* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Case 2 - Ram Pages
Heart failure wikipedia , lookup
Remote ischemic conditioning wikipedia , lookup
Electrocardiography wikipedia , lookup
History of invasive and interventional cardiology wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Cardiac surgery wikipedia , lookup
Jatene procedure wikipedia , lookup
Ventricular fibrillation wikipedia , lookup
Coronary artery disease wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
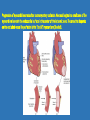
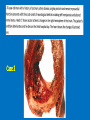
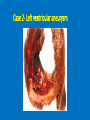
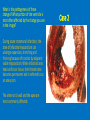
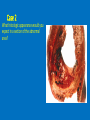
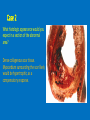
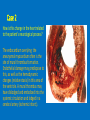
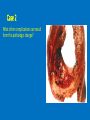
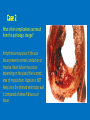
Ischemic Heart Disease Acute Myocardial Infarction Cora Uram-Tuculescu, MD Department of Pathology, VCUHS [email protected] Infarct modification by reperfusion Case 1 1. What changes do you observe in the heart? 2. What is the most likely diagnosis and what was the likely cause? 3. What histologic features would you expect in a section of the grossly abnormal myocardium? 4. Describe the likely pathophysiology leading to death in this case. 5. Describe the sequence of histologic changes that occurs in the myocardium as a result of acute occlusion of flow in a coronary artery. Case 1 Case 1- Acute myocardial infarction with rupture What changes do you observe in the heart? Case 1 What changes do you observe in the heart? The myocardium of the anterior left ventricular wall is diffusely yellow in appearance in contrast to the normal red myocardium adjacent to it. Case 1 What is the most likely diagnosis and what was the likely cause? Case 1 What is the most likely diagnosis and what was the likely cause? Acute transmural MI. The probe demonstrates rupture of the wall. Most likely cause is coronary artery atherosclerosis with occlusive thrombus over a ruptured plaque. Case 1 Describe the likely pathophysiology leading to death in this case. Case 1 Describe the likely pathophysiology leading to death in this case. Acute rupture of the ventricular wall would result in accumulation of blood in the pericardial sac (hemopericardium). This would prevent the heart from filling normally in diastole (tamponade). Case 1 Describe the sequence of histologic changes that occurs in the myocardium as a result of acute occlusion of flow in a coronary artery. Case 1 Temporal sequence of early biochemical findings and progression of necrosis after onset of severe myocardial ischemia. A, Early changes include loss of adenosine triphosphate (ATP) and accumulation of lactate. B, For approximately 30 minutes after the onset of even the most severe ischemia, myocardial injury is potentially reversible. Thereafter, progressive loss of viability occurs that is complete by 6 to 12 hours. The benefits of reperfusion are greatest when it is achieved early, and are progressively lost when reperfusion is delayed. (Modified with permission from Antman E: Acute myocardial infarction. In Braunwald E, et al [eds]: Heart Disease: A Textbook of Cardiovascular Medicine, 6th ed. Philadelphia, WB Saunders, 2001, pp 1114-1231.) The temporal evolution of the morphologic changes in acute MI and subsequent healing Time Gross features Light microscopy EM 0-1/2 hrs None None Relaxation of myofibrils Glycogen loss Mitochondrial swelling ½-4 hrs None Usually none; variable waviness of fibers at border Sarcolemmal disruption Mitochondrial amorphous densities 4-12 hrs Dark mottling (occasional) Early coagulation necrosis; edema; hemorrhage 12-24 hrs Dark mottling Ongoing coagulation necrosis; pyknosis of nuclei; myocyte hypereosinophilia; marginal contraction band necrosis 1-3 days Mottling with yellow-tan infarct center Coagulation necrosis, with loss of nuclei and striations; brisk interstitial infiltrate of neutrophils 3-7 days Hyperemic border; central yellow-tan softening Beginning disintegration of dead myofibers, with dying neutrophils; early phagocytosis of dead cells by macrophages at infarct border 7-10 days Maximally yellow-tan and soft, with depressed red-tan margins Well developed phagocytosis of dead cells; granulation tissue at margins 10-14 