* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Human Physiology An Integrated Approach 6/E
Survey
Document related concepts
Biochemistry wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Gaseous signaling molecules wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
High-altitude adaptation in humans wikipedia , lookup
Human genetic resistance to malaria wikipedia , lookup
Transcript
18
Gas Exchange
and Transport
Gas Exchange in the Lungs and Tissues
Lower Alveolar PO2 Decreases Oxygen Uptake
Diffusion Problems Cause Hypoxia
Gas Solubility Affects Diffusion
Gas Transport in the Blood
Hemoglobin Binds to Oxygen
Oxygen Binding Obeys the Law of Mass Action
Hemoglobin Transports Most Oxygen to the Tissues
PO2 Determines Oxygen-Hb Binding
Oxygen Binding Is Expressed As a Percentage
Several Factors Affect Oxygen-Hb Binding
Carbon Dioxide Is Transported in Three Ways
Regulation of Ventilation
Neurons in the Medulla Control Breathing
Carbon Dioxide, Oxygen, and pH Influence Ventilation
Protective Reflexes Guard the Lungs
Higher Brain Centers Affect Patterns of Ventilation
The successful
ascent of
Everest without
supplementary
oxygen is one of the
great sagas of the
20th century.
—John B. West, Climbing
with O’s, NOVA Online
(www.pbs.org)
Background Basics
Exchange epithelia
pH and buffers
Law of mass action
Cerebrospinal fluid
Simple diffusion
Autonomic and somatic
motor neurons
Structure of the brain
stem
Red blood cells and
hemoglobin
Blood-brain barrier
Giant liposomes
of pulmonary
surfactant (40X)
From Chapter 18 of Human Physiology: An Integrated Approach, Sixth Edition. Dee Unglaub Silverthorn.
Copyright © 2013 by Pearson Education, Inc. All rights reserved.
633
Gas Exchange and Transport
T
he book Into Thin Air by Jon Krakauer chronicles an illfated trek to the top of Mt. Everest. To reach the summit of
Mt. Everest, climbers must pass through the “death zone”
located at about 8000 meters (over 26,000 ft). Of the thousands of
people who have attempted the summit, only about 2000 have been
successful, and more than 185 have died. What are the physiological challenges of climbing Mt. Everest (8850 m or 29,035 ft), and
why did it take so many years before humans successfully reached
the top? The lack of oxygen at high altitude is part of the answer.
The mechanics of breathing includes the events that create bulk flow of air into and out of the lungs. In this chapter we
focus on the two gases most significant to human physiology,
oxygen and carbon dioxide, and look at how they move between
alveolar air spaces and the cells of the body. The process can be
divided into two components: the exchange of gases between
compartments, which requires diffusion across cell membranes,
and the transport of gases in the blood. Figure 18.1 presents
an overview of the topics that we cover in this chapter.
If the diffusion of gases between alveoli and blood is significantly impaired, or if oxygen transport in the blood is inadequate, hypoxia (a state of too little oxygen) results. Hypoxia
frequently (but not always!) goes hand in hand with hypercapnia,
RUNNING PROBLEM
High Altitude
In 1981 a group of 20 physiologists, physicians, and
climbers, supported by 42 Sherpa assistants, formed the
American Medical Research Expedition to Mt. Everest. The
purpose of the expedition was to study human physiology
at extreme altitudes, starting with the base camp at 5400 m
(18,000 ft) and continuing on to the summit at 8850 m (over
29,000 ft). From the work of these scientists and others, we
now have a good picture of the physiology of high-altitude
acclimatization.
elevated concentrations of carbon dioxide. These two conditions are clinical signs, not diseases, and clinicians must gather
additional information to pinpoint their cause. Table 18.1
lists several types of hypoxia and some typical causes.
To avoid hypoxia and hypercapnia, the body uses sensors
that monitor arterial blood composition. These sensors respond
to three regulated variables:
1
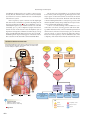
PULMONARY GAS EXCHANGE AND TRANSPORT
CO2
2
O2
Airways
Alveoli of lungs
3
CO2 O2
6 CO2 enters alveoli
at alveolar-capillary
interface.
1 Oxygen enters the
blood at alveolarcapillary interface.
O2
CO2
Pulmonary
circulation
5 CO2 is transported dissolved,
bound to
hemoglobin, or
as HCO3–.
Systemic
circulation
CO2
2 Oxygen is transported in blood
dissolved in plasma
or bound to
hemoglobin inside
RBCs.
3 Oxygen diffuses
into cells.
Cells
ATP
Fig. 18.1
634
CO2
Cellular
respiration
determines
metabolic CO2
production.
The normal values for these three parameters are given in
Table 18.2. In this chapter we will consider the mechanisms
by which oxygen and CO2 move from the lungs to the cells and
back again.
Gas Exchange in the Lungs
and Tissues
O2
4 CO2 diffuses
out of cells.
Oxygen. Arterial oxygen delivery to the cells must be adequate to support aerobic respiration and ATP production.
Carbon dioxide (CO2) is produced as a waste product during the citric acid cycle. Excretion of CO2 by the lungs is
important for two reasons: high levels of CO2 are a central nervous system depressant, and elevated CO2 causes a
state of acidosis (low pH) through the following reaction:
CO2 + H2O Δ H2CO3 Δ H+ + HCO3-.
pH. Maintaining pH homeostasis is critical to prevent denaturation of proteins. The respiratory system monitors
plasma pH and uses changes in ventilation to alter pH.
This process is discussed later along with renal contributions to pH homeostasis.
O2
Nutrients
Breathing is the bulk flow of air into and out of the lungs. Once
air reaches the alveoli, individual gases such as oxygen and CO2
diffuse from the alveolar air space into the blood. Recall that diffusion is movement of a molecule from a region of higher concentration to one of lower concentration.
Gas Exchange and Transport
Table
18.1
Classification of Hypoxias
Type
Definition
Typical Causes
Hypoxic hypoxia
Low arterial PO2
High altitude; alveolar hypoventilation;
decreased lung diffusion capacity;
abnormal ventilation-perfusion ratio
Anemic hypoxia
Decreased total amount of O2 bound to
hemoglobin
Blood loss; anemia (low [Hb] or altered
HbO2 binding); carbon monoxide
poisoning
Ischemic hypoxia
Reduced blood flow
Heart failure (whole-body hypoxia);
shock (peripheral hypoxia); thrombosis
(hypoxia in a single organ)
Histotoxic hypoxia
Failure of cells to use O2 because cells
have been poisoned
Cyanide and other metabolic poisons
Normal Blood Values in Pulmonary Medicine
Table
18.2
Arterial
Venous
P O2
95 mm Hg
(85–100)
40 mm Hg
PCO2
40 mm Hg
(35–45)
46 mm Hg
pH
7.4 (7.38–7.42)
7.37
When we think of concentrations of solutions, units such
as moles/liter and milliosmoles/liter come to mind. However,
respiratory physiologists commonly express plasma gas concentrations in partial pressures to establish whether there is a
concentration gradient between the alveoli and the blood. Gases
move from regions of higher partial pressure to regions of lower
partial pressure.
Figure 18.2 shows the partial pressures of oxygen and
CO2 in air, the alveoli, and inside the body. Normal alveolar PO2
at sea level is about 100 mm Hg. The PO2 of “deoxygenated” venous blood arriving at the lungs is 40 mm Hg. Oxygen therefore
diffuses down its partial pressure (concentration) gradient from
the alveoli into the capillaries. Diffusion goes to equilibrium,
and the PO2 of arterial blood leaving the lungs is the same as in
the alveoli: 100 mm Hg.
When arterial blood reaches tissue capillaries, the gradient
is reversed. Cells are continuously using oxygen for oxidative phosphorylation. In the cells of a person at rest, intracellular PO2 averages 40 mm Hg. Arterial blood arriving at the
cells has a PO2 of 100 mm Hg. Because PO2 is lower in the cells,
oxygen diffuses down its partial pressure gradient from plasma
into cells. Once again, diffusion goes to equilibrium. As a result,
venous blood has the same PO2 as the cells it just passed.
Conversely, PCO2 is higher in tissues than in systemic capillary blood because of CO2 production during metabolism
(Fig. 18.2). Cellular PCO2 in a person at rest is about 46 mm Hg,
compared to an arterial plasma PCO2 of 40 mm Hg. The gradient
causes CO2 to diffuse out of cells into the capillaries. Diffusion
goes to equilibrium, and systemic venous blood averages a PCO2
of 46 mm Hg.
At the pulmonary capillaries, the process reverses.
Venous blood bringing waste CO2 from the cells has a PCO2 of
46 mm Hg. Alveolar PCO2 is 40 mm Hg. Because PCO2 is higher
in the plasma, CO2 moves from the capillaries into the alveoli.
By the time blood leaves the alveoli, it has a PCO2 of 40 mm Hg,
identical to the PCO2 of the alveoli.
In the sections that follow we will consider some of the
other factors that affect the transfer of gases between the alveoli
and the body’s cells.
Concept Check
18
Answers: End of Chapter
1. Cellular metabolism review: which of the following three metabolic
pathways—glycolysis, the citric acid cycle, and the electron transport
system—is directly associated with (a) O2 consumption and with
(b) CO2 production?
2. Why doesn’t the movement of oxygen from the alveoli to the plasma
decrease the PO2 of the alveoli?
3. If nitrogen is 78% of atmospheric air, what is the partial pressure of this
gas when the dry atmospheric pressure is 720 mm Hg?
635
Gas Exchange and Transport
GASES DIFFUSE DOWN CONCENTRATION GRADIENTS
Hypoxia is the primary problem that people experience
when ascending to high altitude. High altitude is considered
anything above 1500 m (5000 ft), but most pathological
responses to altitude occur above 2500 m (about 8000
ft). By one estimate, 25% of people arriving at 2590 m will
experience some form of altitude sickness.
Dry air = 760 mm Hg
PO = 160 mm Hg
2
PCO = 0.25 mm Hg
2
Alveoli
Q1: If water vapor contributes 47 mm Hg to the pressure
of fully humidified air, what is the PO2 of inspired air
reaching the alveoli at 2500 m, where dry atmospheric
pressure is 542 mm Hg? How does this value for PO2
compare with that of fully humidified air at sea level?
