* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 5: The Working Cell
William Flynn Martin wikipedia , lookup
Open energy system models wikipedia , lookup
Energy subsidies wikipedia , lookup
100% renewable energy wikipedia , lookup
Energy storage wikipedia , lookup
Kinetic energy wikipedia , lookup
Potential energy wikipedia , lookup
Regenerative brake wikipedia , lookup
Public schemes for energy efficient refurbishment wikipedia , lookup
Low-Income Home Energy Assistance Program wikipedia , lookup
Zero-energy building wikipedia , lookup
World energy consumption wikipedia , lookup
Low-carbon economy wikipedia , lookup
Energy Charter Treaty wikipedia , lookup
Alternative energy wikipedia , lookup
Life-cycle greenhouse-gas emissions of energy sources wikipedia , lookup
International Energy Agency wikipedia , lookup
Energy policy of the United Kingdom wikipedia , lookup
Energy returned on energy invested wikipedia , lookup
Energy harvesting wikipedia , lookup
Distributed generation wikipedia , lookup
Energy efficiency in transport wikipedia , lookup
Energy policy of Finland wikipedia , lookup
Internal energy wikipedia , lookup
Energy in the United Kingdom wikipedia , lookup
Negawatt power wikipedia , lookup
Gibbs free energy wikipedia , lookup
Conservation of energy wikipedia , lookup
Energy policy of the European Union wikipedia , lookup
United States energy law wikipedia , lookup
Energy efficiency in British housing wikipedia , lookup
Energy Independence and Security Act of 2007 wikipedia , lookup
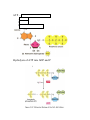
Chapter 5: The Working Cell Energy and Work Thousands of chemical reactions taking place inside cells and organisms. o Many of these reactions require an investment of energy. Energy – the capacity to perform work. o Work – when an object is moved against an opposing force: gravity, friction, rearrange atoms in matter. Two Types of Energy o Kinetic Energy – Energy of motion Moving objects perform work by transferring motion to other matter. o Potential Energy – Energy of position or stored energy Based on location or structure. http://www.youtube.com/watch?v=vl4g7T5gw1M http://www.schooltube.com/video/f147084ab011fc64a890/ Kinetic vs. Potential: Circle Potential and Box Kinetic (Both?) Questions: Quiz Yourself 1. Which letter shows the ball when it has the maximum kinetic energy? D 2. Which letter shows the ball when it has the maximum potential energy? A 3. Which letter shows the ball when it has just a little more kinetic energy than A? G 4. Which letter shows the ball when it has the least potential energy? D 5. Which letter shows the ball when it has just a little less potential energy than letter F? C 6. Which sequence correctly shows an increase in kinetic energy? A. E, F, B, G B. B, F, E, C C. D, E, B, F D. A, G, F, C D Types of Potential Energy Gravitation potential energy Elastic potential energy Electric potential energy Magnetic potential energy Nuclear potential energy Chemical potential energy o Molecules have potential energy because …. of the stored energy in the covalent bonds between atoms. Types of Kinetic Energy Thermal energy Light energy THERMODYNAMICS The study of energy transformations that occur in a collection of matter. Matter – “system” could be 1 cell or the whole planet All else – “surrounding” Living organisms are open systems Exchange both energy and matter with their surroundings. 1st Law of Thermodynamics Total amount of energy in the universe is constant. Energy is not created or destroyed, it is transformed and transferred. Convert sun energy into chemical energy (energy stored in the bonds between a molecule’s atoms) 2nd Law of Thermodynamics Energy conversions reduce the order of the universe and increase Entropy. Entropy – the amount of disorder in a system. During energy conversions some energy becomes unusable (not used for work). This is usually in the form of heat. Heat is a “low” form of energy. Cells use energy to form ordered structures from less organized starting materials. Where does the energy come from? Cells convert “high” ordered forms of energy into “lower” ordered energy and molecules. The difference in energy is used to form ordered structures. Ex: Cellular Respiration Glucose + O2 ATP + CO2 + H2O + Heat (High energy) (Usable energy) (Low energy) Chemical Reactions Store/Release Energy Two types of energy reactions: 1. Endergonic Reactions – Chemical reactions that require a net investment/input of energy. Molecules with low potential absorb energy from the surroundings producing products that have greater stored potential energy. o Energy stored in the covalent bonds between atoms. 