* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Occurrence and Fruit and Seed Biology of
Survey
Document related concepts
Transcript
Aust. J. Mar. Freshwater Res., 1993, 44, 43-57
Occurrence and Fruit and Seed Biology of
Halophila tricostata Greenway (Hydrocharitaceae)
J. K U O ~W.
, Lee ~ o n and
g ~R. G. colesB
Centre for Microscopy and Microanalysis, The University of Western Australia,
Nedlands, WA 6009, Australia.
Northern Fisheries Centre, Queensland Department of Primary Industries,
Box 5396, Cairns, Qld 4871, Australia.
A
Abstract
Halophila tricostata Greenway appears to be endemic to eastern Queensland, Australia, and occurs
between 14O11'S and 23'45's. It was found at depths from 1 . 4 to 30 m in well sheltered habitats,
including in shallow coastal sites near mangrove-lined estuaries, on the lee side of continental and coralreef islands, and on some commercial prawn-trawling grounds within the Great Barrier Reef lagoon.
It grows on predominantly fine mud substratum in small monospecific meadows or mixed with other
tropical seagrasses, mostly other Halophila species. Field observations indicate that H. tricostata is an
annual angiosperm and produces an estimated 70 000 seeds m-2 year-'.
Halophila tricostata is dioecious. The plant has a horizontal rhizome bearing an erect shoot with
eight to twelve nodes and a root at each rhizome node. Except at the first two or three nodes, the
mature plants produce a reproductive organ at each node of their rarely branched erect shoot. The
reproductive organs and fruits develop and mature acropetally along the erect shoot. There are
24-60 seeds, with a mean of 41 seeds, per fruit. The seed has a coiled embryo protected by a cotyledon,
and an enlarged hypocotyl. The hypocotyl acts as a nutrient store and contains starch, protein and
lipid. The seed covering consists of pericarp remains and two thin cuticular layers of seed coat.
The surface of the seed covering has numerous fine protrusions. The seed covering becomes loose
and is discarded during germination, exposing the hypocotyl. The surface of the hypocotyl develops
hair-like unicellular structures during seedling development. The majority of the seeds begin to
germinate at 26-2g0C after two weeks of culturing, but germination is not synchronized. The culturing
of H. tricostata seedlings beyond the three-leaf stage was not successful.
Introduction
The distribution and reproductive biology of Halophila tricostata Greenway are among
the least known for the Halophila species. Since the first description of H. tricostata from
sledge samples near Lizard Island, north-eastern Queensland (Greenway 1979), its presence
has been recorded over a much wider distributional range (Coles et al. 1987a, 1987b, 1992;
Lee Long et al. 1989). Observations during these surveys indicated that the plant may be
much more ephemeral than most other seagrasses, and a monitoring study was implemented
to examine this aspect of its biology.
Morphological descriptions of fruits and seeds in Halophila are few (Balfour 1878; den
Hartog 1970). This has been attributed to the inconspicuous fruits and seeds, which have
probably been overlooked (den Hartog 1970). However, recent studies of fruit and seed
morphology have been carried out on H. engelmannii Aschers. (McMillan 1986, 1987a,
1988a), H. decipiens Ostenfeld (McMillan 1986~;Parthasarathy et al. 1988b; McMillan
and Soong 1989), H. beccarii Aschers. (Parthasarathy et al. 1988a) and H. ovalis (R. Br.)
Hook. f. (Kuo and Kirkman 1992). The mature fruits and seeds of H. tricostata have
J. Kuo et al.
not been previously described (Greenway 1979). The anatomy of fruits and seeds of the
Halophila species, in particular the embryo and seed reserves, is not well understood and
has been investigated only in H. ovalis (Kuo and Kirkman 1992).
The morphology of germination and the establishment of seedlings in Halophila have
been described for H. spinulosa (R. Br.) Aschers. (Birch 1981), H. engelmannii (McMillan
1987b; McMillan and Jewett-Smith 1988; Jewett-Smith and McMillan 1990), H. decipiens
(McMillan 1988b) and H. ovalis (Kuo and Kirkman 1992).
This paper reports on the distribution and habitats of Halophila tricostata and describes
the morphology and anatomy of the mature fruits, seeds and seedlings of this species.
Materials and Methods
Information on the occurrence and distribution of H. tricostata was based mainly on Queensland
Department of Primary Industries surveys of Queensland coastal and island areas conducted from 1984
to 1989 (see Coles et al. 1987a, 1987b, 1992; Lee Long et al. 1989, 1992). A further monthly or
bimonthly survey was conducted at a permanent site, 16 m below mean sea level on the north-western
lee side of Fitzroy Island (a small continental island in the Great Barrier Reef lagoon, 6 km from the
mainland, 16"56'S), from October 1987 to February 1990.