PO = 100 mm Hg
2
PCO2 = 40 mm Hg
O2
CO2
Pulmonary
circulation
Venous blood
Arterial blood
PO2 ≤ 40 mm Hg
PCO2 ≥ 46 mm Hg
PO2 = 100 mm Hg
PCO2 = 40 mm Hg
Systemic
circulation
O2
CO2
Cells
PO ≤ 40 mm Hg
2
PCO2 ≥ 46 mm Hg
Aerobic metabolism consumes
O2 and produces CO2.
Fig. 18.2
Lower Alveolar PO2 Decreases Oxygen Uptake
Many variables influence the efficiency of alveolar gas exchange and determine whether arterial blood gases are normal
( Fig. 18.3a). First, adequate oxygen must reach the alveoli.
A decrease in alveolar PO2 means that less oxygen is available
to enter the blood. There can also be problems with the transfer
of gases between the alveoli and pulmonary capillaries. Finally,
blood flow, or perfusion, of the alveoli must be adequate. If
something impairs blood flow to the lung, then the body is unable to acquire the oxygen it needs. Let’s look in more detail at
these factors.
There are two possible causes of low alveolar PO2: either
(1) the inspired air has low oxygen content or (2) alveolar ventilation, is inadequate.
636
RUNNING PROBLEM
Composition of the Inspired Air The first requirement for adequate oxygen delivery to the tissues is adequate oxygen intake
from the atmosphere. The main factor that affects atmospheric
oxygen content is altitude. The partial pressure of oxygen in air
decreases along with total atmospheric pressure as you move from
sea level (where normal atmospheric pressure is 760 mm Hg) to
higher altitudes.
For example, Denver, 1609 m above sea level, has an atmospheric pressure of about 628 mm Hg. The PO2 of dry air in
Denver is 132 mm Hg, down from 160 mm Hg at sea level. For
fully humidified atmospheric air reaching the alveoli, the PO2 is
even lower: Patm 628 mm Hg - PH2O 47 mm Hg) = 581 mm Hg *
21% = PO2 of 122 mm Hg, down from 150 mm Hg at sea level.
Notice that water vapor pressure is the same no matter what the
altitude, making its contribution to total pressure in the lungs
more important as you go higher.
Alveolar Ventilation Unless a person is traveling, altitude remains constant. If the composition of inspired air is normal but
alveolar PO2 is low, then the problem must lie with alveolar ventilation. Low alveolar ventilation is also known as hypoventilation and is characterized by lower-than-normal volumes of fresh
air entering the alveoli. Pathological changes that can result in
alveolar hypoventilation (Fig. 18.3c) include decreased lung
compliance, increased airway resistance, or CNS depression that
slows ventilation rate and decreases depth. Common causes of
CNS depression in young people include alcohol poisoning and
drug overdoses.
Concept Check
Answers: End of Chapter
4. At the summit of Mt. Everest, an altitude of 8850 m, atmospheric pressure
is only 250 mm Hg. What is the PO2 of dry atmospheric air atop Everest? If
water vapor added to inhaled air at the summit has a partial pressure of
47 mm Hg, what is the PO2 of the inhaled air when it reaches the alveoli?
Gas Exchange and Transport
GAS EXCHANGE IN THE ALVEOLI
(a) Alveolar gas exchange
Alveolar Gas Exchange
is influenced by
O2 reaching
the aveoli
Alveolar
ventilation
Composition of
inspired air
Rate and
depth of
breathing
Airway
resistance
Lung
compliance
Gas diffusion
between alveoli
and blood
Surface
area
Adequate
perfusion
of alveoli
Diffusion
distance
Barrier
thickness
Amount of
fluid
(b) Cells form a diffusion barrier between lung and blood.
18
Surfactant
Alveoli
Alveolar
air space
CO2
O2
Alveolar
epithelium
Fused basement
membranes
Capillary
0.1–1.5 μm
Nucleus of
endothelial
cell
O2
CO2
Capillary
lumen
Plasma
RBC
(c) Pathologies that cause hypoxia
Diffusion ∝ surface area × barrier permeability/distance2
Normal lung
Emphysema
Fibrotic lung disease
Pulmonary edema
Asthma
Destruction of alveoli
means less surface
area for gas exchange.
Thickened alveolar membrane
slows gas exchange. Loss
of lung compliance may
decrease alveolar ventilation.
Fluid in interstitial space increases
diffusion distance. Arterial PCO2
may be normal due to higher
CO2 solubility in water.
Increased airway resistance
decreases alveolar
ventilation.
Bronchioles
constricted
PO2
normal
PO normal
2
PO
2
normal
or low
PO2 low
PO2
normal
or low
PO2
normal
Exchange
surface
normal
Increased
diffusion
distance
PO2 low
PO2
low
PO2 low
PO2 low
Fig. 18.3
637
Gas Exchange and Transport
Diffusion Problems Cause Hypoxia
If hypoxia is not caused by hypoventilation, then the problem
usually lies with some aspect of gas exchange between alveoli
and blood. In these situations, alveolar PO2 may be normal, but
the PO2 of arterial blood leaving the lungs is low. The transfer of
oxygen from alveoli to blood requires diffusion across the barrier created by type I alveolar cells and the capillary endothelium (Fig. 18.3b).
The exchange of oxygen and carbon dioxide across this diffusion barrier obeys the same rules as simple diffusion across
a membrane. The diffusion rate is directly proportional to the
available surface area, the concentration gradient of the gas, and
the permeability of the barrier:
Diffusion rate r
surface area : concentration gradient : barrier permeability
From the general rules for diffusion, we can add a fourth factor: diffusion distance. Diffusion is inversely proportional to the
square of the distance or, in simpler terms—diffusion is most
rapid over short distances
B I O T E C H N O LO G Y
The Pulse Oximeter
One important clinical indicator of the effectiveness of
gas exchange in the lungs is the concentration of oxygen in
arterial blood. Obtaining an arterial blood sample is difficult
for the clinician and painful for the patient because it means
finding an accessible artery. (Most blood is drawn from
superficial veins rather than from arteries, which lie deeper
within the body). Over the years, however, scientists have
developed instruments that quickly and painlessly measure
blood oxygen levels through the surface of the skin on a
finger or earlobe. One such instrument, the pulse oximeter,
clips onto the skin and in seconds gives a digital reading of
arterial hemoglobin saturation. The oximeter works by measuring light absorbance of the tissue at two wavelengths.
Another instrument, the transcutaneous oxygen sensor,
measures dissolved oxygen using a variant of traditional gasmeasuring electrodes. Both methods have limitations but are
popular because they provide a rapid, noninvasive means of
estimating arterial oxygen content.
Diffusion rate r 1>distance2
Under most circumstances, diffusion distance, surface
area, and barrier permeability in the body are constants and are
maximized to facilitate diffusion. Gas exchange in the lungs is
rapid, blood flow through pulmonary capillaries is slow, and
diffusion reaches equilibrium in less than 1 second. This leaves
the concentration gradient between alveoli and blood as the primary factor affecting gas exchange in healthy people.
The factors of surface area, diffusion distance, and membrane permeability do come into play with various diseases.
Pathological changes that adversely affect gas exchange include
(1) a decrease in the amount of alveolar surface area available
for gas exchange, (2) an increase in the thickness of the alveolarcapillary exchange barrier, and (3) an increase in the diffusion
distance between the alveolar air space and the blood.
Surface Area Physical loss of alveolar surface area can have
devastating effects in emphysema, a degenerative lung disease
most often caused by cigarette smoking (Fig. 18.3c). The irritating effect of smoke chemicals and tar in the alveoli activates
alveolar macrophages that release elastase and other proteolytic enzymes. These enzymes destroy the elastic fibers of the
lung and induce apoptosis of cells, breaking down the walls of
the alveoli. The result is a high-compliance/low-elastic recoil
lung with fewer and larger alveoli and less surface area for gas
exchange.
Diffusion Barrier Permeability Pathological changes in the
alveolar-capillary diffusion barrier may alter its properties and
slow gas exchange. For example, in fibrotic lung diseases, scar
638
tissue thickens the alveolar wall (Fig. 18.3c). Diffusion of gases
through this scar tissue is much slower than normal. However,
because the lungs have a built-in reserve capacity, one-third of
the exchange epithelium must be incapacitated before arterial
PO2 falls significantly.
Diffusion Distance Normally the pulmonary diffusion distance is small because the alveolar and endothelial cells are thin
and there is little or no interstitial fluid between the two cell layers (Fig. 18.3b). However, in certain pathological states, excess
fluid increases the diffusion distance between the alveolar air
space and the blood. Fluid accumulation may occur inside the
alveoli or in the interstitial compartment between the alveolar
epithelium and the capillary.
In pulmonary edema, accumulation of interstitial fluid increases the diffusion distance and slows gas exchange (Fig. 18.3c).
Gas Exchange and Transport
Normally, only small amounts of interstitial fluid are present in
the lungs, the result of low pulmonary blood pressure and effective lymph drainage. However, if pulmonary blood pressure
rises for some reason, such as left ventricular failure or mitral
valve dysfunction, the normal filtration/reabsorption balance at
the capillary is disrupted.
When capillary hydrostatic pressure increases, more fluid
filters out of the capillary. If filtration increases too much, the lymphatics are unable to remove all the fluid, and excess accumulates
in the pulmonary interstitial space, creating pulmonary edema. In
severe cases, if edema exceeds the tissue’s ability to retain it, fluid
leaks from the interstitial space into the alveolar air space, flooding the alveoli. Normally the inside of the alveoli is a moist surface
lined by a very thin (about 2–5 mm) layer of fluid with surfactant
(see Fig. 18.3b). With alveolar flooding, this fluid layer can become much thicker and seriously impair gas exchange. Alveolar
flooding can also occur with leakage when alveolar epithelium is
damaged, such as from inflammation or inhaling toxic gases. If
hypoxia due to alveolar fluid accumulation is severe and cannot
be corrected by oxygen therapy, the condition may be called adult
respiratory distress syndrome or ARDS.
Concept Check
Answers: End of Chapter
5. Why would left ventricular failure or mitral valve dysfunction cause
elevated pulmonary blood pressure?
6. If alveolar ventilation increases, what happens to arterial PO2? To arterial
PCO2? To venous PO2 and PCO2? Explain your answers.
Gas Solubility Affects Diffusion
A final factor that can affect gas exchange in the alveoli is the
solubility of the gas. The movement of gas molecules from air
into a liquid is directly proportional to three factors: (1) the
pressure gradient of the gas, (2) the solubility of the gas in the
liquid, and (3) temperature. Because temperature is relatively
constant in mammals, we can ignore its contribution in this
discussion.
