* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Percutaneous Mitral Valve Repair
Cardiac contractility modulation wikipedia , lookup
Remote ischemic conditioning wikipedia , lookup
Cardiothoracic surgery wikipedia , lookup
Pericardial heart valves wikipedia , lookup
Coronary artery disease wikipedia , lookup
Artificial heart valve wikipedia , lookup
History of invasive and interventional cardiology wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
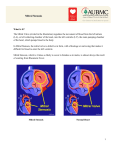
JACC: CARDIOVASCULAR INTERVENTIONS VOL. 4, NO. 7, 2011 © 2011 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/$36.00 PUBLISHED BY ELSEVIER INC. DOI: 10.1016/j.jcin.2011.05.010 ACC INTERVENTIONAL SCIENTIFIC COUNCIL: NEWS AND VIEWS Percutaneous Mitral Valve Repair Lessons From the EVEREST II (Endovascular Valve Edge-to-Edge REpair Study) and Beyond Jon C. George, MD,* Vincent Varghese, DO,* George Dangas, MD, PHD,† Ted E. Feldman, MD‡ Browns Mills, New Jersey; New York, New York; and Evanston, Illinois The 24-month results of the EVEREST II (Endovascular Valve Edge-to-Edge REpair Study), presented at the American College of Cardiology 2011 Scientific Sessions/i2 Summit and recently published (1), investigating the safety and efficacy of the MitraClip (Abbott Vascular, Santa Clara, California) device, validated the interest in percutaneous approaches to the treatment of mitral regurgitation (MR) compared with the current gold standard, which is surgical mitral valve repair or replacement (2). The MitraClip is a percutaneously inserted device that mimics the surgical edge-to-edge repair developed by Dr. Ottavio Alfieri in 1991, by attaching to the tips of the anterior and posterior mitral leaflets, securing them together to form a double-orifice mitral inlet, and thereby reducing the amount of regurgitation (3). Trial Results and Implications The EVEREST II randomized 279 patients with severe (3 to 4⫹) MR to percutaneous mitral valve clip or open surgical valve repair or replacement. Patients were followed for 30 days and 1 year with primary endpoints of major adverse events and clinical success rate, respectively. Clinical success was defined as freedom from death, open mitral valve repair or replacement, and improvement of at least 2 grades of MR. At 30 days, there was a significant reduction in the composite endpoint of major adverse events (including death, myocardial infarction, stroke, reoperation, blood transfusion, and From the *Division of Interventional Cardiology and Endovascular Medicine, Deborah Heart and Lung Center, Browns Mills, New Jersey; †Division of Interventional Cardiology, Mount Sinai Medical Center, New York, New York; and the ‡Cardiology Division, Evanston Hospital, Evanston, Illinois. Dr. George is a consultant for Boston Scientific and ev3 Endovascular. Dr. Feldman received research grants from and is a consultant for Abbott, Boston Scientific, and Edwards Lifesciences. All other authors have reported that they have no relationships to disclose. renal failure) at 9.6% in the percutaneous mitral clip group compared with 57% in the surgical group. At 1 year, clinical success was higher in the surgically treated group compared with the mitral clip arm. At 2 years, intention-to-treat analysis revealed that the primary composite endpoints were still significantly better in the surgery group compared with the mitral clip group. The conclusion of the EVEREST II is that percutaneous mitral valve repair, although less effective at reducing MR, is not inferior to surgical repair or replacement, with a significantly reduced adverse event rate; however, several questions remain. First, an edge-to-edge repair would only be suitable for a select group of patients with central MR or flail leaflet and would not address the other leaflet morphologies of MR. Almost one-third of patients in the MitraClip group had functional MR with reportedly no difference in outcome (reduced left ventricular volume/dimension and improved symptoms) compared with the degenerative MR patients; however, the durability of this result