days Red-gray depressed infarct borders Well- established granulation tissue with new blood vessels and collagen deposition 2-8 wks Gray-white scar, progressive from border toward core of infarct Increased collagen deposition, with decreased cellularity >2 month Scarring complete Dense collagenous scar Progression of myocardial necrosis after coronary artery occlusion. Necrosis begins in a small zone of the myocardium beneath the endocardial surface in the center of the ischemic zone. The area that depends on the occluded vessel for perfusion is the “at risk” myocardium (shaded). Case 2 1. Describe the gross pathologic changes? What is the most likely diagnosis? 2. What is the pathogenesis of these changes? What portion of the ventricle is most often affected by the change you see in the image? 3. What histologic appearance would you expect in a section of the abnormal area? 4. How is this change in the heart related to the patient’s neurological process? 5. What other complications can result from this pathologic change? Case 2 Case 2- Left ventricular aneurysm Case 2 Describe the gross pathologic changes? What is the most likely diagnosis? Case 2 Describe the gross pathologic changes? What is the most likely diagnosis? A portion of the left ventricular wall is white rather than red in color, and thinner than the adjacent normal myocardium. What is the pathogenesis of these changes? What portion of the ventricle is most often affected by the change you see in the image? Case 2 What is the pathogenesis of these changes? What portion of the ventricle is most often affected by the change you see in the image? During acute transmural infarction, the zone of infarcted myocardium can undergo expansion, stretching and thinning because of traction by adjacent viable myocardium. When infarcted area heals with scar tissue, the thinned zone becomes permanent and is referred to as an aneurysm. The anterior LV wall and the apex are most commonly affected. Case 2 Case 2 What histologic appearance would you expect in a section of the abnormal area? Case 2 What histologic appearance would you expect in a section of the abnormal area? Dense collagenous scar tissue. Myocardium surrounding the scar likely would be hypertrophic, as a compensatory response. Case 2 How is this change in the heart related to the patient’s neurological process? Case 2 How is this change in the heart related to the patient’s neurological process? The endocardium overlying the aneurysmal myocardium often is the site of mural thrombus formation. Endothelial damage may predispose to this, as well as the hemodynamic changes (relative stasis) in this area of the ventricle. A mural thrombus may have dislodged and embolized into the systemic circulation and lodged in a cerebral artery (ischemic infarct). Case 2 What other complications can result from this pathologic change? Case 2 What other complications can result from this pathologic change? Arrhythmias may occur if the scar tissue prevents normal conduction of impulse. Heart failure may occur depending on the size of the scarred area of myocardium. Rupture is NOT likely since the thinned ventricular wall is composed of dense fibrous scar tissue. Key Concepts The vast majority of ischemic heart disease is due to coronary artery atherosclerosis; vasospasm, vasculitis, or embolism are less common causes. Cardiac ischemia results from a mismatch in coronary supply and myocardial demand, and presents as different, albeit overlapping syndromes Acute myocardial infarction typically results from acute thrombosis after plaque disruption; most occur in plaques that did not previously exhibit critical stenosis. Key Concepts Myocardial ischemia leads to loss of function within 1 to 2 minutes, but causes necrosis only after 20 to 40 minutes. Myocardial infarction is diagnosed based on symptoms, EKG changes, and measurement of serum CK-MB and troponins. Gross and histologic changes of infarction require hours to days to develop. Infarction can be modified by therapeutic intervention (e.g., thrombolysis or stenting), which salvages myocardium at risk, but potentially induces reperfusion-related injury. Complications of infarction include: ventricular rupture, papillary muscle rupture, aneurysm formation, mural thrombus, arrhythmia, pericarditis, and CHF.