP, LVEDP, ⫾d(LVP)/dt, as well as SV and decreased the number of aborted beats (Table 2, compare the asterisk-labeled values with the values for AF). It should be noted that in contrast to the sinus rate, AF was associated with smaller LVSP than SBP values (63 ⫾ 12 vs. 77 ⫾ 17 mmHg). This apparent “discrepancy” resulted from the presence of large (43 ⫾ 9%) number of aborted beats, for which LVSP is always less than SBP. Because an aborted beat produced no AoF, its (apparent) SBP was defined as equal to the DBP of previous cycle. On the other hand, because the aortic valve did not open, it is obvious that the LVSP for the AJP-Heart Circ Physiol • VOL aborted beat was far below the DBP of the previous beat. Slowing of the VR by the VS reduced the number of aborted beats from 43% to 2% (Table 2) and resulted in a progressive reduction of the difference between the averaged LVSP and SBP. A more detailed analysis of the dependency of the hemodynamics on the VR is shown in Fig. 4, where the experimental data points are superimposed on curves obtained with a polynomial fit procedure for each of the measured parameters (see METHODS). The regression lines for CO, SBP, LVSP, ⫾d(LVP)/dt, and DBP had a discernible maximum. The maximum occurred between 83% and 140% of SCL but was most clear for CO with a maximum at 87% SCL (Fig. 4A). The remaining parameters exhibited rather monotonic relationships. Although VR slowing in the entire range from 75% to 150% SCL tended to improve each parameter, there was no statistically significant additional beneficial effect at R-R⬎100% SCL for SBP, LVSP, ⫾d(LVP)/dt, DBP, and LVEDP (Fig. 4, D–I, and Table 2). Statistically significant increase in this range was evident only for the SV and especially for the number of aborted beats [Fig. 4, B and C, and Table 2 (compare the †-labeled values in columns 4, 6, and 7 with the values in column 5)]. Importantly, despite the increased SV, Fig. 3. Representative hemodynamic traces during AF (A), AF with VR slowing to 75% (B), 100% (C), 125% (D), and 150% (E) SCL. Electrocardiogram (ECG), standard surface ECG lead II; RA, right atrial electrogram; RV, right ventricle electrogram; AoP, aortic blood pressure; LVP, left ventricular pressure; AoF, aortic blood flow. The asterisk marks the aborted beats (see RESULTS). The delivery of AVN-VS could be seen by the artifacts in surface ECG at the end of QRS (B–E). 282 • MARCH 2002 • www.ajpheart.org Downloaded from http://ajpheart.physiology.org/ by 10.220.32.246 on May 3, 2017 Fig. 2. R-R intervals during spontaneous sinus rhythm, atrial fibrillation (AF), and AF with different levels of ventricular rate (VR) slowing achieved by selective AVN-VS. Each episode contains 500 R-R intervals. Beats 1–500, 501–1,000, 1,001–1,500, 1,501–2,000, 2,001–2,500, and 2,501–3,000 were collected during sinus rhythm, AF, and AF with VR slowing to 75%, 100%, 125%, and 150% of the spontaneous sinus cycle length (SCL), respectively. The SCL was 463 ms. The corresponding average R-R intervals in each subsequent period were 270 ms (AF), 348 ms (achieved 75% SCL), 462 ms (achieved 99.9% SCL), 577 ms (achieved 124.6% SCL), and 691 ms (achieved 149.2% SCL), respectively. H1105 H1106 OPTIMAL VENTRICULAR RATE IN AF Table 2. Hemodynamic changes (compared with the spontaneous sinus rhythm) during AF alone, and during AF with 4 different levels of feedback-controlled VR slowing by selective AVN-VS in 10 dogs AF ⫹ AVN-VS Parameter Sinus Rhythm AF TL ⫽ 75% SCL TL ⫽ 100% SCL TL ⫽ 125% SCL TL ⫽ 150% SCL 1 2 3 4 5 6 7 SBP, mmHg DBP, mmHg LVSP, mmHg LVEDP, mmHg ⫹d(LVP)/dt, mmHg/s ⫺d(LVP)/dt, mmHg/s SV, ml/beat % Aborted beats CO, l/min 100 ⫾ 16* 77 ⫾ 14* 101 ⫾ 12* 5.4 ⫾ 2.8* 1,691 ⫾ 364* ⫺1,789 ⫾ 373* 22.8 ⫾ 8.0* 0* 2.7 ⫾ 0.7* 77 ⫾ 17 61 ⫾ 14 63 ⫾ 12 7.6 ⫾ 3.0 1,327 ⫾ 379 ⫺952 ⫾ 356 8.6 ⫾ 2.9 43 ⫾ 9 1.8 ⫾ 0.4 96 ⫾ 21* 77 ⫾ 19* 80 ⫾ 11* 6.0 ⫾ 2.4* 1,624 ⫾ 342* ⫺1,315 ⫾ 429* 14.3 ⫾ 5.6*† 33 ⫾ 10*† 2.2 ⫾ 0.7* 98 ⫾ 20* 74 ⫾ 17* 89 ⫾ 13*‡ 6.2 ⫾ 2.2 1,683 ⫾ 414* ⫺1,448 ⫾ 461*‡ 19.0 ⫾ 6.7*‡ 20 ⫾ 8*‡ 2.3 ⫾ 0.6*‡ 96 ⫾ 17* 69 ⫾ 14 91 ⫾ 10* 5.5 ⫾ 2.0* 1,635 ⫾ 340* ⫺1,435 ⫾ 310* 21.4 ⫾ 7.4†* 10 ⫾ 8†* 2.1 ⫾ 0.5†* 97 ⫾ 21* 66 ⫾ 17† 95 ⫾ 12* 5.7 ⫾ 2.2* 1,674 ⫾ 376* ⫺1,539 ⫾ 484 23.6 ⫾ 7.1*† 2 ⫾ 6*† 1.9 ⫾ 0.5† the slowing of the VR beyond 100% SCL resulted in a significant decline of the CO [Fig. 4A, and Table 2 (compare the †-labeled values in columns 6 and 7 with the values in column 5)]. This negative outcome was a combined result of the increased SV and the substantially decreased VR. Thus slowing of VR to 100% SCL increased most hemodynamic parameters and produced the best CO. However, these improvements were still insufficient to reach the values measured during the spontaneous sinus rhythm [(Table 2 (compare ‡-labeled values in column 5 with column 2)]. This, at least in part, may be because the slowing of the VR by AVN-VS did not eliminate the irregularity of the heart rhythm. In fact, the example illustrated in Fig. 2 demonstrates that in absolute terms there was larger beat-to-beat variability at the slower VR levels. The summarized data from all 10 dogs are shown in Fig. 5. The relative irregularity is represented by the normalized standard deviation of the 500 measured R-R intervals (as a percentage of SD/R-R, closed symbols). The CO, normalized versus the values at sinus rate, is also shown (as percent, open symbols). Compared with AF, it is clear that the CO increased from 66% in AF to 85% of the maximum when AVN-VS slowed the rate to 100% SCL. At that level of slowing, there was an SD/R-R ⫽ 25%. It is interesting to note that further slowing of the VR to 150% SCL actually reduced CO to 70% despite the fact that the variability declined to 17%. Although the present experimental protocol does not permit direct evaluation of the hemodynamic contributions of irregularity (see DISCUSSION), the data suggest a predominant role of the optimal slowing of the VR in maintaining high CO during AF. DISCUSSION Major findings. This study confirmed that selective AVN parasympathetic stimulation through the epicardial fat pad approach could be used to control (i.e., to AJP-Heart Circ Physiol • VOL slow down to a predetermined level) the average VR during AF. AF worsened all hemodynamic parameters compared with those observed during sinus rhythm. We identified a range of vagally induced VR slowing that was associated with improvement of hemodynamics. Although a substantial improvement (vs. AF) of all major hemodynamic parameters was observed at any level of AVN-VS, the best results were achieved when VR was maintained equal to, or slightly faster than, the spontaneous sinus rate. In particular, the CO, which is a product of the SV and VR, was suboptimal at 75% SCL due to insufficient SV, as well as at 125% and 150% SCL due to excessive slowing of VR. Therefore, we concluded that 100% SCL should be a preferable target during AF. Fast VR during AF and its effect on the hemodynamics. Multiple mechanisms are responsible for the detrimental hemodynamic consequences during AF. These include the increase in heart rate, the loss of atrial contribution to ventricular filling, the reduced interval for passive diastolic filling, and the irregular ventricular rhythm (3, 7, 9, 15, 24, 26, 33, 35, 40). Clinically, VR slowing is considered to be the first therapeutic step in a majority of patients with both acute and chronic AF (16). The importance of this parameter is further accentuated by the observation that in patients with successful conversion of AF, CO increase was observed only when the conversion was associated with slowing of the VR (1, 20, 32). The detrimental hemodynamic response during AF is a complex function of the rapid irregular sequence of shorter and longer R-R intervals. Mechanistically, this produces interaction of two phenomena. First, after a short R-R interval, the