When a gas is placed in contact with water and there is a
pressure gradient, gas molecules move from one phase to the
other. If gas pressure is higher in the water than in the gaseous
phase, then gas molecules leave the water. If gas pressure is
higher in the gaseous phase than in water, then the gas dissolves
into the water.
For example, consider a container of water exposed to air
with a PO2 of 100 mm Hg ( Fig. 18.4a). Initially, the water has
no oxygen dissolved in it (water PO2 = 0 mm Hg). As the air stays
in contact with the water, some of the moving oxygen molecules
in the air diffuse into the water and dissolve (Fig. 18.4b). This
process continues until equilibrium is reached. At equilibrium
(Fig. 18.4c), the movement of oxygen from the air into the water
RUNNING PROBLEM
Acute mountain sickness is the mildest illness caused by
altitude hypoxia. The primary symptom is a headache
that may be accompanied by dizziness, nausea, fatigue, or
confusion. More severe illnesses are high-altitude pulmonary
edema (HAPE) and high-altitude cerebral edema. HAPE is the
major cause of death from altitude sickness. It is characterized
by high pulmonary arterial pressure, extreme shortness of
breath, and sometimes a productive cough yielding a pink,
frothy fluid. Treatment is immediate relocation to lower
altitude and administration of oxygen.
Q2: Why would someone with HAPE be short of breath?
Q3: Based on what you learned about the mechanisms for
matching ventilation and perfusion in the lung, can
you explain why patients with HAPE have elevated
pulmonary arterial blood pressure?
is equal to the movement of oxygen from the water back into
the air.
We refer to the concentration of oxygen dissolved in the
water at any given PO2 as the partial pressure of the gas in solution.
In our example, therefore, if the air has a PO2 of 100 mm Hg, at
equilibrium the water also has a PO2 of 100 mm Hg.
Note that this does not mean that the concentration of
oxygen is the same in the air and in the water! The concentration of dissolved oxygen also depends on the solubility of oxygen
in water. The ease with which a gas dissolves in a liquid is the
solubility of the gas in that liquid. If a gas is very soluble, large
numbers of gas molecules go into solution at a low gas partial
pressure. With less soluble gases, even a high partial pressure may
cause only a few molecules of the gas to dissolve in the liquid.
For example, when PO2 is 100 mm Hg in both the air and
the water, air contains 5.2 mmol O2>L air, but water contains only
0.15 mmol O2>L water (Fig. 18.4c). As you can see, oxygen is not
very soluble in water and, by extension, in any aqueous solution.
Its low solubility was a driving force for the evolution of oxygencarrying molecules in the aqueous solution we call blood.
Now compare oxygen solubility with CO2 solubility
(Fig. 18.4d). Carbon dioxide is 20 times more soluble in water
than oxygen is. At a PCO2 of 100 mm Hg, the CO2 concentration
in air is 5.2 mmol CO2>L air, and its concentration in water is 3.0
mmol>L water. So although PO2 and PCO2 are both 100 mm Hg
in the water, the amount of each gas that dissolves in the water is
very different.
Why is solubility important in physiology? The answer is
that oxygen’s low solubility in aqueous solutions means that very
little oxygen can be carried dissolved in plasma. Its low solubility
18
639
Gas Exchange and Transport
GASES IN SOLUTION
When temperature remains constant, the amount of a gas that dissolves in a liquid depends on both the
solubility of the gas in the liquid and the partial pressure of the gas.
Oxygen solubility
(a) Initial state: no O2 in solution
(b) Oxygen dissolves.
(c) At equilibrium, PO2 in air and water are equal. Low O2
solubility means concentrations are not equal.
PO2 = 100 mm Hg
[O2] = 5.20 mmol/L
PO2 = 100 mm Hg
PO2 = 100 mm Hg
[O2] = 0.15 mmol/L
PO = 0 mm Hg
2
CO2 solubility
(d) When CO2 is at equilibrium at the same partial pressure
(100 mm Hg), more CO2 dissolves.
PCO2 = 100 mm Hg
[CO2] = 5.20 mmol/L
PCO2 = 100 mm Hg
[CO2] = 3.00 mmol/L
FIGURE QUESTION
Physiologists also express dissolved gases in
blood using the following equation:
[Gas]diss = α [Pgas]
α for oxygen is (0.03 mL O2/L blood)/mm Hg PO2
α for CO2 is (0.7 mL CO2/L blood)/mm Hg PCO2
If arterial blood has a PO2 of 95 mm Hg and a
PCO2 of 40 mm Hg, what are the oxygen and
CO2 concentrations (in mL gas/L blood)?
Fig. 18.4
also means oxygen is slower to cross the increased diffusion distance present in pulmonary edema. Diffusion of oxygen into
alveolar capillaries does not have time to come to equilibrium
before the blood has left the capillaries. The result is decreased
arterial PO2 even though alveolar PO2 may be normal.
Carbon dioxide, in contrast, is relatively soluble in body
fluids, so increased diffusion distance may not significantly affect CO2 exchange. In some cases of pulmonary edema, arterial
PO2 is low but arterial PCO2 is normal because of the different
solubilities of the two gases.
Concept Check
Answers: End of Chapter
7. True or false? Plasma with a PO2 of 40 mm Hg and a PCO2 of 40 mm Hg
has the same concentrations of oxygen and carbon dioxide.
8. A saline solution is exposed to a mixture of nitrogen gas and hydrogen
gas in which PH2 = PN2. What information do you need to predict
whether equal amounts of H2 and N2 dissolve in the solution?
640
Gas Transport in the Blood
Now that we have described how gases enter and leave the
capillaries, we turn our attention to oxygen and carbon dioxide transport in the blood. Gases that enter the capillaries
first dissolve in the plasma. But dissolved gases play only a
small part in providing the cells with oxygen. The red blood
cells, or erythrocytes, have a critical role in ensuring that gas
transport between lung and cells is adequate to meet cell
needs. Without hemoglobin in the red blood cells, the blood
would be unable to transport sufficient oxygen to sustain life
( Fig. 18.5).
Oxygen transport in the circulation and oxygen consumption by tissues are excellent ways to illustrate the general principles of mass flow and mass balance. Mass flow
is defined as amount of x moving per minute, where mass
flow = concentration * volume flow. We can calculate the
mass flow of oxygen traveling from lungs to the cells by using the oxygen content of the arterial blood * cardiac output.
Gas Exchange and Transport
OXYGEN TRANSPORT
MASS BALANCE AND THE FICK EQUATION
More than 98% of the oxygen in blood is bound to hemoglobin in
red blood cells, and less than 2% is dissolved in plasma.
Venous O2
transport
(mL O2/min)
ARTERIAL BLOOD
O2 dissolved in plasma (~ PO2) < 2%
Red blood cell
O2 + Hb
HbO2
> 98%
O2
Arterial O2
transport
(mL O2/min)
Cellular oxygen
consumption (QO2)
(mL O2/min)
Alveolus
Alveolar
membrane
Capillary
endothelium
Transport
to cells
Mass Balance
Cells
Arterial O2 transport – QO2 = Venous O2 transport
HbO2
Hb + O2
Rearranges to:
O2 dissolved
in plasma
O2
18
Arterial O2 transport – Venous O2 transport = QO
2
Used in
cellular
respiration
Mass Flow
O2 transport = Cardiac output (CO)
(L blood/min)
× O2 concentration
(mL O2/L blood)
FIGURE QUESTION
How many cell membranes will O2 cross in its
passage between the airspace of the alveolus
and binding to hemoglobin?
Fig. 18.5
Fick Equation
Substitute the mass flow equation for O2
transport in the mass balance equation:
If arterial blood contains, on average, 200 mL O2 >L blood and
the cardiac output is 5 L>min:
mL O2 >min to cells = 200 mL O2 >L blood * 5 L blood>min
= 1000 mL O2 >min delivered to tissues
If we know the mass flow of oxygen in the venous blood
leaving the cells, we can use the principle of mass balance
to calculate the uptake and consumption of oxygen by the cells
( Fig. 18.6):
Arterial O2 transport - cell use of O2 = venous O2 transport
where oxygen transport is mass flow, mL O2 being transported
per minute. This equation rearranges to:
Arterial O2 transport - venous O2 transport = cell use of O2
(CO × Arterial [O2] ) – (CO × Venous [O2] ) = QO2
Using algebra (AB) – (AC) = A(B – C):
CO × ( Arterial [O2] – Venous [O2] ) = QO2
Fig. 18.6
Adolph Fick, the nineteenth-century physiologist who derived Fick’s law of diffusion, combined the mass flow and mass
balance equations above to relate oxygen consumption (QO2),
cardiac output (CO), and blood oxygen content. The result is the
Fick equation:
QO2 = CO * (arterial oxygen content - venous oxygen content)
The Fick equation can be used to estimate cardiac output or
oxygen consumption, assuming that arterial and venous blood
gases can be measured.
641
Gas Exchange and Transport
Hemoglobin Binds to Oxygen
Oxygen transport in the blood has two components: the oxygen
that is dissolved in the plasma (the PO2) and oxygen bound to
hemoglobin (Hb). In other words:
Total blood O2 content = dissolved O2 + O2 bound to Hb
As you learned in the previous section, oxygen is only slightly
soluble in aqueous solutions, and less than 2% of all oxygen in
the blood is dissolved. That means hemoglobin transports more
than 98% of our oxygen (Fig. 18.5).
Hemoglobin, the oxygen-binding protein that gives red
blood cells their color, binds reversibly to oxygen, as summarized in the equation
Once arterial blood reaches the tissues, the exchange process that took place in the lungs reverses. Dissolved oxygen
diffuses out of systemic capillaries into cells, and the resultant
decrease in plasma PO2 disturbs the equilibrium of the oxygenhemoglobin binding reaction by removing oxygen from the
left side of the equation. The equilibrium shifts to the left according to the law of mass action, and the hemoglobin molecules release their oxygen stores, as represented in the bottom
half of Figure 18.5.
Like oxygen loading at the lungs, this process of transferring oxygen to the body’s cells takes place very rapidly and
goes to equilibrium. The PO2 of the cells determines how much
oxygen is unloaded from hemoglobin. As cells increase their
metabolic activity, their PO2 decreases, and hemoglobin releases
more oxygen to them.