Flowering and fruiting plants of H. tricostata were collected from Fitzroy Island on 23 November
1990. Mature fruits and seeds were separated and stored in the dark in aerated recirculating sea water
(salinity 35) at 2 8 T for 35 days. Seed counts were carried out for 30 fruits, and seed diameter was
measured on 70 seeds.
For the germination trials, seeds were sterilized in 90% ethanol for 20 min, then rinsed twice in
autoclaved sea water. Seeds were transferred to axenic conditions in 200-mL flasks containing 150 mL
of sea water and kept at 26 to 28OC on a 12-h light/dark cycle. Ambient illumination on the experimental jars was 100-125 pE m-2 s-' (1 pE= 1 pmol), monitored with a Li-Cor Model LI-188B
quantum radiometer-photometer. Seed germination and seedling development were recorded and
photographed periodically and were also examined by scanning electron microscopy (SEM).
For SEM, seeds and seedlings were fixed in 2.5% glutaraldehyde in sea water and dehydrated with
graded acetone, then critical-point-dried with C02. Specimens were examined with a Philips Model 505
scanning electron microscope.
For anatomical and histochmeical studies, mature fruits with maturing seeds were fixed in 2.5%
glutaraldehyde in sea water and embedded in glycol and methacrylate (O'Brien and McCully 1981).
Serial sections (2.5 pm thick) were stained with saturated Sudan black B in 70% ethanol for lipids and
cuticle, 1% amido black 10B in 7% acetic acid for protein, periodic acid-Schiff's reaction (PAS) for
starch and other polysaccharides, and 0.05% toluidine blue (pH 4.4) for general cell organization and
phenols (see Kuo et al. 1990, 1991; Kuo and Kirkman 1992).
Results
Habitats and Distribution of H. tricostata
Field observations of Halophila tricostata appear as occasional records in a series of
reports on the distribution of seagrasses on the eastern Queensland coast and are compiled
here (Table 1). The known latitudinal distribution of H. tricostata is from 14'11'S to 23'45's
(Fig. 1). The species was found during surveys at depths between 1.4 and 30 m, always in
sheltered waters. Sites included shallow-water ( < 5 m) coastal habitats near mangrove
wetlands and estuaries, and deep waters of the Great Barrier Reef lagoon. Additional records
have come from benthic otter-trawls engaged in commercial prawn-fishing operations at
20 m depth 6 nautical miles east of Cape Flattery (15's) in November and December 1989
and 1990 and south-east of Fitzroy Island (about 17"s) in December 1986.
Habitats where H. tricostata were observed and collected all include a substratum of
predominantly fine mud, with some carbonate sand and shell grit. Halophila tricostata
occurred in small patches of monospecific meadows but was more commonly found mixed
with H. decipiens and sometimes with H. ovata Gaud. and H. spinulosa of the Hydrocharitaceae, Halodule uninveris (Forsk.) Aschers., Syringodium isoetifolium (Aschers.)
Hinders Islands Group*
Howick Islands Group*
Low Islets#
Fitzroy Island*
Hutchison Island*
Dunk Island*
Hinchinbrook Island Channelt
Magnetic Island*
Upstart Bay+
Hook Island*
Gladstone Harbour+
~ocality~
("3
Latitude
Depth
(m)
Vegetation cover
(all species) (96)
H. tricostata
(shoots m-')
HD, HO, HS, HT
CS, HD, HO, HS
HD
HD
HD, HT
HD, HS, HU, SI
HD, HO, HU
HD, HO, HU
HO, HS, HU
None
HD, HO, HU, ZC
Other speciesC
A
Based on Queensland Department of Primary Industries surveys (Coles et al. 1987u, 1987b, 1992; Lee Long et al. 1989, 1992).
Locality types: *, continental island; #, coral island; +,mangrove-lined estuary and bay.
Other seagrasses occurring with Halophila tricostata: CS, Cymodocea serrulata; HD, Halophila decipiens; HO, Halophila ovalis; HS, Halophila
spinulosa; HT, Halophila ovata; HU, Halodule uninervk (wide-leafed); SI, Syringodium isoetifolium; ZC, Zostera cupricorni.
Date
Table 1. Records of occurrence of Halophila tricostata and associated seagrass species in surveys of Queensland coastal and island areasA
J. Kuo et al.
Dandy and Cymodocea serrulata (R. Br.) Aschers. of the Cymodoceaceae, and Zostera
capricorni Aschers. of the Zosteraceae (Table 1).