inotropic response is decreased due to incomplete mechanical restitution reflecting the dynamics of calcium release from the sarcoplasmic reticulum (46). In addition, according to the FrankStarling principle (4), the short R-R intervals are associated with reduced end-diastolic volume that fur- 282 • MARCH 2002 • www.ajpheart.org Downloaded from http://ajpheart.physiology.org/ by 10.220.32.246 on May 3, 2017 Values are means ⫾ SD. TL, target level of the average R-R interval as a percentage of the SCL. SBP and DBP: systolic and diastolic blood pressures determined from the aortic pressure (AoP) signal; LVSP and LVEDP, left ventricular systolic and left ventricular end-diastolic pressures determined from the LVP signal; ⫾d(LVP)/dt, ⫾ maximal rate of change of LV pressure; SV, stroke volume; CO, cardiac output. P ⬍ 0.05 was considered to be significant. * Statistically significant differences vs. AF; † statistically significant differences between the values at 75%, 125%, and 150% SCL vs. the values at 100% SCL; ‡ statistically significant differences between the values at 100% SCL vs. the values at sinus rhythm. OPTIMAL VENTRICULAR RATE IN AF H1107 ther attenuates the subsequent SV. Second, when a longer R-R interval occurs after a shorter one, the mechanical response after the former exhibits a relative potentiation (34, 46). The random occurrence of shorter and longer R-R intervals during AF results in a complex interaction between the above two phenomena. The overall decreased cardiac index (15) indicates that the persistent, adverse effects of shorter R-R intervals on ventricular performance dominate the hemodynamic outcome. Although the present study was not designed to directly evaluate the effects of irregularity, the data in Fig. 5 suggest that vagally induced slowing of the VR during AF was associated with an increased standard deviation of the R-R intervals. However, the present observations preclude conclusion about the separate AJP-Heart Circ Physiol • VOL Fig. 5. Normalized cardiac output in percent vs. the values at the spontaneous sinus rate (E) and heart rate variability represented as a normalized standard deviation of 500 measured R-R intervals (in %SD/R-R, F) during AF and 4 levels of VR slowing to 75%, 100%, 125%, and 150% of the SCL. AVN-VS increased the normalized CO from 66% during AF to 85% at slowing of 100% SCL, although this was accompanied by an increase in irregularity from 17% to 25% (P ⬍ 0.001 for both). See RESULTS for details. 282 • MARCH 2002 • www.ajpheart.org Downloaded from http://ajpheart.physiology.org/ by 10.220.32.246 on May 3, 2017 Fig. 4. Polynomial contrasts analysis of the measured hemodynamic parameters during AF and 4 levels of VR slowing expressed as %SCL. The VR corresponding to the AF is considered an additional level (the experimental value for it in 10 animals was 58 ⫾ 7% SCL). Solid circles represent mean experimental data (see Table 2). The lines represent the fit obtained with a combination of significant linear, quadratic, cubic, and quartic components. Optimal %SCL, where detected, is indicated with dashed lines. The optimum in the CO curve at 87% SCL (A). Excessive VR slowing (R-R ⬎100% SCL), despite the progressive increase in the SV (B) and the decline in the number of aborted beats (C), resulted in a decrease of the CO (A). See RESULTS for details. H1108 OPTIMAL VENTRICULAR RATE IN AF AJP-Heart Circ Physiol • VOL well as in dogs in vivo (44). The present study extended these findings and established that precise VR slowing can be achieved by feedback-controlled AVN-VS. The superiority of this approach is that it avoids the unwanted effects carried by drugs. Moreover, in our experiments, AVN-VS exerted its effects exclusively on the AVN. That is, when applied during sinus rhythm it did not change the sinus rate (44), and when applied during atrioventricular sequential pacing it did not alter any hemodynamic