beyond 2 years remains to be seen. Second, the improvement in symptoms and left ventricular size, although similar in both groups, may begin to diverge as patients are followed because a 3-fold greater percentage of device-treated patients had 2⫹ or greater MR severity compared with surgically treated patients. In addition, although most patients remain candidates for surgical repair after MitraClip therapy, there have also been reports of mitral clip patients with persistent severe MR who, due to significant mitral leaflet scarring, are unable to have surgical mitral valve repair and must undergo valve implantation. More than 15% of patients in the device arm subsequently underwent mitral valve surgery due to persistent severe MR. The inclusion of requiring blood transfusions, instead of the incidence of major bleeding, as an 826 George et al. Percutaneous Mitral Valve Repair adverse event in the study has drawn criticism; however, it should be noted that adverse events continue to favor device-treated patients even if major bleeding is taken into account and that transfusion after cardiac surgery is associated with increased acute and long-term mortality (4 – 8). Summary and Future Direction The EVEREST II was a novel evaluation that compared percutaneous mitral valve repair with surgical repair or replacement. Although the study demonstrated the feasibility of a percutaneous treatment option for select patients with severe functional MR, it is currently not yet approved by the U.S. Food and Drug Administration (FDA) to supplant surgical mitral valve repair or replacement. Although the EVEREST II has completed enrollment, further study and long-term follow-up are ongoing with the REALISM (Real World Expanded Multicenter Study of the MitraClip System), a continued access registry. The MitraClip device has received CE Mark approval in Europe and is currently under review for FDA approval in the United States. Meanwhile several other technologies are being developed as alternative strategies for the management of MR (9). The AMADEUS (cArillon Mitral Annuloplasty Device European Union Study) (10) evaluated the use of the Carillon device (Cardiac Dimensions, Kirkland, Washington), which consists of an arched nitinol wire with looped anchors at each end that is delivered percutaneously via the coronary sinus and anchored in the great cardiac vein, restoring the natural shape of the mitral annulus by modifying the tension of the device. At 6 months, MR reduction and improvement in 6-min walk distance and quality of life were achieved with permanent implantation in 30 of 48 patients enrolled (11). Two indirect annuloplasty devices are no longer being studied, but some clinical results are reported. The EVOLUTION phase I study (Clinical Evaluation of the Edwards Lifesciences Percutaneous Mitral Annuloplasty System for the Treatment of Mitral Regurgitation) (12) used the MONARC coronary sinus device (Edwards Lifesciences, Irvine, California), which consists of 2 stent anchors with a connecting spring bridge and a biodegradable element within the bridge. At 12 months, MR reduction more than 1 grade was observed in 50% of implanted patients and in 85.7% of patients with baseline MR more than 3 grades (12). The EVOLUTION phase II study of a second-generation coronary sinus ring for patients with at least 3⫹ MR has been concluded, but results have not yet been made available. The Percutaneous Mitral Annuloplasty (PTMA) device (Viacor Inc., Wilmington, Massachusetts) is a trilumen plastic cannula that is used to deliver nitinol rods into the coronary sinus to compress the septolateral dimension and allow improved leaflet coaptation. MR reduction was observed in 13 of 19 patients who received a diagnostic PTMA device (13). JACC: CARDIOVASCULAR INTERVENTIONS, VOL. 4, NO. 7, 2011 JULY 2011:825–7 Two direct mitral annuloplasty approaches including the Mitralign system (Mitralign, Tewksbury, Massachusetts) and the Guided Delivery Systems device (Guided Delivery Systems, Santa Clara, California) have undergone first-inhuman feasibility studies, but