Halophila tricostata abundance at the Fitzoy Island site was greatest in October and
November, and plants were almost absent from the site between February and September
(autumn and winter) each year (Fig. 2). However, the abundance of H. tricostata at the site
also varied between years, and the bottom vegetation cover was sometimes less than that
at adjacent sites (Fig. 2; Table 1). A density of about 284 erect shoots m-2 was counted
at Fitzroy Island in December 1989, of which only about 5-10% were male shoots. From
the seed production rate (see below) this equates to an estimated supply of about 70000
seeds m-2.
Plant Morphology in H. tricostata
Halophila tricostata is dioecious, although the morphology and appearance of the mature
plant are similar in both male and female plants. Mature plants have a horizontal rhizome
that produces an unbranched root and an erect shoot at each rhizome node (Fig. 3).
Each erect shoot has eight to twelve unbranched or rarely branched nodes. Each of the first
AUSTRALIA
'6
-LOW ISLglS
-PITZROY ISLAND
-HINCHINBROOK ISLAND
UPSTART BAY
QUEENSLAND
Fig. 1. Area map, showing major localities for collections of Halophila tricostata during
seagrass surveys and monitoring (1984-89).
Occurrence and Fruits and Seeds of H. tricostata
I
I
1
I
I
I
I
1
,
I
I
I
I
I
l
I
I
I
,
I
/
,
I
I
1
1
1
1
1
,
Oct Dec Feb Apr Jun Aug Oct Dec Feb Apr Jun Aug Oct Dec
1987
1
1988
I
1989
11990
Changes in bottom vegetation cover of Halophila tricostata at Fitzroy Island
from October 1987 to January 1990.
Fig. 2.
two to three basal nodes normally bear two leaf blades representing 'junior leaves', but these
leaves, particularly those from the first node, are usually abscissed in mature reproductive
plants (Fig. 3). From the third or fourth node upwards, there is both a rosette of three
leaf blades representing 'adult leaves' and a reproductive organ at each node (Fig. 3).
The vegetative and reproductive organs develop and mature acropetally along the erect
shoot (Figs 4-6). Each male or female flower is covered by two bracts and supported by
the rosetted leaves (Figs 4 and 6). The male flower has a short pedicel, which becomes
elongated during pollination, and three pale-yellow fused anthers (Fig. 4). The female flower
has an ovoid ovary with a hypanthium bearing four to six styles up to 1 cm long (Fig. 5).
After fertilization, these styles are detached, leaving the hypanthium on the apical end of
the developing fruit (Fig. 6).
Fruit and Seed Morphology in H. tricostata
Five to eight (average 6.8) fruits are produced per erect shoot of H. tricostata at the
site near Fitzroy Island. The maturing fruit has a globose to subglobose shape and is
protected by a pair of bracts. A short (- 3 mm) stylar beak or hypanthium drops off after
the fruit is detached from the parental erect shoot. The surface of the fruit is smooth and
yellowish to brown in colour.
From 30 fruits, seed counts vary from 24 to 60 (mean 41) mature seeds per fruit.
There are also, on average, five immature or undeveloped seeds per fruit. The seed of
H. tricostata is spherical, usually bears short fruit-stalk remains at the apical end, and is
slightly apiculated at the basal end (Figs 7, 8 and 21). Seeds average 0.38k0.003 mm
(range 0.33-0.45 mm) in diameter. The surface of the seed covering in H. tricostata has
numerous fine protrusions, each protrusion being about 2-3 pm in height and some of them
being joined to each other (Figs 9 and 10).
Fruit and Seed Anatomy in H. tricostata
The fruit wall of H. tricostata consists of enlarged epidermal cells and three to four
layers of parenchymatous cells (Figs 11 and 12). The outer wall of each epidermal cell is
J. Kuo et al.
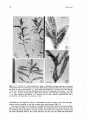
Figs 3-6. 3: Portion of a female reproductive plant of Halophila tricostata, showing a horizontal
rhizome (Rh) with a root (R) and an erect shoot (ES) bearing a developing fruit (F) at each node from
the third or fourth node upwards. x 1 . Scale marked in millimetres. 4: Erect shoot with portions
of the bracts removed to show the developing male flowers (A) containing fused anthers. x 8. Scale
bar, 1 mm. 5: Erect shoot bearing a female flower with four styles (St) and a developing fruit (F).
x 1.4. Scale marked in millimetres. 6: A maturing fruit (F) has a distinct hypanthium (H) and is
protected by two bracts (B). x6. Scale bar, 2 mm.
thickened but not lignified, without a detectable cuticular covering. The inner parenchymatous cells are smaller in size but contain more starch grains (Fig. 12).
