* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download One slide per page
Survey
Document related concepts
Transcript
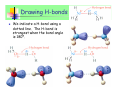
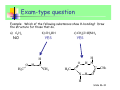
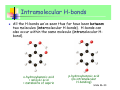
Unless otherwise stated, all images in this file have been reproduced from: Blackman, Bottle, Schmid, Mocerino and Wille, Chemistry, 2007 (John Wiley) ISBN: 9 78047081 0866 Slide 36-1 Chem 1101 A/Prof Sébastien Perrier Room: 351 Phone: 9351-3366 Email: [email protected] Prof Scott Kable Room: 311 Phone: 9351-2756 Email: [email protected] A/Prof Adam Bridgeman Room: 222 Phone: 9351-2731 Slide 36-2 Email: [email protected] Highlights of last lecture Batteries and Corrosion… CONCEPTS Difference between primary and secondary batteries How common batteries work Chemical reactions for common batteries Fuel cells and how they work Corrosion and corrosion protection CALCULATIONS None specifically, except redox calculations involving batteries. Slide 36-3 Intermolecular Forces Themes: Forces that hold molecules together Biopolymers and their structure References: Most general chemistry texts will have a satisfactory chapter on intermolecular forces, e.g. Silberberg: Chap 12.3 (IM Forces) and pp.468-73 (Polymers) Slide 36-4 IM Forces Key chemical concepts: • Different types of forces between molecules • H-bonding • primary, secondary and tertiary structure of polymers Key Calculations: • none Slide 36-5 Q: If matter is so attracted to itself, why doesn’t it squash down to nothing? Answer: There is a balance in energy between the forces of attraction and the strong force of repulsion that atoms experience when they get very close together. The separation distance is known either as the - Bond length (for intramolecular), or the - Van der Waals (VDW) distance (for intermolecular) - these are larger than bond lengths since the forces of attraction are weaker We can assign atomic radii (= dist/2): - Covalent radius and Van der Waals (VDW) radius Slide 36-6 Q: If matter is so attracted to itself, why doesn’t it squash down to nothing? Answer: There is a balance in energy between the forces of attraction and the strong force of repulsion that atoms experience when they get very close together. The separation distance is known either as the - Bond length (for intramolecular), or the - Van der Waals (VDW) distance (for Covalent radius VDW radius intermolecular) - these are larger than bond lengths since the forces of attraction are weaker We can assign atomic radii (= dist/2): - Covalent radius and Van der Waals (VDW) radius Slide 36-7 Intramolecular Forces All attractive and repulsive interaction between atoms arise from electrostatic forces. We have already examined the forces that hold atoms together to form a molecule, which we separate into ionic, covalent and metallic bonds, although there is a continuum between all three of these descriptions. Force Attraction Energy Example Ionic Cation – anion 400-4000 kJ/mol NaCl Covalent Nuclei sharing 150-1100 kJ/mol e- pair Metallic Model Delocalised electrons 75-1000 kJ/mol H-H Fe Slide 36-8 Intramolecular Forces One thing they have in common, however, is that they are all quite STRONG (typically 80-4000 kJ/mol). Making and breaking these bonds requires a lot of energy, which we call a chemical reaction. INTRAmolecular forces are NOT all there is to chemical species. Individual molecules ALSO feel attraction or repulsion from each other. A trivial example is water as a solid (ice), liquid (water) and gas (steam). If there were no attraction between individual water molecules then water would only exist as a gas. Interactions between water molecules (INTERmolecular interactions) are responsible for liquid and solid water. Slide 36-9 Charge distribution Intramolecular bonding is due to electrostatic forces, it should come as no surprise that INTERmolecular forces also arise through electrostatics. We recognise three different ways that the electron distribution can affect the electrostatic effects: 1-Ionic Charge 2-Dipole moment 3-Polarizability 1) Ionic charge In this case the molecule itself has an excess of negative (anion) or positive (cation) charge. Slide 36-10 2) Dipole moment Ionic and polar covalent bonds have an unequal sharing of electrons between the two atoms. In these cases one end of the bond is more negative and the other more positive. If the molecule is a diatomic species then we call the molecule “polar” These “bond dipoles” can be added up in more complicated molecules. Frequently the sum of the bond dipoles gives rise to an overall dipole moment in a polyatomic molecule. However, sometimes, due to symmetry or accident, the bond dipoles can cancel to give rise to a non-polar molecule (even though the individual bonds are polar). Slide 36-11 2) Dipole moment Slide 36-12 2) Dipole moment Blackman Figure 6.28 Slide 36-13 3) Polarizability The electrons on a molecule are never stationary, nor rigidly held. When a molecule is brought into the vicinity of other charges, the electrons on the molecule will move in response to this charge. The freedom of the electrons to move around is called the “polarizability” of the molecule. Small and first row atoms hold their electrons tightly and are not very polarizable. Larger atoms, with electrons in outer orbitals are more polarizable. If an atom or molecule is polarizable, then a small dipole can be induced in the atom/molecule when brought up to another molecule. We call this an “induced dipole”. Slide 36-14 3) Polarizability A dipole may be induced in an