Hb + O2 m HbO2
Why is hemoglobin an effective oxygen carrier? The answer lies in its molecular structure. Hemoglobin (Hb) is a tetramer with four globular protein chains (globins), each centered
around an iron-containing heme group. The central iron atom
of each heme group can bind reversibly with one oxygen molecule. With four heme groups per hemoglobin molecule, one
hemoglobin molecule has the potential to bind four oxygen
molecules. The iron-oxygen interaction is a weak bond that can
be easily broken without altering either the hemoglobin or the
oxygen.
Hemoglobin bound to oxygen is known as oxyhemoglobin, abbreviated HbO2. It would be more accurate to show
the number of oxygen molecules carried on each hemoglobin
molecule—Hb(O2)1 - 4—but we use the simpler abbreviation because the number of bound oxygen molecules varies from one
hemoglobin molecule to another.
Oxygen Binding Obeys the Law of Mass Action
The hemoglobin binding reaction Hb + O2 m HbO2 obeys
the law of mass action. As the concentration of free O2
increases, more oxygen binds to hemoglobin and the equation
shifts to the right, producing more HbO2. If the concentration
of O2 decreases, the equation shifts to the left. Hemoglobin releases oxygen and the amount of oxyhemoglobin decreases.
In the blood, the free oxygen available to bind to hemoglobin is dissolved oxygen, indicated by the PO2 of plasma
(Fig. 18.5). In the pulmonary capillaries, oxygen from the alveoli dissolves in plasma. Dissolved O2 then diffuses into the red
blood cells, where it can binds to hemoglobin. The hemoglobin
acts like a sponge, soaking up oxygen from the plasma until the
reaction Hb + O2 m HbO2 reaches equilibrium.
The transfer of oxygen from alveolar air to plasma to red
blood cells and onto hemoglobin occurs so rapidly that blood in
the pulmonary capillaries normally picks up as much oxygen as
the PO2 of the plasma and the number of red blood cells permit.
642
Hemoglobin Transports Most Oxygen
to the Tissues
To understand why we must have adequate amounts of hemoglobin in our blood to survive, consider the following example.
Assume that a person’s oxygen consumption at rest is about
250 mL O2 >min and the cardiac output is 5 L blood >min. To
meet the cells’ needs for oxygen, the 5 L of blood >min coming to the tissues would need to contain at least 250 mL O2, or
50 mL O2 >L blood.
The low solubility of oxygen means that only 3 mL of O2
will dissolve in the plasma fraction of 1 liter of arterial blood
( Fig. 18.7a). The dissolved oxygen delivery to the cells is
3 mL O2 >L blood * 5 L blood>min = 15 mL O2 >min
The cells use at least 50 mL O2 >min, so the small amount of oxygen that dissolves in plasma cannot meet the needs of the tissues
at rest.
Now let’s consider the difference in oxygen delivery if hemoglobin is available. At normal hemoglobin levels, red blood
cells carry about 197 mL O2 >L blood (Fig. 18.7b).
Total blood O2 content = dissolved O2 + O2 bound to Hb
= 3 mL O2 >L blood + 197 mL HbO2 >L blood
= 200 mL O2 >L blood
If cardiac output remains 5 L>min, hemoglobin-assisted oxygen
delivery to cells is 1000 mL>min:
200 mL O2 >L blood * 5 L blood>min = 1000 mL O2 >min
This is four times the oxygen consumption needed by the tissues
at rest. The extra serves as a reserve for times when oxygen demand increases, such as with exercise.
Gas Exchange and Transport
HEMOGLOBIN INCREASES OXYGEN TRANSPORT
(a) Oxygen transport in blood without
hemoglobin. Alveolar PO2 = arterial PO2
PO2 = 100 mm Hg
(b) Oxygen transport at normal
PO2 in blood with hemoglobin
PO = 100 mm Hg
2
Alveoli
O2 molecule
Arterial
plasma
PO = 100 mm Hg
2
Oxygen dissolves in plasma.
(c) Oxygen transport at reduced PO
2
in blood with hemoglobin
PO = 28 mm Hg
2
PO2 = 100 mm Hg
Red blood cells with hemoglobin are carrying
98% of their maximum load of oxygen.
PO2 = 28 mmHg
Red blood cells carrying 50% of
their maximum load of oxygen.
18
O2 content of plasma = 3 mL O2/L blood
O2 content of plasma =
O2 content of red
blood cells
O2 content of red
blood cells
=0
Total O2 carrying
capacity
3 mL O2/L blood
Total O2 carrying
capacity
3 mL O2/L blood
= 197 mL O2/L blood
200 mL O2/L blood
O2 content of plasma =
O2 content of red
blood cells
Total O2 carrying
capacity
0.8 mL O2/L blood
= 99.5 mL O2/L blood
100.3 mL O2/L blood
Fig. 18.7
EMERGING CONCEPTS
Blood Substitutes
Physiologists have been attempting to find a substitute
for blood ever since 1878, when an intrepid physician named
T. Gaillard Thomas transfused a patient with whole milk
in place of blood. (It helped but the patient died anyway.)
Although milk seems an unlikely replacement for blood, it
has two important properties: proteins to provide colloid
osmotic pressure and molecules (emulsified lipids) capable
of binding to oxygen. In the development of hemoglobin
substitutes, oxygen transport is the most difficult property
to mimic. A hemoglobin solution would seem to be the obvious answer, but hemoglobin that is not compartmentalized
in red blood cells behaves differently than hemoglobin that
is compartmentalized. Investigators are making progress by
polymerizing hemoglobin into larger, more stable molecules
and loading these hemoglobin polymers into phospholipid
liposomes. Perfluorocarbon emulsions are also being tested
as oxygen carriers. To learn more about this research, read
“Physiological properties of blood substitutes,” News Physiol
Sci 16(1): 38–41, 2001 Feb (http://nips.physiology.org).
PO2 Determines Oxygen-Hb Binding
The amount of oxygen that binds to hemoglobin depends on
two factors: (1) the PO2 in the plasma surrounding the red blood
cells and (2) the number of potential Hb binding sites available
in the red blood cells ( Fig. 18.8). Plasma PO2 is the primary
factor determining what percentage of the available hemoglobin binding sites are occupied by oxygen, known as the percent
saturation of hemoglobin. As you learned in previous sections,
arterial PO2 is established by (1) the composition of inspired air,
(2) the alveolar ventilation rate, and (3) the efficiency of gas exchange from alveoli to blood. Figure 18.7c shows what happens
to O2 transport when PO2 decreases.
The total number of oxygen-binding sites depends on the
number of hemoglobin molecules in red blood cells. Clinically, this number can be estimated either by counting the red
blood cells and quantifying the amount of hemoglobin per
red blood cell (mean corpuscular hemoglobin) or by determining the blood hemoglobin content (g Hb>dL whole blood). Any
pathological condition that decreases the amount of hemoglobin in the cells or the number of red blood cells adversely affects
the blood’s oxygen-transporting capacity.
People who have lost large amounts of blood need to replace hemoglobin for oxygen transport. A blood transfusion is
643
Gas Exchange and Transport
The amount of oxygen
bound to Hb depends on
RUNNING PROBLEM
Plasma O2
The amount of
hemoglobin
which determines
which determines
% Saturation
of Hb
×
Total number of
Hb binding sites
calculated from
Hb content
per RBC
×
Number
of RBCs
In most people arriving at high altitude, normal physiological
responses kick in to help acclimatize the body to the chronic
hypoxia. Within two hours of arrival, hypoxia triggers the
release of erythropoietin from the kidneys and liver. This
hormone stimulates red blood cell production, and as a
result, new erythrocytes appear in the blood within days.
Q4: How does adding erythrocytes to the blood help a
person acclimatize to high altitude?
Q5: What does adding erythrocytes to the blood do to the
viscosity of the blood? What effect will that change in
viscosity have on blood flow?
Fig. 18.8
the ideal replacement for blood loss, but in emergencies this is
not always possible. Saline infusions can replace lost blood volume, but saline (like plasma) cannot transport sufficient quantities of oxygen to support cellular respiration. Faced with this
problem, researchers are currently testing artificial oxygen carriers to replace hemoglobin. In times of large-scale disasters,
these hemoglobin substitutes would eliminate the need to identify a patient’s blood type before giving transfusions.
Oxygen Binding Is Expressed As a Percentage
As you just learned, the amount of oxygen bound to hemoglobin at any given PO2 is expressed as the percent saturation of
hemoglobin, where
(Amount of O2 bound>maximum that could be bound) * 100
= percent saturation of hemoglobin
If all binding sites on all hemoglobin molecules are occupied by
oxygen molecules, the blood is 100% oxygenated, or saturated
with oxygen. If half the available binding sites are carrying oxygen, the hemoglobin is 50% saturated, and so on.
The relationship between plasma PO2 and percent saturation of hemoglobin can be explained with the following analogy.
The hemoglobin molecules carrying oxygen are like students
moving books from an old library to a new one. Each student
(a hemoglobin molecule) can carry a maximum of four books
(100% saturation). The librarian in charge controls how many
books (O2 molecules) each student will carry, just as plasma PO2
determines the percent saturation of hemoglobin.
The total number of books being carried depends on the
number of available students, just as the amount of oxygen delivered to the tissues depends on the number of available hemoglobin molecules. For example, if there are 100 students, and the
644
librarian gives each of them four books (100% saturation), then
400 books are carried to the new library. If the librarian gives
three books to each student (decreased plasma PO2), then only
300 books go to the new library, even though each student could
carry four. (Students carrying only three of a possible four books
correspond to 75% saturation of hemoglobin.) If the librarian is
handing out four books per student but only 50 students show
up (fewer hemoglobin molecules), then only 200 books get to
the new library, even though the students are taking the maximum number of books they can carry.
The physical relationship between PO2 and how much oxygen binds to hemoglobin can be studied in vitro. Researchers
expose samples of hemoglobin to various PO2 levels and quantitatively determine the amount of oxygen that binds. Oxyhemoglobin saturation curves , such as the ones shown in
Figure 18.9, are the result of these in vitro binding studies.
(These curves are also called dissociation curves.)
The shape of the Hb # O2 saturation curve reflects the properties of the hemoglobin molecule and its affinity for oxygen.
If you look at the curve, you find that at normal alveolar and
arterial PO2 (100 mm Hg), 98% of the hemoglobin is bound to
oxygen (Fig. 18.9a). In other words, as blood passes through the
lungs under normal conditions, hemoglobin picks up nearly the
maximum amount of oxygen that it can carry.