The seed covering of H. tricostata is a complex structure and consists of pericarp remains
and the seed coat (Figs 13-16). Pericarp remains are represented by several cell layers that
are flattened and lie parallel to the seed surface. The walls of the outermost cell layer and
the outer tangential walls of the second cell layer are thin, but the radial and particularly
Occurrence and Fruits and Seeds of H. tricostata
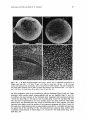
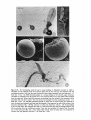
Figs 7-10. 7, 8: SEM photomicrographs show that a mature seed of Halophila tricostata has a
distinct seed stalk (Sk). x 135 (Fig. 7) and x 117 (Fig. 8). Scale bars, 100 pm. 9, 10: The seed
surface has numerous fine protrusions, some of which may be fused together. The seed covering also
has some debris (asterisk), and it splits to expose the surface of the hypocotyl (Hc). ~ 4 7 7(Fig. 9)
and x 990 (Fig. 10). Scale bars, 20 am (Fig. 9) and 10 pm (Fig. 10).
the inner tangential walls of the second-layer cells are thickened (Figs 13 and 14). These
thickened walls contain mainly polysaccharides and are not lignified (Figs 13 and 14).
In addition, the basal portion of the thickened inner tangential walls possesses numerous
vertical fine lines (Figs 13-16) that are rich in protein (Figs 14 and 15) and lipid (Fig. 16).
The seed coat is represented by two thin cuticular layers that stain positively with Sudan
black B (Fig. 16), indicating that they consist of cuticular and/or fatty material. The inner
cuticular layer tightly covers the surface of the hypocotyl epidermal cells (Figs 15 and 16).
The seed has a large hypocotyl and a coiled embryo protected by a cotyledon situated near
the apical end of the seed (Figs 17-20). The hypocotyl has numerous large uniform cells
containing a large amount of starch (Figs 9, 10, 13 and 14), with little protein (Fig. 15) or
lipid (Fig. 16).
J. Kuo et al.
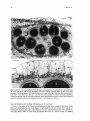
Figs 11 and 12. 11: Longitudinal section through a maturing fruit of Halophila tricostata, showing
the fruit wall (FW) and several developing starch-rich (darkly stained) seeds (S) that have been
sectioned in different planes. Each seed is attached to a seed stalk (Sk). Toluidine blue counterstained
with PAS. x50. Scale bar, 250 gm. 12: The fruit wall consists of several cell layers containing
enlarged epidermal cells (E) and starch (Sh)-rich inner parenchymatous cells covering the developing
seeds (S). Toluidine blue counterstained with PAS. x 126. Scale bar, 100 pm.
Seed Germination and Seedling Morphology in H. tricostata
None of the seeds of H. tricostata germinated during dark storage in the seven weeks
after seed collection, but the seeds began to germinate one to two weeks after transfer to
illuminated experimental jars. Germination was not synchronized; after two months of
culturing, about 30% of seeds had germinated and were at various stages of seedling
Occurrence and Fruits and Seeds of H. tricostata
51
Figs 13-20. 13-16. The seed covering in Halophila tricostata consists of pericarp remains and the two
layers of the seed coat (arrows and arrowheads). The pericarp remains have epidermal cells (E), and
the third parenchyma cell layer has thickened inner tangential walls (asterisks) with numerous fine
vertical lines. Sh, starch-rich cells. Toluidine blue counterstained with PAS (Fig. 13), amido black 10B
counterstained with PAS (Fig. 14), amido black 10B (Fig. 15), and Sudan black B (Fig. 16). All x 435.
Scale bars, all 15 pm. 17-20: Serial sections of a mature seed, showing the relationship between
the hypocotyl (Hc), the embryo (Eb) and a cotyledon (C). All toluidine blue. All x 110. Scale bars,
all 100 pm.
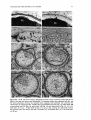
Figs 21-25. 21: Germinating seeds (S) and a young seedling of Halophila tricostata in which a
cotyledon (C) has emerged from the hypocotyl (Hc) and the first leaf (L) has emerged from the
cotyledonary pocket. Note that the seed covering (Sc) has already separated from the hypocotyl. x 2.
Scale bar, 5 mm. 22: SEM photomicrograph of a young seedling at the one-leaf stage, showing a
cotyledon (C) emerging from the apical end of the hypocotyl (Hc) and a long radicle (Ra) emerging
from the basal end. Several hair-like structures are growing from the surface of the hypocotyl, and a
discarded seed coat (Sc) is nearby. The first leaf (L) has emerged from the cotyledonary pocket. x 16.