atom or non-polar molecule by the presence of an electric field. This process is called polarisation. + - + - + - + - Induced dipole ∝ electric field strength ∝ polarisability of the non-polar species Polarisability is a measure of the ease with which an electron cloud of an atom or molecule is distorted. - increases with number of electrons - increases with size Slide 36-15 Type of IM Forces Slide 36-16 INTERmolecular forces 1) Ion-Dipole: - dipoles align (partially or fully) in the electric field of an ion - typical energy 40-600 kJ/mol (per bond) Slide 36-17 INTERmolecular forces Hydration shells around ions in water Blackman Figure 10.8 Slide 36-18 INTERmolecular forces 2) Dipole-dipole - dipoles align (partially or fully) in the electric field of the neighboring dipole. - typical energy 5-25 kJ/mol Solid Liquid Slide 36-20 INTERmolecular forces 3) Ion-induced dipole - a dipole is induced in a polarizable molecule by the presence of an ion. - typical energy: 3-15 kJ/mol Point of note: - A comparatively rare force, since ions do not often exist in the presence of non-polar species (due to the insolubility of ionic solids in non-polar solvents). Slide 36-21 INTERmolecular forces 4) Dipole – induced dipole - a dipole is induced in a polarizable molecule by the presence of another dipole. - typical energy: 2-10 kJ/mol Points of note: - A comparatively rare force, since dipoles do not often exist in the presence of non-polar species (due to the insolubility/immiscibility of polar molecules in non-polar solvents and vice versa). - Some highly polarisable molecules can have significant solubility in polar solvents (e.g. Xe is 25 times more soluble in H2O than is He) Slide 36-22 INTERmolecular forces 5) Induced dipole – induced dipole (or Dispersion or London forces) - Two polarizable molecules induce transient dipole moments in each other. - typical energy: 0.05 – upwards kJ/mol Points of note: - may be considered as an instantaneous dipole - induced dipole force - occurs between all atoms and molecules Slide 36-23 INTERmolecular forces 5) Induced dipole – induced dipole (or Dispersion or London forces) Generally regarded as being weak, but can in fact be very strong for large molecules e.g. benzene and bromine are liquid at RT I2 and S8 are solid at RT The strength of the interaction depends on two features: • polarisability • surface contact area Slide 36-24 Summary of IM Forces Dispersion forces exist between ALL molecules. The force increase in strength with molecular mass. Forces associated with permanent dipoles are found only in substances with overall dipole moments (polar molecules). Their existence adds to the dispersion forces. When comparing substances of widely different masses, dispersion forces are usually more significant than dipolar forces. When comparing substances of similar molecular mass, dipole forces can produce significant differences in molecular properties (e.g. boiling point). Molecule MW (amu) Dipole moment, (Debye) dispersion dipolar BP (K) F2 38 0 100% 0% 85 HCl 36.5 1.08 81% 19% 188.1 HBr 80.9 0.82 94.5% 5.5% 206.4 HI 127.9 0.44 99.5% 0.5% 237.8 Slide 36-25 Type of IM Forces INTERmolecular forces arises from the combination of any pair of the three interactions we have just examined.* * except ion-ion, which is ionic bonding Slide 36-26 Hydrogen bonds A H-bond arises from an unusually strong dipole-dipole force. When H is bonded to a very electronegative element (F, O, N) the bond is polar covalent. H is unusual because with only one electron, it leaves a partially exposed nucleus (H has no other core electrons to shield the nucleus). The bond can be thought of as forming between the hydrogen atom and the lone pairs of the F, N, or O. HF H2O NH3 Slide 36-27 Only F, O, and N? The H-bond occurs between H-atom and a lone pair. Can other elements with lone pairs form H-bonds? e.g. Cl? F, O and N are all 2nd row elements, which conveys certain properties: They are the most electronegative elements They are small (only 2s, 2p in outer shell) They have lone pairs Consequently, although, e.g. Cl lone pairs DO contribute to the dipole-dipole force, this force is not unusual in size (see plot later in lecture). So we consider H-bonds ONLY to form between bonds involving FH, OH and NH Slide 36-28 Drawing H-bonds We indicate a H-bond using a dotted line. The H-bond is strongest when the bond angle is 180º. Slide 36-29 The Ammonia Fountain NH3 (g) + H2O ⇔ NH3 (aq) NH3 (aq) + H2O ⇔ NH4+ (aq) + OH- (aq) Slide 36-30 Exam-type question Example: Which of the following substances show H-bonding? Draw the structure for those that do. a) C2H6 b) CH3OH NO YES c) CH3(C=O)NH2 YES H H O H H3C O O CH3 H3C H N C C N H CH3 O H Slide 36-31 The special case of water Water is perhaps the most unusual liquid. Each water molecule is H-bonded to FOUR other water molecules (donating 2 Hatoms and accepting two H-atoms to the lone pairs), forming a tetrahedral network. 1 2 4 3 Slide 36-32 Intramolecular H-bonds All the H-bonds we’ve seen thus far have been between two molecules (intermolecular H-bonds). H-bonds can also occur within the same molecule (intramolecular Hbond). o-hydroxybenzoic acid = salicylic acid = metabolite of aspirin p-hydroxybenzoic acid (no intramolecular H-bonding) Slide 36-33 Summary CONCEPTS How to determine whether a molecule is polar Three type of charge distribution: ion, dipole, polarizability Different type of IM Forces Which forces are present in different molecules Which forces are more important Effect of IM Forces on boiling point CALCULATIONS None Slide 36-34