Notice that the curve is nearly flat at PO2 levels higher than
100 mm Hg (that is, the slope approaches zero). At PO2 above
100 mm Hg, even large changes in PO2 cause only minor changes
in percent saturation. In fact, hemoglobin is not 100% saturated
until the PO2 reaches nearly 650 mm Hg, a partial pressure far
higher than anything we encounter in everyday life.
The flattening of the saturation curve at higher PO2 also
means that alveolar PO2 can fall a good bit below 100 mm Hg
Fig. 18.9 E S S E N T I A L S
Oxygen-hemoglobin Binding Curves
Binding properties of adult and fetal hemoglobin
(b) Maternal and fetal hemoglobin have different oxygenbinding properties.
100
100
90
90
Hemoglobin saturation, %
Hemoglobin saturation, %
(a) The oxyhemoglobin saturation curve is determined in vitro
in the laboratory.
80
70
60
50
40
30
20
Fetal
hemoglobin
80
70
60
Maternal hemoglobin
50
40
30
20
10
10
0
20
40
60
80
Resting cell
PO (mm Hg)
2
0
100
Alveoli
20
40
60
80
PO (mm Hg)
2
100
120
Physical factors alter hemoglobin’s affinity for oxygen
(d) Effect of temperature
(e) Effect of PCO
100
100
100
80
7.6
7.4
60
7.2
40
20
0
20
40
60
PO2 (mm Hg)
80
100
2
37° C
43° C
80
60
40
20
0
20
40
60
PO2 (mm Hg)
80
100
Hemoglobin saturation, %
20° C
Hemoglobin saturation, %
Hemoglobin saturation, %
(c) Effect of pH
80
PCO2 = 20 mm Hg
60
PCO2 = 40 mm Hg
PCO2 = 80 mm Hg
40
20
0
20
40
60
PO2 (mm Hg)
80
100
(f) Effect of the metabolic compound 2,3-DPG
GRAPH QUESTIONS
Hemoglobin saturation, %
100
1. For the graph in (a):
(a) When the PO2 is 20 mm Hg, what is the percent O2 saturation of hemoglobin?
(b) At what PO2 is hemoglobin 50% saturated with O2?
80
2. At a PO2 of 20 mm Hg, how much more oxygen is released at an exercising
muscle cell whose pH is 7.2 than at a cell with a pH of 7.4?
No 2,3-DPG
60
Normal 2,3-DPG
Added 2,3-DPG
40
3. What happens to oxygen release when the exercising muscle cell warms up?
4. Blood stored in blood banks loses its normal content of 2,3-DPG. Is this
good or bad? Explain.
20
5. Because of incomplete gas exchange across the thick membranes of the placenta,
hemoglobin in fetal blood leaving the placenta is 80% saturated with oxygen.
What is the PO2 of that placental blood?
0
20
40
60
PO2 (mm Hg)
80
100
6. Blood in the vena cava of the fetus has a PO2 around 10 mm Hg. What is the
percent O2 saturation of maternal hemoglobin at the same PO2?
645
Gas Exchange and Transport
without significantly lowering hemoglobin saturation. As long
as PO2 in the alveoli (and thus in the pulmonary capillaries) stays
above 60 mm Hg, hemoglobin is more than 90% saturated and
maintains near-normal levels of oxygen transport. However,
once PO2 falls below 60 mm Hg, the curve becomes steeper. The
steep slope means that a small decrease in PO2 causes a relatively
large release of oxygen.
For example, if PO2 falls from 100 mm Hg to 60 mm Hg,
the percent saturation of hemoglobin goes from 98% to about
90%, a decrease of 8%. This is equivalent to a saturation change
of 2% for each 10 mm Hg change. If PO2 falls further, from 60 to
40 mm Hg, the percent saturation goes from 90% to 75%, a decrease of 7.5% for each 10 mm Hg. In the 40–20 mm Hg range,
the curve is even steeper. Hemoglobin saturation declines from
75% to 35%, a change of 20% for each 10 mm Hg change.
What is the physiological significance of the shape of the
saturation curve? In blood leaving systemic capillaries with a
PO2 of 40 mm Hg (an average value for venous blood in a person
at rest), hemoglobin is still 75% saturated, which means that at
the cells it released only one-fourth of the oxygen it is capable of
carrying. The oxygen that remains bound serves as a reservoir
that cells can draw on if metabolism increases.
When metabolically active tissues use additional oxygen,
their cellular PO2 decreases, and hemoglobin releases additional
oxygen at the cells. At a PO2 of 20 mm Hg (an average value for
exercising muscle), hemoglobin saturation falls to about 35%.
With this 20 mm Hg decrease in PO2 (40 mm Hg to 20 mm Hg),
hemoglobin releases an additional 40% of the oxygen it is capable of carrying. This is another example of the built-in reserve
capacity of the body.
Several Factors Affect Oxygen-Hb Binding
Any factor that changes the conformation of the hemoglobin
protein may affect its ability to bind oxygen. In humans, physiological changes in plasma pH, PCO2, and temperature all alter
the oxygen-binding affinity of hemoglobin. Changes in binding
affinity are reflected by changes in the shape of the HbO2 saturation curve.
Increased temperature, increased PCO2, or decreased pH
decrease the affinity of hemoglobin for oxygen and shift the
oxygen-hemoglobin saturation curve to the right (Fig. 18.9c–e).
When these factors change in the opposite direction, binding affinity increases, and the curve shifts to the left. Notice that when
the curve shifts in either direction, the changes are much more
pronounced in the steep part of the curve. Physiologically, this
means that oxygen binding at the lungs (in the 90–100 mm Hg
PO2 range) is not greatly affected, but oxygen delivery at the tissues (in the 20–40 mm Hg range) is significantly altered.
Let’s examine one situation, the affinity shift that takes
place when pH decreases from 7.4 (normal) to 7.2 (more acidic).
(The normal range for blood pH is 7.38–7.42, but a pH of 7.2 is
646
compatible with life). Look at the graph in Figure 18.c. At a PO2
of 40 mm Hg (equivalent to a resting cell) and pH of 7.4, hemoglobin is about 75% saturated. At the same PO2, if the pH falls to
7.2, the percent saturation decreases to about 62%. This means
that hemoglobin molecules release 13% more oxygen at pH 7.2
than they do at pH 7.4.
When does the body undergo shifts in blood pH? One situation is with maximal exertion that pushes cells into anaerobic
metabolism. Anaerobic metabolism in exercising muscle fibers
releases H + into the cytoplasm and extracellular fluid. As H +
concentrations increase, pH falls, the affinity of hemoglobin for
oxygen decreases, and the HbO2 saturation curve shifts to the
right. More oxygen is released at the tissues as the blood becomes more acidic (pH decreases). A shift in the hemoglobin
saturation curve that results from a change in pH is called the
Bohr effect.
An additional factor that affects oxygen-hemoglobin
binding is 2,3-diphosphoglycerate (2,3-DPG; also called
2,3-bisphosphoglycerate or 2,3-BPG), a compound made from
an intermediate of the glycolysis pathway. Chronic hypoxia (extended periods of low oxygen) triggers an increase in 2,3-DPG
production in red blood cells. Increased levels of 2,3-DPG lower
the binding affinity of hemoglobin and shift the HbO2 saturation curve to the right (Fig. 18.9f). Ascent to high altitude and
anemia are two situations that increase 2,3-DPG production.
Changes in hemoglobin’s structure also change its oxygenbinding affinity. For example, fetal hemoglobin (HbF) has two
gamma protein chains in place of the two beta chains found in
adult hemoglobin. The presence of gamma chains enhances the
ability of fetal hemoglobin to bind oxygen in the low-oxygen
environment of the placenta. The altered binding affinity is reflected by the different shape of the fetal HbO2 saturation curve
(Fig. 18.9b). At any given placental PO2 , oxygen released by
maternal hemoglobin is picked up by the higher-affinity fetal
hemoglobin for delivery to the developing fetus. Shortly after
birth, fetal hemoglobin is replaced with the adult form as new
red blood cells are made.
Figure 18.10 summarizes all the factors that influence
the total oxygen content of arterial blood.
Concept Check
Answers: End of Chapter
9. Can a person breathing 100% oxygen at sea level achieve 100%
saturation of her hemoglobin?
10. What effect does hyperventilation have on the percent saturation of
arterial hemoglobin?
11. A muscle that is actively contracting may have a cellular PO2 of 25 mm Hg.
What happens to oxygen binding to hemoglobin at this low PO2? What
is the PO2 of the venous blood leaving the active muscle?
Gas Exchange and Transport
ARTERIAL OXYGEN
The total oxygen content of arterial blood
depends on the amount of oxygen dissolved
in plasma and bound to hemoglobin.
TOTAL ARTERIAL
O2 CONTENT
Oxygen dissolved in
plasma (PO2 of plasma)
Oxygen
bound to Hb
helps
determine
is influenced by
Composition of
inspired air
Rate and
depth of
breathing
Alveolar
ventilation
Airway
resistance
Oxygen diffusion
between alveoli
and blood
Lung
compliance
Surface
area
Membrane
thickness
Adequate
perfusion
of alveoli
% Saturation
of Hb
x
Total number of
binding sites
affected by
Diffusion
distance
PCO
2
pH
Temperature
2,3-DPG
Hb content
per RBC
Number
x of RBCs
Amount of
interstitial fluid
18
Fig. 18.10
Carbon Dioxide Is Transported in Three Ways
Gas transport in the blood includes carbon dioxide removal
from the cells as well as oxygen delivery to cells, and hemoglobin also plays an important role in CO2 transport. Carbon dioxide is a by-product of cellular respiration. It is more soluble in body fluids than oxygen is, but the cells produce far
more CO2 than can dissolve in the plasma. Only about 7% of
the CO2 carried by venous blood is dissolved in the blood. The
remaining 93% diffuses into red blood cells, where 70% is converted to bicarbonate ion, as explained below, and 23% binds to
hemoglobin (HbCO2). Figure 18.11 summarizes these three
mechanisms of carbon dioxide transport in the blood.
Why is removing CO2 from the body so important? First,
elevated PCO2 (hypercapnia) causes the pH disturbance known
as acidosis . Extremes of pH interfere with hydrogen bonding of molecules and can denature proteins. Abnormally high
PCO2 levels also depress central nervous system function, causing confusion, coma, or even death. For these reasons, CO2 is
a potentially toxic waste product that must be removed by the
lungs.