larger clinical outcome trials are pending. The MitraClip is the first step in catheter-based strategy for percutaneous treatment of MR. The initial study describing the surgical Alfieri technique for treatment of MR (14) demonstrated 22% greater freedom from reoperation when double-orifice repair was combined with an annuloplasty procedure. The effectiveness of percutaneous mitral valve repair may be improved when performed in conjunction with the use of other catheter-based annuloplasty devices that are currently in development. At this point, it is clear that the MitraClip procedure is safe, with clinical benefits in selected patients who are stable at least for 2 years. The regulatory steps involved in evaluation for FDA approval and the subsequent rate of incorporation into clinical practice will determine future developments of the MitraClip and other mitral valve devices. Reprint requests and correspondence: Dr. Jon C. George, Division of Interventional Cardiology and Endovascular Medicine, Deborah Heart and Lung Center, 200 Trenton Road, Browns Mills, New Jersey 08015. E-mail: [email protected]. REFERENCES 1. Feldman T, Foster E, Glower DG, et al. Percutaneous repair or surgery for mitral regurgitation. N Engl J Med 2011;364:1395– 406. 2. Bonow RO, Carabello BA, Chaterjee K, et al. ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1998 Guidelines for the Management of Patients With Valvular Heart Disease). J Am Coll Cardiol 2006;48:e1–148. 3. Feldman T, Kar S, Rinaldi M, et al. Percutaneous mitral repair with the MitraClip System. Safety and midterm durability in the initial EVEREST (Endovascular Valve Edge-to-Edge REpair Study) cohort. J Am Coll Cardiol 2009;54:686 –94. 4. Engoren MC, Habib RH, Zacharias A, et al. Effect of blood transfusion on long-term survival after cardiac operation. Ann Thorac Surg 2002;74:1180 – 6. 5. Koch CG, Li L, Duncan AI, et al. Transfusion in coronary artery bypass grafting is associated with reduced long-term survival. Ann Thorac Surg 2006;81:1650 –7. 6. Surgenor SD, DeFoe GR, Fillinger MP, et al. Intraoperative red blood cell transfusion during coronary artery bypass graft surgery increases the risk of postoperative low-output heart failure. Circulation 2006;114 Suppl:I43– 8. 7. Kuduvalli M, Oo AY, Newall N, et al. Effect of peri-operative red blood cell transfusion on 30-day and 1-year mortality following coronary artery bypass surgery. Eur J Cardiothorac Surg 2005;27: 592– 8. 8. Koch CG, Li L, Duncan AI, et al. Morbidity and mortality risk associated with red blood cell and blood-component transfusion in isolated coronary artery bypass grafting. Crit Care Med 2006; 34:1608 –16. 9. Feldman T, Cilingiroglu M. Percutaneous leaflet repair and annuloplasty for mitral regurgitation. J Am Coll Cardiol 2011;57:529 –37. JACC: CARDIOVASCULAR INTERVENTIONS, VOL. 4, NO. 7, 2011 JULY 2011:825–7 10. Siminiak T, Hoppe UC, Schofer J, et al. Effectiveness and safety of percutaneous coronary sinus-based mitral valve repair in patients with dilated cardiomyopathy (from the AMADEUS trial). Am J Cardiol 2009;104:565–70. 11. Schofer J, Siminiak T, Haude M, et al. Percutaneous mitral annuloplasty for functional mitral regurgitation: results of the CARILLON Mitral Annuloplasty Device European Union Study. Circulation 2009; 120:326 –33. 12. Harnek J, Webb JG, Kuck KH, et al. Transcatheter implantation of the MONARC coronary sinus device for mitral regurgitation 1-year results George et al. Percutaneous Mitral Valve Repair 827 from the EVOLUTION phase I study (Clinical Evaluation of the Edwards Lifesciences Percutaneous Mitral Annuloplasty System for the Treatment of Mitral Regurgitation). J Am Coll Cardiol Intv 2011;4:115–22. 13. Sack S, Kahlert P, Bilodeau L, et al. Percutaneous transvenous mitral annuloplasty: initial human experience with a novel coronary sinus implant device. Circ Cardiovasc Interv 2009;2:277– 84. 14. Alfieri O, Maisano F, De Bonis M, et al. The double-orifice technique in mitral valve repair: a simple solution for complex problems. J Thorac Cardiovasc Surg 2001;122:674 – 81.