Scale bar, 5 mm. 23, 24: SEM photomicrographs of seeds with the seed covering (Sc) removed to
show the apical end (asterisk) and basal end (arrowhead) of the hypocotyl as well as the surface of the
hypocotyl epidermal cells. x 88 (Fig. 23) and x 112 (Fig. 24). Scale bars, 100 pm. 25: Young seedling
at the two-leaf stage. The elongated first leaf (Ll) and second leaf (L2) and the emerging lateral root
(R)are growing from the cotyledonary pocket. Note that the cotyledon (C) extends from the apical
end of the hypocotyl (Hc) and a long radicle (Ra) extends from the basal end. The hypocotyl is
covered with long hair-like structures. x 14. Scale bar, 1 mm.
Occurrence and Fruits and Seeds of H. tricostata
development. Unfortunately, none of the seedlings developed beyond the three-leaf stage to
produce a horizontal rhizome for further development.
The initial sign of germination in H. tricostata was a swelling of the seeds, followed by
splitting and finally discarding of the outer seed covering (Figs 21 and 22) to expose the
rather smooth surface of the hypocotyl (Fig. 23). After the protrusion of the chalaza1 end
of the seed, the cotyledon and then the coiled embryo emerged through the split seed coat
(Fig. 24). The radicle then developed from the base of the hypocotyl, and there was
enlargement of the cotyledonary pocket (Figs 21 and 22). The first true leaf, with only a
single midrib, emerged from the cotyledonary pocket (Figs 21 and 22). By this stage, long
unicellular hairs were usually growing from the surface of the hypocotyl (Fig. 22). The
second and third leaves emerged successively from the sheath of the preceding leaf, and a
lateral root developed at the base of the first leaf on the enlarged cotyledonary pocket
(Fig. 25).
Discussion
Greenway (1979) did not find mature fruits of Halophila tricostata, but described the
species as having numerous ovules with reticulate testa. The present study found the surface
of the seed covering in H. tricostata to have numerous fine protrusions up to 2-3 pm in
height. These protrusions appear to resemble what has been described as peg-like projections
(about 10-15 pm in height) in H. spinulosa (Birch 1981) but are different from the reticulate
surface of H. engelmannii (McMillan 1987b) and H. ovalis (Birch 1981; Kuo and Kirkman
1992). Whether the appearance of the seed surface can be used for taxonomic and phylogenetic purposes in the genus Halophila remains to be determined. It is still not certain
whether the surface sculpturing of the seed covering in Halophila species has any functional
significance. The peg-like projections may provide surface friction against the substratum
to loosen the seed covering (Birch 1981). On the other hand, the reticulate surface may
facilitate positive buoyancy, by the entrapment of air bubbles, for further dispersal
(McMillan 1987b).
Despite the morphology and appearance of the mature plants and the differing
appearance of the seed surface among the Halophila species, the seed anatomy and the
morphology and initial development of seedlings are similar in the species that have been
studied, including H. spinulosa (Birch 1981), H. engelmannii (McMillan 1987b; McMillan
and Jewett-Smith 1988; Jewett-Smith and McMillan 1990), H. decipiens (McMillan 1988b)
and H. ovalis (Kuo and Kirkman 1992). Seeds of H. tricostata have a complex seed covering
consisting of pericarp remains and a two-layered seed coat, and each seed consists of
both a coiled embryo protected by a cotyledon and a large hypocotyl. This type of seed
organization appears to be typical of the genus Halophila (Lakshmanan 1963; den Hartog
1970; McMillan 19876; Kuo and Kirkman 1992), although Birch (1981) claimed that only
a single integument is present in H. spinulosa. Both layers of the seed coat derive from
inner and outer integuments during seed development in H. ovata (Lakshmanan 1963).
Probably the most interesting feature during seedling development in the Halophila species
is that the discarding of the seed covering ensures the early development of unicellular
hair-like structures from the hypocotyl epidermal cells. Similar hairs, known as 'hypocotylar
hairs' or the ring of 'anchoring hairs' on the 'hypocotylar collar', are apparently typical
of freshwater monocotyledonous seedlings and also occur in some aquatic dicotyledonous
seedlings and in a few terrestrial plants (see Arber 1925; Kaul 1978). Seeds of the tropical
seagrass Thalassia species have an enlarged pyriform hypocotyl with a flattened base
(Maiden and Betche 1909; Orpurt and Boral 1964; Kuo et al. 1991), and apparently only
from this flattened portion of the hypocotyl do the anchoring hairs develop (Orpurt and
Boral 1964). These anchoring hairs have been shown to be efficient anchors of seeds before
the radicle emerges (Birch 1981). The ontogeny and the development of the seed covering
J. Kuo et al.
in Halophila, and that o f the hair-like structure on the hypocotyl during seedling development, deserve further microscopical investigation.