indexes (not shown in the reported data). Furthermore, AVN-VS allowed VR reduction to several predetermined levels to be achieved promptly during AF. Notably, by preserving the anterograde His-Purkinje conduction, we were able to evaluate the hemodynamic response in a model much closer to the real natural AF compared with models utilizing retrograde ventricular pacing (22). Implications of the reported findings and study limitations. The anatomic basis of the epicardial approach for selective parasympathetic stimulation has been well established in animal models (2, 6, 12, 13, 21, 28–30). Although only endocardial stimulation has been so far employed to produce dromotropic effects in humans (11, 17–19, 43), it is possible that the technique used in the present study could be applied in some patients, especially postoperative patients with AF. The optimal VR level observed in this study could be considered as a clinical target VR level during AF. However, due to the nature of animal studies, certain limitations should be considered. First, these are acute experiments. Whether the acutely determined rates would also apply to a chronic stage, and whether enhanced sympathetic tone during exercise would compete with the AVN-VS remain to be determined in chronic experiments. Second, whether the optimal rate derived from healthy animals can be recommended in a diseased state is still not clear. This is important because clinical AF is frequently accompanied by other heart diseases (i.e., coronary heart disease and heart failure). Third, the prolonged effects of AVN-VS during AF need further investigation in chronic experiments. Finally, the slowed ventricular rhythm observed in this study was irregular. It is interesting that the slight increase of variability with the slowing of the VR (Figs. 2 and 5) has also been documented in patients undergoing slow pathway AVN modification during AF (41). Specifically, the ablation prolonged the average R-R interval from 481 to 640 ms and increased the average SD/R-R from 16.8% to 20%. Although the mechanism of this phenomenon is not known, it might represent an inherent property of conduction of the depressed AVN during AF. A comparison of the hemodynamic outcome during controlled slowing of the anterograde irregular ventricular activation (as achieved in the present experiments) with complete AVN ablation and retrograde but regular pacing at several comparable slow rates has never been done and deserves a separate investigation. Such studies are needed to evaluate whether or not the price paid in AVN destruction and pacemaker dependency is worth a potential hemodynamic benefit. 282 • MARCH 2002 • www.ajpheart.org Downloaded from http://ajpheart.physiology.org/ by 10.220.32.246 on May 3, 2017 negative hemodynamic contribution of this factor. In particular, the best CO was achieved at 100% SCL (Table 2 and Fig. 5), despite the fact that at this level of AVN-VS the irregularity was higher than at lesser (75% SCL) or higher (150% SCL) degrees of VR slowing (Fig. 5). Optimal anterograde VR slowing during AF. Although the beneficial effect of the slower VR during AF is axiomatic, the relationship between the VR and the parameters of cardiac function in AF has not been clearly quantified. In this study, we were able to achieve different levels of VR slowing during AF by utilizing vagal modulation of AVN conductivity without directly affecting ventricular performance. Our results clearly showed that the detrimental, rapid, irregular ventricular response could be successfully attenuated by selective AVN-VS. Slowing the VR to the sinus rate or slightly faster (average R-R interval in the range of 75%-100% of the SCL) appeared to yield best overall results (Table 2). The polynomial fit indicated a presence of an optimum for the CO at 87% of the SCL (Fig. 4). Slowing to 125% and 150% of the SCL further improved some of the other measured