CO2 and Bicarbonate Ions As we just noted, about 70% of the
CO2 that enters the blood is transported to the lungs as bicarbonate ions (HCO3- ) dissolved in the plasma. The conversion of
CO2 to HCO3- serves two purposes: (1) it provides an additional
means of CO2 transport from cells to lungs, and (2) HCO3- is
available to act as a buffer for metabolic acids, thereby
helping stabilize the body’s pH.
How does CO2 turn into HCO3- ? The rapid conversion depends on the presence of carbonic anhydrase (CA), an enzyme
found concentrated in red blood cells. Let’s see how this happens. Dissolved CO2 in the plasma diffuses into red blood cells,
where it may react with water in the presence of carbonic anhydrase to form carbonic acid (H2CO3, top portion of Fig. 18.11).
Carbonic acid then dissociates into a hydrogen ion and a bicarbonate ion:
Carbonic
anhydrase
CO2 + H2O m H2CO3 m H + + HCO3Carbonic
acid
Because carbonic acid dissociates readily, we sometimes ignore
the intermediate step and summarize the reaction as:
CO2 + H2O m H + + HCO3This reaction is reversible. The rate in either direction depends
on the relative concentrations of the substrates and obeys the
law of mass action.
The conversion of carbon dioxide to H + and HCO3- continues until equilibrium is reached. (Water is always in excess
in the body, so water concentration plays no role in the dynamic equilibrium of this reaction.) To keep the reaction going, the products (H + and HCO3- ) must be removed from the
cytoplasm of the red blood cell. If the product concentrations
are kept low, the reaction cannot reach equilibrium. Carbon
dioxide continues to move out of plasma into the red blood
cells, which in turn allows more CO2 to diffuse out of tissues
into the blood.
647
Gas Exchange and Transport
CARBON DIOXIDE TRANSPORT
Most CO2 in the blood has been converted
to bicarbonate ion, HCO3–.
1 CO2 diffuses out of cells into systemic
capillaries.
2 Only 7% of the CO2 remains dissolved in plasma.
3 Nearly a fourth of the CO2 binds to
hemoglobin, forming carbaminohemoglobin.
VENOUS BLOOD
1
2
CO2
Cellular
respiration
in
peripheral
tissues
Dissolved CO2
(7%)
Red blood cell
3 CO2 + Hb
4 CO2 + H2O
CA
Cell membrane
5 HCO3 enters the plasma in exchange for
Cl– (the chloride shift).
8 The carbonic acid reaction reverses, pulling
HCO3– back into the RBC and converting it
back to CO2.
H+ + Hb
HbH
HCO3– in
plasma (70%)
Transport
to lungs
–
7 By the law of mass action, CO2 unbinds from
hemoglobin and diffuses out of the RBC.
HCO3–
H2CO3
5
Capillary
endothelium
4 70% of the CO2 load is converted to
bicarbonate and H+. Hemoglobin buffers H+.
6 At the lungs, dissolved CO2 diffuses out of
the plasma.
Cl–
HbCO2 (23%)
6
Dissolved CO2
Dissolved CO2
–
8 HCO3
in
plasma
Hb + CO2
HbCO2
Cl–
HCO3–
HbH
H2CO3
CA
7
CO2
Alveoli
H2O + CO2
H+ + Hb
KEY
CA = carbonic anhydrase
Fig. 18.11
Two separate mechanisms remove free H + and HCO3- .
In the first, bicarbonate leaves the red blood cell on an antiport
protein. This transport process, known as the chloride
shift, exchanges HCO3- for Cl - . The anion exchange maintains the cell’s electrical neutrality. The transfer of HCO3- into
the plasma makes this buffer available to moderate pH changes
caused by the production of metabolic acids. Bicarbonate is the
most important extracellular buffer in the body.
produced from the reaction of CO2 and water. In those cases,
excess H + accumulates in the plasma, causing the condition
known as respiratory acidosis. You will learn more about the
role of the respiratory system in maintaining pH homeostasis
when you study acid-base balance.
Hemoglobin and H+ The second mechanism removes free
H + from the red blood cell cytoplasm. Hemoglobin within the
red blood cell acts as a buffer and binds hydrogen ions in the
reaction
Hemoglobin and CO2 Although most carbon dioxide that enters red blood cells is converted to bicarbonate ions, about 23%
of the CO2 in venous blood binds directly to hemoglobin. At the
cells, when oxygen leaves its binding sites on the hemoglobin
molecule, CO2 binds with free hemoglobin at exposed amino
groups (-NH2), forming carbaminohemoglobin:
H+ + Hb m HbH
CO2 + Hb m HbCO2 (carbaminohemoglobin)
Hemoglobin’s buffering of H + is an important step that prevents
large changes in the body’s pH. If blood PCO2 is elevated much
above normal, the hemoglobin buffer cannot soak up all the H +
The presence of CO2 and H + facilitates formation of carbaminohemoglobin because both these factors decrease hemoglobin’s binding affinity for oxygen (see Fig. 18.9).
648
Gas Exchange and Transport
RUNNING PROBLEM
SUMMARY OF O2 AND CO2 EXCHANGE AND TRANSPORT
The usual homeostatic response to high-altitude hypoxia is
hyperventilation, which begins on arrival. Hyperventilation
enhances alveolar ventilation, but this may not help elevate
arterial PO2 levels significantly when atmospheric PO2 is low.
However, hyperventilation does lower plasma PCO2.
Dry air = 760 mm Hg
PO = 160 mm Hg
2
PCO2 = 0.25 mm Hg
Q6: What happens to plasma pH during hyperventilation?
(Hint: Apply the law of mass action to figure out what
happens to the balance between CO2 and H+ + HCO3-).
Alveoli
PO2 = 100 mm Hg
PCO2 = 40 mm Hg
Q7: How does this change in pH affect oxygen binding at
the lungs when PO2 is decreased? How does it affect
unloading of oxygen at the cells?
CO2 O2
O2 transport
CO2 transport
HCO3– = 70%
HbCO2 = 23%
Dissolved CO2 = 7%
CO2 Removal at the Lungs When venous blood reaches the
lungs, the processes that took place in the systemic capillaries
reverse (bottom portion of Fig. 18.11). The PCO2 of the alveoli
is lower than that of venous blood in the pulmonary capillaries.
Therefore, CO2 diffuses down its pressure gradient—in other
words, out of plasma into the alveoli—and the plasma PCO2 begins to fall.
The decrease in plasma PCO2 allows dissolved CO2 to diffuse out of the red blood cells. As CO2 levels in the red blood
cells decrease, the equilibrium of the CO2 -HCO3- reaction is
disturbed, shifting toward production of more CO2. Removal
of CO2 causes H + to leave the hemoglobin molecules, and the
chloride shift reverses: Cl - returns to the plasma in exchange
for HCO3- moving back into the red blood cells. The HCO3and newly released H + re-form into carbonic acid, which is
then converted into water and CO2. This CO2 is then free to diffuse out of the red blood cell and into the alveoli.
Figure 18.12 shows the combined transport of CO2
and O2 in the blood. At the alveoli, O2 diffuses down its pressure gradient, moving from the alveoli into the plasma and then
from the plasma into the red blood cells. Hemoglobin binds to
O2, increasing the amount of oxygen that can be transported to
the cells.
At the cells, the process reverses. Because PO2 is lower in
cells than in the arterial blood, O2 diffuses from the plasma into
the cells. The decrease in plasma PO2 causes hemoglobin to release O2, making additional oxygen available to enter cells.
Carbon dioxide from aerobic metabolism simultaneously
leaves cells and enters the blood, dissolving in the plasma. From
there, CO2 enters red blood cells, where most is converted to
HCO3- and H + . The HCO3- is returned to the plasma in exchange for a Cl - while the H + binds to hemoglobin. A fraction of the CO2 that enters red blood cells also binds directly to
Pulmonary
circulation
HbO2 > 98%
Dissolved O2 < 2%
(~PO2)
18
Venous blood
Arterial blood
PO2 ≤ 40 mm Hg
PCO2 ≥ 46 mm Hg
PO2 = 100 mm Hg
PCO2 = 40 mm Hg
Systemic
circulation
O2
CO2
Cells
PO2 ≤ 40 mm Hg
PCO2 ≥ 46 mm Hg
Fig. 18.12
hemoglobin. At the lungs, the process reverses as CO2 diffuses
out of the pulmonary capillaries and into the alveoli.
To understand fully how the respiratory system coordinates delivery of oxygen to the lungs with transport of oxygen
in the circulation, we now consider the central nervous system
control of ventilation.
Concept Check
Answers: End of Chapter
12. How would an obstruction of the airways affect alveolar ventilation,
arterial PCO2, and the body’s pH?
Regulation of Ventilation
Breathing is a rhythmic process that usually occurs without conscious thought or awareness. In that respect, it resembles the
rhythmic beating of the heart. However, skeletal muscles, unlike
649
Gas Exchange and Transport
autorhythmic cardiac muscles, are not able to contract spontaneously. Instead, skeletal muscle contraction must be initiated
by somatic motor neurons, which in turn are controlled by the
central nervous system.
In the respiratory system, contraction of the diaphragm
and other muscles is initiated by a spontaneously firing network
of neurons in the brain stem ( Fig. 18.13). Breathing occurs automatically throughout a person’s life but can also be controlled
voluntarily, up to a point. Complicated synaptic interactions
between neurons in the network create the rhythmic cycles of
inspiration and expiration, influenced continuously by sensory
input, especially that from chemoreceptors for CO2, O2, and H + .
Ventilation pattern depends in large part on the levels of those
three substances in the arterial blood and extracellular fluid.
The neural control of breathing is one of the few “black
boxes” left in systems-level physiology. Although we know the
major regions of the brain stem that are involved, the details
remain elusive and controversial. The brain stem network that
controls breathing behaves like a central pattern generator, with
intrinsic rhythmic activity that probably arises from pacemaker
neurons with unstable membrane potentials.
Some of our understanding of how ventilation is controlled has come from observing patients with brain damage. Other information has come from animal experiments in
which neural connections between major parts of the brain
stem are severed, or sections of brain are studied in isolation.
Research on CNS respiratory control is difficult because of the
complexity of the neural network and its anatomical location,
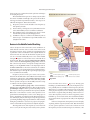
THE REFLEX CONTROL OF VENTILATION
Central and peripheral chemoreceptors monitor blood gases and pH.
Control networks in the brain stem regulate activity in somatic motor
neurons leading to respiratory muscles.