A study on nutrient utilization in seed reserves during seedling development in H. ovalis
indicates that starch and protein in the hypocotyl are used for the germination and initial
development o f the seedling until it reaches about the three-leaf stage (Kuo and Kirkman
1992). The nutrients required for further development and growth o f the Halophila seedling
appear to be obtained from the substratum and the water surrounding the seedling. This
phenomenon could explain why, under normal laboratory conditions, successful germination
and early development o f H. tricostata seedlings are rather easily achieved but are rarely
followed by continued growth, as is also the case for H. spinulosa (Birch 1981) and
H. ovalis (Kuo and Kirkman 1992). Successful culturing o f young seedlings has led to
rosettes o f six leaves and beyond in H. decipiens (McMillan 1988b) and H. engelmannii
(McMillan 1987b; McMillan and Jewett-Smith 1988; Jewett-Smith and McMillan 1990).
Field observations at Fitzroy Island indicated that seeds o f H. tricostata have a winter
dormancy requirement and that the timing o f germination is controlled primarily by the
temperature and light patterns in the area. It is impossible to determine whether the plants
observed at Fitzroy Island in 1989 and 1990 were germinated from seeds produced in
previous years (i.e. 1988 and 1989, respectively). Germination in the laboratory at 26-28OC
suggests that germination in H. tricostata is least likely in the low temperatures o f autumn
and winter and most likely when water temperatures rise with spring conditions. The wide
variation in the timing o f germination at 26-28OC assures continuous germination during
summer. Furthermore, seeds o f H. tricostata remain dormant i f buried in the sediment or
kept in darkness and germinate only when exposed to light. Seeds o f H. engelmannii are
similar in this respect (McMillan 1987b, 1988a; Jewett-Smith and McMillan 1990), but the
mechanisms o f both intrinsic and extrinsic factors in seagrass seed germination, as discussed
by Birch (1981), are still little understood.
Although the phenology o f H. tricostata in the Great Barrier Reef is not fully recorded,
the continuous field observations at Fitzroy Island indicate that, unlike most other Halophila
species, which are perennial, H. tricostata is an annual species. Halophila tricostata
germinates from seed, grows, and produces flowers and fruits all within a period o f a few
months, from about September-October to December-January each year. At the time
o f seed collection in November-December 1989 and 1990, all plants were reproductive.
However, living plants were almost absent from the Fitzroy Island site during the other
periods o f the year. Late austral spring blooms o f H. tricostata appear to germinate from
presumably dense beds o f seeds, which remain dormant in the fine mud and carbonate
substratum during the austral autumn to early spring. This would indicate that there is only
a brief period in which conditions are suitable for seed germination, seedling establishment,
plant growth and development, and reproduction in H. tricostata.
Plants o f H. tricostata from Fitzroy Island had, on average, 41 seeds per fruit, higher
than any recorded in the literature for the Halophila species. High numbers o f seeds per
fruit have also been observed in the annual H. decipiens from Panama (McMillan and Soong
1989) and south-western Australia (Kuo and Kirkman 1992 and unpublished observation).
The mean number o f H. decipiens fruits per square metre reported varies with depth at
St Croix in the US Virgin Islands: 10.4 fruits m-2 at 15 m , 9.3 fruits m-2 at 21 m and
7.1 fruits m-2 at 27 m, with the mean number o f seeds per fruit being 35.3 (Josselyn
et al. 1986). McMillan (1988~)
recorded 27-48 (mean 36.8) seeds per fruit from 20 fruits o f
H. decipiens from shallow water in Panama. In Halophila species, the lowest average
numbers o f seeds per fruit reported were 8.6 for H. ovalis from south-western Australia
(Kuo and Kirkman 1992), 7.1 for H. engelmannii from Redfish Bay, Texas (McMillan
1987a),and 1-4 for H. beccarii from southern India (Parthasarathy et al. 1988~).
The mean
seed production for H. tricostata at Fitzroy Island, in the Great Barrier Reef, is calculated
as 70000 seeds m - 2 , which far exceeds the reported 13 500 seeds m - 2 for H. decipiens
at Toro Point, Panama (McMillan 1988b), and 74 seeds m - 2 for H. engelmannii in
Occurrence and Fruits and Seeds of H. tricostata
Redfish Bay, Texas (McMillan 1986, 1987a, 1988~).The high number of seeds per fruit in
H. tricostata further supports this species as being annual and its only means of survival
as being seed germination. Keddy (1987) found that the seed production of the annual
eelgrass Zostera marina L. was seven times that of perennial shoots. Most other seagrasses
are perennial and can therefore be propagated both through sexual reproduction by seed
production and through asexual reproduction by rhizome extension or vegetative propagule
formation (see Kuo and McComb 1989).