parameters, although only for SV and the number of aborted beats the additional changes reached statistical significance (Table 2, Fig. 4). However, the overall hemodynamic effectiveness as reflected in the CO decreased due to excessive reduction of the number of cardiac contractions per unit time. This is in agreement with previous studies suggesting that lengthening of the diastolic interval beyond ⬃700 ms does not appreciably increase the LV end-diastolic volume, whereas shortening of the diastolic interval to ⬍500 ms impairs LV filling and SV (14). Interestingly, the empirically recommended target rate of 60–90 beats/min for humans during AF (45, 47) corresponds closely to the data obtained in this study (i.e., for an average sinus rate of 70 beats/min, or SCL ⫽ 857 ms, the recommended target of 75–100% SCL during AF would be 70–93 beats/min). Selective AVN-VS as a useful novel method for slowing VR during AF. As previously indicated, the use of drugs to slow the VR during AF has limitations because VR changes are unpredictable in response to treatment (22), and agents such as -blockers, verapamil, diltiazem, and digitalis, have a direct effect on cardiac function (31). Moreover, drugs lack efficacy in some subjects and are intolerable by others (8). The approach used in the present study was based on the well-established dromotropic effects of the vagus on AVN. The cell bodies of the postganglionic parasympathetic nerve fibers, which selectively innervate limited regions of the heart, have been identified for both animals and humans (2, 5, 6, 12, 13, 21, 28–30). For example, the fibers that innervate the AVN course through “. . .a smaller fat pad overlying epicardium at the junction of inferior vena cava-inferior left atrium” (29). It is known that stimulation of these parasympathetic projections can slow AV conduction without affecting other cardiac functions (2, 6, 12, 13, 21, 28–30). Using this method, we have previously demonstrated the possibility to slow VR during AF in vitro (23) as OPTIMAL VENTRICULAR RATE IN AF The authors thank Donald G. Hills and William J. Kowalewski for expert help during the surgical preparation and assistance in the experiments. This study was supported in part by National Heart, Lung, and Blood Institute Grant RO1-HL-60833-01A1. REFERENCES AJP-Heart Circ Physiol • VOL 19. Keim S, Mehdirad AA, Rist K, Mazgalev T, and Tchou P. Subthreshold burst stimulation of the AV node unmasks latent preexcitation in a concealed ectopic nodoventricular accessory pathway (Abstract). Pacing Clin Electrophysiol 17: 336, 1994. 20. Killip T and Baer RA. Hemodynamic effects after reversion from atrial fibrillation to sinus rhythm by precordial shock. J Clin Invest 45: 658–671, 1966. 21. Lazzara R, Scherlag BJ, Robinson MJ, and Samet P. Selective in situ parasympathetic control of the canine sinoatrial and atrioventricular nodes. Circ Res 32: 393–401, 1973. 22. Liau CS, Chen MF, Lin FY, Tsai CH, and Lee YT. Relationship between ventricular rate and cardiac output in mimic experimental atrial fibrillation. J Electrocardiol 27: 163–168, 1994. 23. Mazgalev TN, Garrigue S, Mowrey KA, Yamanouchi Y, and Tchou PJ. Autonomic modification of the atrioventricular node during atrial fibrillation: role in the slowing of ventricular rate. Circulation 99: 2806–2814, 1999. 24. Mitchell JH and Shapiro W. Atrial function and the hemodynamic consequences of atrial fibrillation in man. Am J Cardiol 23: 556–567, 1969. 25. Morady F, Hasse C, Strickberger SA, Man KC, Daoud E, Bogun F, Goyal R, Harvey M, Knight BP, Weiss R, and Bahu M. Long-term follow-up after radiofrequency modification of the atrioventricular node in patients with atrial fibrillation. J Am Coll Cardiol 29: 113–121, 1997. 26. Naito M, David D, Michelson EL, Schaffenburg M, and Dreifus LS. The hemodynamic consequences of cardiac arrhythmias: evaluation of the relative roles of abnormal atrioventricular sequencing, irregularity of ventricular rhythm and atrial fibrillation in a canine model. Am Heart J 106: 284–291, 1983. 