* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Acetic anhydride Product Number A6404 Store at - Sigma
Survey
Document related concepts
Transcript
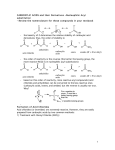
Acetic anhydride Product Number A6404 Store at Room Temperature Product Description Molecular Formula: C4 H6O3 Molecular Weight: 102.1 CAS Number: 108-24-7 1 Boiling Point: 139 °C 1 Density: 1.082 g/ml Molarity: 10.6 M (calculated from density and molecular weight) 1 Synonyms: acetic oxide, acetyl oxide Acetic anhydride is a reagent that is used in the manufacturing of acetyl compounds and cellulose acetates. It is also utilized as a solvent in the examination of wool fat, glycerol, fatty and volatile oils, 1 and resins. In the synthesis of organic compounds, acetic anhydride is used in various reactions, including the cleavage of dialkyl ethers, the formation of anhydrides from carboxylic acids, the addition of methylthiomethyl groups to phenol rings, and the addition of acyl and acyloxy groups across double bonds. Acetic anhydride will also react with such 2 acetals, alcohols, and sulfoxides. A wide variety of substrates and materials has been synthesized using acetic anhydride. Small molecule syntheses include the preparation of substituted 2-pyridones, acylated silyl ketene acetals, and estrogen receptor ligands derived from 3,4,5 2,3-diarylpiperazines. Larger materials which have been prepared with acetic anhydride include cellulose and poly(ethylene oxide)-block-poly(N-hexyl-L6,7 aspartamide)-acyl conjugates. Acetic anhydride has been used to hyperacetylate histones to investigate transcriptional effects of the NAD-dependent histone deacetylase Sir2 in Drosophila on a complex of the hyperacetylated 8 histones with DNA. An affinity chromatography/mass spectrometry method for the analysis of neuroendocrine peptides from carboxypeptidase E 9 deficient mice has been reported. A microarray of oligonucleotide probes linked to a solid support that uses acetic anhydride in the preparation 10 has been described. Acetic anhydride is utilized in an assay of a fluorescently labeled peptide catalyst library for application to the resolution of secondary 11 alcohols. Precautions and Disclaimer For Laboratory Use Only. Not for drug, household or other uses. Preparation Instructions This product is miscible in chloroform and ether. In alcohol, this product will form ethyl acetate. In water (0.1 ml/ml, v/v), this product will slowly form acetic 1 acid. References 1. The Merck Index, 12th ed., Entry# 53. 2. Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 4th ed., March, J., John Wiley & Sons (New York, NY: 1992), pp. 600, 822, 1173. 3. Padwa, A., et al., Synthesis of substituted 2-pyridones via the pummerer cyclizationdeprotonation-cycloaddition cascade of imidosulfoxides. J. Org. Chem., 64(6), 2038-2049 (1999). 4. Mermerian, A. H., and Fu, G. C., Catalytic enantioselective synthesis of quaternary stereocenters via intermolecular C-acylation of silyl ketene acetals: dual activation of the electrophile and the nucleophile. J. Am. Chem. Soc., 125(14), 4050-4051 (2003). 5. Gust, R., et al., Synthesis, structural evaluation, and estrogen receptor interaction of 2,3diarylpiperazines. J. Med. Chem., 45(11), 2325-2337 (2002). 6. Chauvelon, G., et al., Preparation of sulfoacetate derivatives of cellulose by direct esterification. Carbohydr. Res., 338(8), 743-750 (2003). 7. 8. 9. Adams, M. L., and Kwon, G. S., The effects of acyl chain length on the micelle properties of poly(ethylene oxide)-block-poly(N-hexyl-Laspartamide)-acyl conjugates. J. Biomater. Sci. Polym. Ed., 13(9), 991-1006 (2002). Parsons, X. H., et al., Histone deacetylation by Sir2 generates a transcriptionally repressed nucleoprotein complex. Proc. Natl. Acad. Sci. USA, 100(4), 1609-1614 (2003). Che, F. Y., and Fricker, L. D., Quantitation of fat fat neuropeptides in Cpe /Cpe mice using differential isotopic tags and mass spectrometry. Anal. Chem., 74(13), 3190-3198 (2002). 10. Belosludtsev, Y., et al., DNA microarrays based on noncovalent oligonucleotide attachment and hybridization in two dimensions. Anal. Biochem., 292(2), 250-256 (2001). 11. Copeland, G. T., and Miller, S. J., Selection of enantioselective acyl transfer catalysts from a pooled peptide library through a fluorescencebased activity assay: an approach to kinetic resolution of secondary alcohols of broad structural scope. J. Am. Chem. Soc., 123(27), 6496-6502 (2001). GCY / RXR 5/06 Sigma brand products are sold through Sigma-Aldrich, Inc. Sigma-Aldrich, Inc. warrants that its products conform to the information contained in this and other Sigma-Aldrich publications. Purchaser must determine the suitability of the product(s) for their particular use. Additional terms and conditions may apply. Please see reverse side of the invoice or packing slip.