The fruits of most Halophila species normally develop on the rhizome, which is buried
in sediment, resulting in narrow seed dispersal. In contrast, the fruits of H. tricostata are
borne on erect shoots at the centre of a rosette of leaves, and the fruits float and are
dispersed by water currents after detachment from the erect shoot, which would result in
the wide dispersal of H. tricostata. The negatively buoyant seeds would settle t o the bottom
after the fruit dehisces. Ongoing collection surveys are required t o augment the list of
known sites for H. tricostata. Extensive surveys to date have failed t o find evidence of
this species outside the latitudinal range reported here (Lee Long et al. 1993). However,
collections from trawler operators indicate that, within the known latitudinal range,
H. tricostata may be a widely distributed annual species.
Evidence of deep-water (up to 23 m) feeding by dugong (Dugong dugon) o n seagrasses
in the Great Barrier Reef lagoon (Lee Long et al. 1989), and the well documented association between seagrasses and commercially important penaeid prawns (Staples 1984; Coles
and Lee Long 1985), warrant further investigations of the ecology of H. tricostata and the
role of this species in the overall ecology of the Great Barrier Reef lagoon.
Acknowledgments
We acknowledge the assistance of G. Chisholm, who was vessel skipper and boatman
during the surveys and monitoring and who also provided valuable assistance in diving-based
studies. We also thank R. Christian, J. Coffey and M. Stevens for technical assistance in
microscopical investigations and B. Bright for reading a n early draft of this paper. This
project was supported by the Australian Research Council, the Great Barrier Reef Marine
Park Authority and the Fishing Industry Research and Development Council.
References
Arber, A. (1925). 'Monocotyledons.' (Cambridge University Press: London.)
Balfour, I. B. (1878). On the genus Halophila. Botanical Society of Edinburgh Transactions 13,
290-343.
Birch, W. R. (1981). Morphology of germinating seeds of the seagrass Halophila spinulosa (R. Br.)
Aschers. (Hydrocharitaceae). Aquatic Botany 11, 79-90.
Coles, R. G., and Lee Long, W. J. (1985). Juvenile prawn biology and the distribution of seagrass
prawn nursery grounds in the south-eastern Gulf of Carpentaria, Queensland. In 'Second Australian
National Prawn Seminar'. (Eds P. C. Rothlisberg, B. J. Hill and D. J. Staples.) pp. 55-60. (NPS2:
Cleveland, Australia.)
Coles, R. G., Lee Long, W. J., Squire, B. A., Squire, L. C., and Bibby, J. M. (1987a). Distribution
of seagrasses and associated juvenile commercial penaeid prawns in north-eastern Queensland waters.
Australian Journal of Marine and Freshwater Research 38, 103-19.
Coles, R. G., ~ e l l o r s ,J. E., Bibby, J. M., and Squire, B. A. (1987b). Seagrass beds and juvenile
prawn nursery grounds between Bowen and Water Park Point. Queensland Department of Primary
Industries Information Series No. QI87021.
Coles, R. G., Lee Long, W. J., Helmke, S. E., Bennett, R. E., and Derbyshire, D. J. (1992). Seagrass
beds and juvenile prawn and fish nursery grounds, Cairns to Bowen. Queensland Department of
Primary Industries Information Series No. QI92012.
den Hartog, C. (1970). 'The Sea-grasses of the World.' (North-Holland Publishing: Amsterdam.)
J. Kuo et al.
Greenway, M. (1979). Halophila tricostata (Hydrocharitaceae), a new species of seagrass from the Great
Barrier Reef region. Aquatic Botany 7, 67-70.
Jewett-Smith, J., and McMillan, C. (1990). Germination and seedling development of Halophila
engelmannii Aschers. (Hydrocharitaceae) under axenic conditions. Aquatic Botany 36, 167-77.
Josselyn, M., Fonseca, M., Niesen, T., and Larson, R. (1986). Biomass, production and decomposition
of a deep water seagrass, Halophila decipiens Ostenf. Aquatic Botany 25, 47-61.
Kaul, R. B. (1978). Morphology of germination and establishment of aquatic seedling in Alismataceae
and Hydrocharitaceae. Aquatic Botany 5, 139-47.
Keddy, C. J. (1987). Reproduction of annual eelgrass: variation among habitats and comparison with
perennial eelgrass (Zostera marina L.). Aquatic Botany 27, 243-56.
Kuo, J., and Kirkman, H. (1992). Fruits, seeds and germination in the seagrass Halophila ovalis
(Hydrocharitaceae). Botanica Marina 35, 197-204.