27. Prystowsky EN, Benson DW Jr, Fuster V, Hart RG, Kay GN, Myerburg RJ, Naccarelli GV, and Wyse DG. Management of patients with atrial fibrillation. A statement for healthcare professionals. The Subcommittee on Electrocardiography and Electrophysiology, American Heart Association. Circulation 93: 1262–1277, 1996. 28. Randall WC, Ardell JL, Calderwood D, Miloslavljevic M, and Goyal SC. Parasympathetic ganglia innervating the canine atrioventricular nodal region. J Auton Nerv Syst 16: 311–323, 1986. 29. Randall WC, Ardell JL, O’Toole MF, and Wurster RD. Differential autonomic control of SAN and AVN regions of the canine heart: structure and function. Prog Clin Biol Res 275: 15–31, 1988. 30. Randall WC, Miloslavljevic M, Wurster RD, Geis GS, and Ardell JL. Selective vagal innervation of the heart. Ann Clin Lab Sci 16: 198–208, 1986. 31. Reiffel JA. Drug choices in the treatment of atrial fibrillation. Am J Cardiol 85: 12–19, 2000. 32. Resnekov L. Haemodynamic studies before and after electrical conversion of atrial fibrillation and flutter to sinus rhythm. Br Heart J 29: 700–708, 1967. 33. Rodman T, Pastor BH, and Figueroa W. Effect on cardiac output of conversion from atrial fibrillation to normal sinus mechanism. Am J Med 41: 249–258, 1966. 34. Samet P. Hemodynamic sequelae of cardiac arrhythmias. Circulation 47: 399–407, 1973. 35. Samet P, Bharati S, and Levy D. Significance of the atrial contribution to ventricular filling. Am J Cardiol 15: 195–202, 1965. 36. Schauerte P, Scherlag BJ, Scherlag MA, Goli S, Jackman WM, and Lazzara R. Ventricular rate control during atrial fibrillation by cardiac parasympathetic nerve stimulation: a transvenous approach. J Am Coll Cardiol 34: 2043–2050, 1999. 37. Schauerte P, Scherlag BJ, Scherlag MA, Jackman WM, and Lazzara R. Transvenous parasympathetic nerve stimulation in the inferior vena cava and atrioventricular conduction. J Cardiovasc Electrophysiol 11: 64–69, 2000. 38. Schauerte PN, Scherlag BJ, Scherlag MA, Goli S, Jackman W, and Lazzara R. Transvenous parasympathetic cardiac 282 • MARCH 2002 • www.ajpheart.org Downloaded from http://ajpheart.physiology.org/ by 10.220.32.246 on May 3, 2017 1. Abildskov JA, Millar K, and Burgess MJ. Atrial fibrillation. Am J Cardiol 28: 263–267, 1971. 2. Ardell JL and Randall WC. Selective vagal innervation of sinoatrial and atrioventricular nodes in canine heart. Am J Physiol Heart Circ Physiol 251: H764–H773, 1986. 3. Benchimol A. Significance of the contribution of atrial systole to cardiac function in man. Am J Cardiol 23: 568–571, 1969. 4. Braunwald E, Frye RL, Atgen AA, and Gilbert JW. Studies on Starling’s law on the heart. III. Observations in patients with mitral stenosis and atrial fibrillation on the relationships between left ventricular end-diastolic segment length, filling pressure, and the characteristics of ventricular contraction. J Clin Invest 39: 1874–1884, 1960. 5. Carlson MD, Geha AS, Hsu J, Martin PJ, Levy MN, Jacobs G, and Waldo AL. Selective stimulation of parasympathetic nerve fibers to the human sinoatrial node. Circulation 85: 1311– 1317, 1992. 6. Chiou CW, Eble JN, and Zipes DP. Efferent vagal innervation of the canine atria and sinus and atrioventricular nodes. The third fat pad. Circulation 95: 2573–2584, 1997. 7. Clark DM, Plumb VJ, Epstein AE, and Kay GN. Hemodynamic effects of an irregular sequence of ventricular cycle lengths during atrial fibrillation. J Am Coll Cardiol 30: 1039– 1045, 1997. 8. Crijns HJ, Van Gelder IC, and Lie KI. Benefits and risks of antiarrhythmic drug therapy after DC electrical cardioversion of atrial fibrillation or flutter. Eur Heart J 15, Suppl: 17–21, 1994. 9. Daoud EG, Weiss R, Bahu M, Knight BP, Bogun F, Goyal R, Harvey M, Strickberger SA, Man KC, and Morady F. Effect of an irregular ventricular rhythm on cardiac output. Am J Cardiol 78: 1433–1436, 1996. 