2. Exergonic Reactions – Chemical reactions that release energy. Reactants start with high potential energy in their bonds. Products have less potential energy. Energy is released when bonds are broken. o Energy released is used for survival, growth and reproduction. Δ G of a Reaction = Change in Free Energy (Energy of Products – Energy of Reactants) Endergonic Reactions have positive Δ G. Exergonic Reactions have negative Δ G. Photosynthesis – uses the energy from sunlight to convert inorganic molecules into high energy organic molecules. Cellular Respiration – uses organic molecules and oxygen to convert stored chemical potential energy in food to “usable” potential chemical energy - ATP. Cellular Metabolism – the sum total of all the chemical reactions (endergonic and exergonic) taking place in an organism. Energy Coupling the use of energy released from exergonic reactions is necessary for endergonic reactions. ATP – Adenosine Triphosphate Adenine Ribose Phosphate Groups (3) Hydrolysis of ATP into ADP and P ATP – ADP + P Cycle Exergonic Reaction: Releases Energy Energy used to synthesize ATP from ADP and P Endergonic Reaction: Require Energy Energy used for cellular work (chemical, mechanical and transport) Three Types of Cellular Work: 1. Chemical Work – Phosphorylation of a reactant molecule drives an endergonic reaction that produces a high energy product molecule. 2. Mechanical Work – Phosphorylation of a motor protein in muscle cells cause the proteins to change shape, pull on microfilaments (actin), causing the cell to contract. 3. Transport Work – Phosphorylation of proteins pumps (transport proteins) embedded in the plasma membrane allows molecules to move from areas of low concentration to areas of high concentration. Enzymes and Chemical Reactions Cells must invest energy to start chemical reactions. This prevents the spontaneous breakdown of molecules with high potential energy. Energy of Activation (EA) or Activation Energy - the energy that must be absorbed to start a chemical reaction. o What does the energy do? Contorts or weaken bonds before the reaction forms new bonds. If cells need certain chemical reactions to happen quickly, why not increase kinetic energy (heat)? Speed up all chemical reactions (not good) Denature proteins Why use enzymes (special enzyme catalysts)? Speed up specific chemical reactions by lowering the Energy of Activation. o How do they work? Position molecules for bonding or weaken bonds before breaking. Enzymes are proteins. Remember protein shape is important to function. Enzymes generally end in –ase and are named after the substrate they bind. Enzymes Are Specific Enzymes are proteins whose 3-D shape determines their function/role in a chemical reaction. The reactant(s) that a specific enzymes acts upon is called the substrate. The specific region on the enzyme where the substrate binds is called the active site. Enzymes catalyze specific reactions because only certain substrate molecules fit into the active site. o When the substrate binds to the active site, the shape changes (induced fit) to weaken bonds for breaking or positions molecules close together for bonding. Enzyme hydrolysis of sucrose Enzymes and Environmental Conditions An enzyme’s shape determines its function, and the shape can be affected by the environment. Enzymes are most effective under specific conditions. Temperature – close to normal cell/body temperature Salinity – high salt ions interfere with chemical bonds pH – close to neutral, H+ interfere with bonding Cofactors (inorganic) and Coenzymes (organic) are ions or molecules that help enzymes function. Enzyme Inhibitors An enzyme’s ability to bond with its substrate can be interfered with by chemicals called inhibitors. Two types of inhibitors: Competitive Inhibitors – have structures like the substrate and bind to the enzyme’s active site before the substrate. Add more substrate to increase productivity. Noncompetitive Inhibitors – bind to a secondary location on the enzyme (allosteric site) that changes the shape of the active site preventing the substrate from binding. http://www.youtube.com/watch?v=PILzvT3spCQ Inhibition Hydrogen bonded inhibitors are weakly held o Reversible Covalently bonded inhibitors are strongly held o Irreversible Enzyme inhibition is not always bad. Some chemical reactions require multiple steps. Products can inhibit further steps if in excess. This is called feedback inhibition. Beneficial drug o Ibuprofen and aspirin – pain inhibition o Protease – HIV inhibitors o Cancer drugs – cell division inhibitors Harmful inhibition Toxins and poisons o Cyanide – ATP inhibition o Sarin gas – nerve impulse inhibition o Pesticides – nerve impulse inhibition o Antibiotics – cell wall inhibition