Kuo, J., and McComb, A. J. (1989). Seagrass taxonomy, structure and development. In 'Biology
of Seagrasses: A Treatise on the Biology of Seagrasses with Special Reference to the Australian
Region'. (Eds A. W. D. Larkum, A. J. McComb and S. A. Shepherd.) pp. 6-73. (Elsevier:
Amsterdam.)
Kuo, J., Iizumi, H., Nilsen, B. E., and Aioi, K. (1990). Fruit anatomy, seed germination and seedling
development in the Japanese seagrass Phyllospadix (Zosteraceae). Aquatic Botany 37, 229-45.
Kuo, J., Coles, R. G., Lee Long, W. J., and Mellors, J. (1991). Fruits and seeds of Thalassia
hemprichii (Hydrocharitaceae) from Queensland, Australia. Aquatic Botany 40, 165-73.
Lakshmanan, K. K. (1963). Embryological studies in the Hydrocharitaceae. 11. Halophila ovata
Gaudich. Journal of the Indian Botanical Society 42, 15-18.
Lee Long, W. J., Coles, R. G., Helmke, S. A., and Bennett, R. E. (1989). Seagrass habitats in coastal,
mid shelf and reef waters from Lookout Point to Barrow Point in north-eastern Queensland.
Queensland Department of Primary Industries Information Series (in press).
Lee Long, W. J., Coles, R. G., Derbyshire, K. J., Miller, K. J., and Vidler, K. P. (1992). Seagrass
beds and juvenile prawn and fish nursery grounds, Water Park Point to Hervey Bay, Queensland.
Queensland Department of Primary Industries Information Series No. QI92011.
Lee Long, W. J., Mellors, J. E., and Coles, R. G. (1993). Seagrasses between Cape York and Hervey
Bay, Queensland, Australia. Australian Journal of Marine and Freshwater Research 44, 19-31.
Maiden, J. H., and Betche, E. (1909). Notes from the Botanical Gardens, No. 15. On a plant, in
fruit, doubtfully referred to Cymodocea. Proceedings of Linnean Society of New South Wales
34, 585-7.
McMillan, C. (1986). Fruits and seeds of Halophila engelmannii (Hydrocharitaceae) in Texas. Contributions in Marine Science 19, 1-8.
McMillan, C. (1987~).Recurrence of fruiting of Halophila engelmannii (Hydrocharitaceae) in Redfish
Bay, Texas. Contributions in Marine Science 30, 105-8.
McMillan, C. (19876). Seed germination and seedling morphology of the seagrass, Halophila engelmannii
(Hydrocharitaceae). Aquatic Botany 28, 179-88.
McMillan, C. (1988~).The seed reserve of Halophila engelmannii (Hydrocharitaceae) in Redfish Bay,
Texas. Aquatic Botany 30, 253-9.
McMillan, C. (19886). Seed germination and seedling development of Halophila decipiens Ostenfeld
(Hydrocharitaceae) from Panama. Aquatic Botany 31, 169-76.
McMillan, C. (1988~).The seed reserve of Halophila decipiens Ostenfeld (Hydrocharitaceae) in Panama.
Aquatic Botany 31, 177-82.
McMillan, C., and Jewett-Smith, J. (1988). The sex ratio and fruit production of laboratory germinated
seedlings of Halophila engelmannii Aschers. (Hydrocharitaceae) from Redfish Bay, Texas. Aquatic
Botany 32, 329-39.
McMillan, C., and Soong, K. (1989). An annual cycle of flowering, fruiting and seed reserve for
Halophila decipiens Ostenfeld (Hydrocharitaceae) in Panama. Aquatic Botany 34, 375-9.
O'Brien, T. P., and McCully, M. E. (1981). 'The Study of Plant Structure. Principles and Selected
Methods.' (Termarcarphi: Melbourne.)
Orpurt, D. A., and Boral, L. L. (1964). The flowers, fruits and seeds of Thalassia testudinum Konig.
Bulletin of Marine Science, the Gulf and Caribbean 14, 296-302.
Parthasarathy, N., Ravikumar, K., and Ramamurthy, K. (1988~).Floral biology and ecology of
Halophila beccarii Aschers. (Hydrocharitaceae). Aquatic Botany 31, 141-51.
Occurrence and Fruits and Seeds of H. tricostata
Parthasarathy, N., Ravikumar, K., and Ramamurthy, K. (1988b). Halophila decipiens Ostenf. in
southern India. Aquatic Botany 32, 179-85.
Staples, D. J. (1984). Habitat requirements of juvenile prawns. In 'The Potential of Aquaculture in
Queensland'. (Eds B. R. Pollock and R. H. Quinn.) pp. 87-102. (Queensland Department of Primary
Industries Conference Workshop Series No. QC83012.)
Manuscript received 14 May 1991; revised and accepted 18 March 1992