10. Feld GK. Radiofrequency catheter ablation versus modification of the AV node for control of rapid ventricular response in atrial fibrillation. J Cardiovasc Electrophysiol 6: 217–228, 1995. 11. Fromer M and Shenasa M. Ultrarapid subthreshold stimulation for termination of atrioventricular node reentrant tachycardia. J Am Coll Cardiol 20: 879–883, 1992. 12. Furukawa Y, Wallick DW, Carlson MD, and Martin PJ. Cardiac electrical responses to vagal stimulation of fibers to discrete cardiac regions. Am J Physiol Heart Circ Physiol 258: H1112–H1118, 1990. 13. Gatti PJ, Johnson TA, Phan P, Jordan IK, Coleman W, and Massari VJ. The physiological and anatomical demonstration of functionally selective parasympathetic ganglia located in discrete fat pads on the feline myocardium. J Auton Nerv Syst 51: 255–259, 1955. 14. Gosselink AT, Blanksma PK, Crijns HJ, Van Gelder IC, De Kam PJ, Hillege HL, Niemeijer MG, Lie KI, and Meijler FL. Left ventricular beat-to-beat performance in atrial fibrillation: contribution of Frank-Starling mechanism after short rather than long RR intervals. J Am Coll Cardiol 26: 1516–1521, 1995. 15. Herbert WH. Cardiac output and the varying R-R interval of atrial fibrillation. J Electrocardiol 6: 131–135, 1973. 16. Jung F and DiMarco JP. Treatment strategies for atrial fibrillation. Am J Med 104: 272–286, 1998. 17. Keim S, Mazgalev T, Kinn R, and Tchou P. Non captured burst stimulation near the AV node: evidence for vagal stimulation as the mechanism for modulation of AV conduction in humans (Abstract). Pacing Clin Electrophysiol 15: 519, 1992. 18. Keim S, Mazgalev T, and Tchou P. Physiologic effects of subthreshold burst stimulation on the human AV node (Abstract). Pacing Clin Electrophysiol 16: 158, 1993. H1109 H1110 39. 40. 41. 42. 43. OPTIMAL VENTRICULAR RATE IN AF nerve stimulation: an approach for stable sinus rate control. J Cardiovasc Electrophysiol 10: 1517–1524, 1999. Scherlag MA, Scherlag BJ, Yamanashi W, Schauerte P, Goli S, Jackman WM, Reynolds D, and Lazzara R. Endovascular neural stimulation via a novel basket electrode catheter: comparison of electrode configurations. J Interv Card Electrophysiol 4: 219–224, 2000. Shapiro W and Klein G. Alterations in cardiac function immediately following electrical conversion of atrial fibrillation to normal sinus rhythm. Circulation 38: 1074–1084, 1968. Simpson C, Yee R, Lee JK, Murgatroyd FD, Basta M, and Krahn AD. The effect of catheter AV node modification on heart rate irregularity in atrial fibrillation (Abstract). Circulation 98: I-181, 1998. Snedecor GW and Cochran WG. Statistical Methods. Ames, IA: Iowa State University Press, 1989, p. 409. Tchou P, Keim S, Kinn R, and Mazgalev T. Non captured burst stimulation near the AV node: selective influence on slow 44. 45. 46. 47. pathway conduction in AV nodal reentrant tachycardia in humans (Abstract). J Am Coll Cardiol 19: 144A, 1992. Wallick DW, Zhang Y, Tabata T, Zhuang S, Mowrey KA, Watanabe J, Greenberg NL, Grimm RA, and Mazgalev TN. Selective AV nodal vagal stimulation improves hemodynamics during acute atrial fibrillation in dogs. Am J Physiol Heart Circ Physiol 281: H1490–H1497, 2001. Wood DL, Hammill SC, and Kopecky SL. Supraventricular arrhythmias. In: Mayo Clinic Practice of Cardiology, edited by Giuliani ER, Gersh BJ, McGoon MD, Hayes DL, and Schaff HV. St. Louis, MO: Mosby, 1996, p. 748–779. Yue DT, Burkhoff D, Franz MR, Hunter WC, and Sagawa K. Postextrasystolic potentiation of the isolated canine left ventricle. Relationship to mechanical restitution. Circ Res 56: 340– 350, 1985. Zipes DP. Specific arrhythmias: diagnosis and treatment. In: Heart Disease–A Textbook of Cardiovascular Medicine, edited by Braunwald E. Philadelphia, PA: Saunders, 1992, p. 667–715. Downloaded from http://ajpheart.physiology.org/ by 10.220.32.246 on May 3, 2017 AJP-Heart Circ Physiol • VOL 282 • MARCH 2002 • www.ajpheart.org