Emotions
and voluntary
control
CO2
O2 and pH
Higher
brain
centers
Medullary
chemoreceptors
Carotid and aortic
chemoreceptors
16
15
1
2
14
13
3
4
Limbic
system
Afferent sensory
neurons
12
5
6
Medulla oblongata
and pons
7
8
11
Somatic
motor neurons
(inspiration)
10
Somatic
motor neurons
(expiration)
9
Inspiration
FIGURE QUESTION
650
Expiration
Scalene and
sternocleidomastoid
muscles
External
intercostals
Diaphragm
Internal
intercostals
Abdominal
muscles
KEY
Match the numbers on the
figure to the boxes of the map.
Stimuli
Integrating centers
Sensors
Efferent neurons
Fig. 18.13
Afferent neurons
Targets
Gas Exchange and Transport
but in recent years scientists have developed better techniques
for studying the system.
The details that follow represent a contemporary model for
the control of ventilation. Although some parts of the model are
well supported with experimental evidence, other aspects are
still under investigation. This model states that:
1
2
3
4
Neural networks in the brain stem control ventilation.
Respiratory neurons in the medulla control inspiratory
and expiratory muscles.
Neurons in the pons integrate sensory information and interact with medullary neurons to influence ventilation.
The rhythmic pattern of breathing arises from a neural
network with spontaneously discharging neurons.
Ventilation is subject to continuous modulation by various
chemoreceptor- and mechanoreceptor-linked reflexes and
by higher brain centers.
Higher
brain
centers
Pons
PRG
NTS
Neurons in the Medulla Control Breathing
Classic descriptions of how the brain controls ventilation divided the brain stem into various control centers. More recent
descriptions, however, are less specific about assigning function
to particular “centers” and instead look at complex interactions
between neurons in a network. We know that respiratory neurons are concentrated bilaterally in two areas of the medulla
oblongata. Figure 18.14 shows these areas on the left side of
the brain stem. One area called the nucleus tractus solitarius
(NTS) contains the dorsal respiratory group (DRG) of neurons
that control mostly muscles of inspiration. Output from the
DRG goes via the phrenic nerves to the diaphragm and via
the intercostal nerves to the intercostal muscles. In addition,
the NTS receives sensory information from peripheral chemoand mechanoreceptors through the vagus and glossopharyngeal
nerves (cranial nerves X and IX).
Respiratory neurons in the pons receive sensory information from the DRG and in turn influence the initiation and
termination of inspiration. The pontine respiratory groups
(previously called the pneumotaxic center) and other pontine
neurons provide tonic input to the medullary networks to help
coordinate a smooth respiratory rhythm.
The ventral respiratory group (VRG) of the medulla has
multiple regions with different functions. One area known as the
pre-Bötzinger complex contains spontaneously firing neurons
that may act as the basic pacemaker for the respiratory rhythm.
Other areas control muscles used for active expiration or for
greater-than-normal inspiration, such as occurs during vigorous
exercise. In addition, nerve fibers from the VRG innervate muscles of the larynx, pharynx, and tongue to keep the upper airways
open during breathing. Inappropriate relaxation of these muscles
during sleep contributes to obstructive sleep apnea, a sleeping disorder associated with snoring and excessive daytime sleepiness.
The integrated action of the respiratory control networks
can be seen by monitoring electrical activity in the phrenic
Medullary chemoreceptors
monitor CO2.
Sensory input
from CN IX, X
(mechanical and
chemosensory)
18
DRG
Medulla
pre-Bötzinger
complex
VRG
Output to expiratory,
some inspiratory,
pharynx, larynx, and
tongue muscles
Output
primarily to
inspiratory
muscles
KEY
PRG = Pontine respiratory group
DRG = Dorsal respiratory group
VRG = Ventral respiratory group
NTS = Nucleus tractus solitarius
Fig. 18.14
nerve and other motor nerves ( Fig. 18.15 ). During quiet
breathing, a pacemaker initiates each cycle, and inspiratory neurons gradually increase stimulation of the inspiratory muscles.
This increase is sometimes called ramping because of the shape
of the graph of inspiratory neuron activity. A few inspiratory
neurons fire to begin the ramp. The firing of these neurons recruits other inspiratory neurons to fire in an apparent positive
feedback loop. As more neurons fire, more skeletal muscle fibers
are recruited. The rib cage expands smoothly as the diaphragm
contracts.
At the end of inspiration, the inspiratory neurons abruptly
stop firing, and the respiratory muscles relax. Over the next few
seconds, passive expiration occurs because of elastic recoil of
the inspiratory muscles and elastic lung tissue. However, some
651
Gas Exchange and Transport
NEURAL ACTIVITY DURING QUIET BREATHING
Tidal volume (liters)
Number of active
inspiratory neurons
During inspiration, the activity of inspiratory neurons increases steadily, apparently
through a positive feedback mechanism. At the end of inspiration, the activity shuts off
abruptly and expiration takes place through recoil of elastic lung tissue.
ive
sit op
po k lo
d
pi bac
Ra ed
fe
Inspiration shuts off
0.5
0
Inspiration
2 sec
Passive expiration
3 sec
Inspiration
2 sec
Time
GRAPH QUESTION
What is the ventilation rate of the person in this example?
Fig. 18.15
motor neuron activity can be observed during passive expiration, suggesting that perhaps muscles in the upper airways contract to slow the flow of air out of the respiratory system.
Many neurons of the VRG remain inactive during quiet
respiration. They function primarily during forced breathing,
when inspiratory movements are exaggerated, and during active
expiration. In forced breathing, increased activity of inspiratory
neurons stimulates accessory muscles, such as the sternocleidomastoids. Contraction of these accessory muscles enhances expansion of the thorax by raising the sternum and upper ribs.
With active expiration, expiratory neurons from the VRG
activate the internal intercostal and abdominal muscles. There
seems to be some communication between inspiratory and expiratory neurons, as inspiratory neurons are inhibited during
active expiration.
Carbon Dioxide, Oxygen, and
pH Influence Ventilation
Sensory input from central and peripheral chemoreceptors
modifies the rhythmicity of the control network to help maintain blood gas homeostasis. Carbon dioxide is the primary
652
stimulus for changes in ventilation. Oxygen and plasma pH play
lesser roles.
The chemoreceptors for oxygen and carbon dioxide are
strategically associated with the arterial circulation. If too little
oxygen is present in arterial blood destined for the brain and
other tissues, the rate and depth of breathing increase. If the rate
of CO2 production by the cells exceeds the rate of CO2 removal
by the lungs, arterial PCO2 increases, and ventilation is intensified to match CO2 removal to production. These homeostatic
reflexes operate constantly, keeping arterial PO2 and PCO2 within
a narrow range.
Peripheral chemoreceptors located in the carotid and
aortic arteries sense changes in the PO2 , pH, and PCO2 of the
plasma (Fig. 18.13). These carotid and aortic bodies are close
to the locations of the baroreceptors involved in reflex control of
blood pressure. Central chemoreceptors in the brain respond to changes in the concentration of CO2 in the cerebrospinal fluid. These central receptors lie on the ventral surface of
the medulla, close to neurons involved in respiratory control.
Peripheral Chemoreceptors When specialized glomus cells
{glomus, a ball-shaped mass} in the carotid and aortic bodies are
activated by a decrease in PO2 or pH or by an increase in PCO2,
Gas Exchange and Transport
they trigger a reflex increase in ventilation. Under most normal
circumstances, oxygen is not an important factor in modulating ventilation because arterial PO2 must fall to less than 60 mm
Hg before ventilation is stimulated. This large decrease in PO2 is
equivalent to ascending to an altitude of 3000 m. (For reference,
Denver is located at an altitude of 1609 m). However, any condition that reduces plasma pH or increases PCO2 will activate the
carotid and aortic glomus cells and increase ventilation.
The details of glomus cell function remain to be worked
out, but the basic mechanism by which these chemoreceptors
respond to low oxygen is similar to the mechanism you learned
for insulin release by pancreatic beta cells or taste transduction
in taste buds.
In all three examples, a stimulus inactivates K + channels,
causing the receptor cell to depolarize ( Fig. 18.16). Depolarization opens voltage-gated Ca2 + channels, and Ca2 + entry
GLOMUS CELLS
The carotid body oxygen sensor releases neurotransmitter
when PO decreases.
2
Blood vessel
Low PO
2
1 Low PO
2 K+ channels close
2
3 Cell
depolarizes
Glomus cell
in carotid
body
5 Ca2+
enters
4 Voltage-gated Ca2+
channel opens
6 Exocytosis of
neurotransmitters
Receptor on
sensory neuron
Action potential
7 Signal to medullary
centers to increase
ventilation
Fig. 18.16
causes exocytosis of neurotransmitter onto the sensory neuron.
In the carotid and aortic bodies, neurotransmitters initiate action potentials in sensory neurons leading to the brain stem respiratory networks, signaling them to increase ventilation.
Because the peripheral chemoreceptors respond only to
dramatic changes in arterial PO2, arterial oxygen concentrations
do not play a role in the everyday regulation of ventilation. However, unusual physiological conditions, such as ascending to high
altitude, and pathological conditions, such as chronic obstructive
pulmonary disease (COPD), can reduce arterial PO2 to levels that
are low enough to activate the peripheral chemoreceptors.
Central Chemoreceptors The most important chemical controller of ventilation is carbon dioxide, mediated both through
the peripheral chemoreceptors just discussed and through central chemoreceptors located in the medulla ( Fig. 18.17). These
receptors set the respiratory pace, providing continuous input
into the control network. When arterial PCO2 increases, CO2
crosses the blood-brain barrier and activates the central chemoreceptors. These receptors signal the control network to increase
the rate and depth of ventilation, thereby enhancing alveolar
ventilation and removing CO2 from the blood.
Although we say that the central chemoreceptors monitor
CO2, they actually respond to pH changes in the cerebrospinal
fluid (CSF). Carbon dioxide that diffuses across the blood-brain
barrier into the CSF is converted to carbonic acid, which dissociates to bicarbonate and H + . Experiments indicate that the H +
produced by this reaction is what initiates the chemoreceptor
reflex, rather than the increased level of CO2.
Note, however, that pH changes in the plasma do not usually influence the central chemoreceptors directly. Although
plasma PCO2 enters the CSF readily, plasma H + crosses the
blood-brain barrier very slowly and therefore has little direct effect on the central chemoreceptors.
When plasma PCO2 increases, the chemoreceptors initially
respond strongly by increasing ventilation. However, if PCO2
remains elevated for several days, ventilation falls back toward
normal rates as the chemoreceptor response adapts by mechanisms that are not clear. Fortunately for people with chronic
lung diseases, the response of peripheral chemoreceptor to low
arterial PO2 remains intact over time, even though the central
chemoreceptor response adapts to high PCO2.
In some situations, low PO2 becomes the primary chemical stimulus for ventilation. For example, patients with severe
chronic lung disease, such as COPD, have chronic hypercapnia and hypoxia. Their arterial PCO2 may rise to 50–55 mm Hg
(normal is 35–45) while their PO2 falls to 45–50 mm Hg (normal 75–100). Because these levels are chronic, the chemoreceptor response adapts to the elevated PCO2. Most of the chemical
stimulus for ventilation in this situation then comes from low
PO2, sensed by the carotid and aortic chemoreceptors. If these
patients are given too much oxygen, they may stop breathing
because their chemical stimulus for ventilation is eliminated.
18
653
Gas Exchange and Transport
CHEMORECEPTOR RESPONSE
Carotid and aortic chemoreceptors monitor CO2, O2, and H+.
Central chemoreceptors monitor CO2 in cerebrospinal fluid.
Plasma
PCO2
KEY
CA = carbonic anhydrase
–
Cerebral capillary
Blood-brain
barrier
2
CO2 + H2O
Cerebrospinal
fluid
CO2
H+
PCO
CA
H2CO3
H+ +
(in plasma)
HCO3–
H+ + HCO3–
Stimulates
peripheral
chemoreceptors
in carotid and
aortic bodies
Plasma PO2
< 60 mm Hg
Central
chemoreceptor
Medulla
oblongata
at brain
Respiratory
control
centers
–
Sensory
neurons
Ventilation
Plasma PO2
Plasma PCO
Negative feedback
2
Fig. 18.17
The central chemoreceptors respond to decreases in arterial PCO2 as well as to increases. If alveolar PCO2 falls, as it might
during hyperventilation, plasma PCO2 and cerebrospinal fluid
PCO2 follow suit. As a result, central chemoreceptor activity declines, and the control network slows the ventilation rate. When
ventilation decreases, carbon dioxide begins to accumulate in
alveoli and the plasma. Eventually, the arterial PCO2 rises above
the threshold level for the chemoreceptors. At that point, the receptors fire, and the control network again increases ventilation.
Protective Reflexes Guard the Lungs
In addition to the chemoreceptor reflexes that help regulate
ventilation, the body has protective reflexes that respond to
physical injury or irritation of the respiratory tract and to overinflation of the lungs. The major protective reflex is bronchoconstriction, mediated through parasympathetic neurons that
innervate bronchiolar smooth muscle. Inhaled particles or noxious gases stimulate irritant receptors in the airway mucosa.
654
The irritant receptors send signals through sensory neurons to
integrating centers in the CNS that trigger bronchoconstriction. Protective reflex responses also include coughing and
sneezing.
The Hering-Breuer inflation reflex was first described in the
late 1800s in anesthetized dogs. In these animals, if tidal volume
exceeded a certain volume, stretch receptors in the lung signaled
the brain stem to terminate inspiration. However, this reflex is
difficult to demonstrate in adult humans and does not operate
during quiet breathing and mild exertion. Studies on human infants, however, suggest that the Hering-Breuer inflation reflex
may play a role in limiting their ventilation volumes.
Higher Brain Centers Affect
Patterns of Ventilation
Conscious and unconscious thought processes also affect respiratory activities. Higher centers in the hypothalamus and cerebrum can alter the activity of the brain stem control network
Gas Exchange and Transport
to change ventilation rate and depth. Voluntary control of
ventilation falls into this category. Higher brain center control
is not a requirement for ventilation, however. Even if the brain
stem above the pons is severely damaged, essentially normal respiratory cycles continue.
Respiration can also be affected by stimulation of portions
of the limbic system. For this reason, emotional and autonomic
activities such as fear and excitement may affect the pace and
depth of respiration. In some of these situations, the neural
pathway goes directly to the somatic motor neurons, bypassing
the control network in the brain stem.
Although we can temporarily alter our respiratory performance, we cannot override the chemoreceptor reflexes. Holding
your breath is a good example. You can hold your breath voluntarily only until elevated PCO2 in the blood and cerebrospinal
fluid activates the chemoreceptor reflex, forcing you to inhale.
Small children having temper tantrums sometimes attempt
to manipulate parents by threatening to hold their breath until
they die. However, the chemoreceptor reflexes make it impossible for the children to carry out that threat. Extremely strongwilled children can continue holding their breath until they turn
blue and pass out from hypoxia, but once they are unconscious,
normal breathing automatically resumes.
RUNNING PROBLEM
The hyperventilation response to hypoxia creates a peculiar
breathing pattern called periodic breathing, in which the
person goes through a 10–15-second period of breathholding followed by a short period of hyperventilation.
Periodic breathing occurs most often during sleep.
Q8: Based on your understanding of how the body controls
ventilation, why do you think periodic breathing occurs
most often during sleep?
Breathing is intimately linked to cardiovascular function.
The integrating centers for both functions are located in the
brain stem, and interneurons project between the two networks,
allowing signaling back and forth. The cardiovascular, respiratory, and renal systems all work together to maintain fluid and
acid-base homeostasis.
18
RUNNING PROBLEM CONCLUSION
High Altitude
On May 29, 1953, Edmund Hillary and Tenzing Norgay of
the British Everest Expedition were the first humans to
reach the summit of Mt. Everest. They carried supplemental
oxygen with them, as it was believed that this feat was
impossible without it. In 1978, however, Reinhold Messner
and Peter Habeler achieved the “impossible.” On May 8,
they struggled to the summit using sheer willpower and no
extra oxygen. In Messner’s words, “I am nothing more than
a single narrow gasping lung, floating over the mists and
summits.” Learn more about these Everest expeditions by
doing a Google search for Hillary Everest or Messner Everest.
To learn more about different types of mountain
sickness, see the International Society for Mountain Medicine
(www.ismmed.org/np_altitude_tutorial.htm); “High altitude
medicine,” Am Fam Physician 1998 Apr. 15 (www.aafp.org/
afp/980415ap/harris.html); and “High-altitude pulmonary
edema” (www.emedicine.com/MED/topic1956.htm).
In this running problem you learned about normal
and abnormal responses to high altitude. Check your
understanding of the physiology behind this respiratory
challenge by comparing your answers with the
information in the following table.
Question
Facts
Integration and Analysis
1. What is the PO2 of inspired air
reaching the alveoli when dry
atmospheric pressure is 542 mm Hg?
How does this value for PO2 compare
with the PO2 value for fully humidified
air at sea level?
Water vapor contributes a partial pressure
of 47 mm Hg to fully humidified air. Oxygen
is 21% of dry air. Normal atmospheric
pressure at sea level is 760 mm Hg.
Correction for water vapor:
542 - 47 = 495 mm Hg * 21% PO2
= 104 mm Hg PO2. In humidified air at sea
level, PO2 = 150 mm Hg.
2. Why would someone with HAPE be
short of breath?
Pulmonary edema increases the diffusion
distance for oxygen.
Slower oxygen diffusion means less oxygen
reaching the blood, which worsens the
normal hypoxia of altitude.
655
Gas Exchange and Transport
R U N N I N G P R O B L E M CO N C LU S I O N (continued)
Question
Facts
Integration and Analysis
3. Based on mechanisms for
matching ventilation and perfusion in
the lung, why do patients with HAPE
have elevated pulmonary arterial
blood pressure?
Low oxygen levels constrict pulmonary
arterioles.
Constriction of pulmonary arterioles causes
blood to collect in the pulmonary arteries
behind the constriction. This increases
pulmonary arterial blood pressure.
4. How does adding erythrocytes to
the blood help a person acclimatize
to high altitude?
98% of arterial oxygen is carried bound to
hemoglobin.
Additional hemoglobin increases the
oxygen-carrying capacity of the blood.
5. What does adding erythrocytes to
the blood do to the viscosity of the
blood? What effect will that change in
viscosity have on blood flow?
Adding cells increases blood viscosity.
According to Poiseuille’s law, increased
viscosity increases resistance to flow, so
blood flow will decrease.
6. What happens to plasma pH during
hyperventilation?
Apply the law of mass action to the
equation CO2 + H2O m H + + HCO3- .
The amount of CO2 in the plasma decreases
during hyperventilation, which means
the equation shifts to the left. This shift
decreases H+, which increases pH (alkalosis).
7. How does this change in pH affect
oxygen binding at the lungs when
PO2 is decreased? How does it affect
unloading of oxygen at the cells?
See Figure 18.9c.
The left shift of the curve means that, at any
given PO2, more O2 binds to hemoglobin.
Less O2 will unbind at the tissues for a given
PO2, but PO2 in the cells is probably lower
than normal, and consequently there may
be no change in unloading.
8. Why do you think periodic
breathing occurs most often during
sleep?
Periodic breathing alternates periods
of breath-holding (apnea) and
hyperventilation.
An awake person is more likely to make
a conscious effort to breathe during the
breath-holding spells, eliminating the cycle
of periodic breathing.
Test your understanding with:
• Practice Tests
• Running Problem Quizzes
• A&PFlixTM Animations
• PhysioExTM Lab Simulations
• Interactive Physiology
Animations
www.masteringaandp.com
Chapter Summary
In this chapter, you learned why climbing Mt. Everest is such a respiratory challenge for the human body, and why people with emphysema
experience the same respiratory challenges at sea level. The exchange
and transport of oxygen and carbon dioxide in the body illustrate the
mass flow of gases along concentration gradients. Homeostasis of these
656
blood gases demonstrates mass balance: the concentration in the blood
varies according to what enters or leaves at the lungs and tissues. The
law of mass action governs the chemical reactions through which hemoglobin binds O2, and carbonic anhydrase catalyzes the conversion of
CO2 and water to carbonic acid.
Gas Exchange and Transport
Gas Exchange in the Lungs and Tissues
Respiratory: Gas Exchange
1. Normal alveolar and arterial PO2 is about 100 mm Hg. Normal alveolar and arterial PCO2 is about 40 mm Hg. Normal venous PO2 is
40 mm Hg, and normal venous PCO2 is 46 mm Hg. (Fig. 18.2)
2. Body sensors monitor blood oxygen, CO2, and pH in an effort to
avoid hypoxia and hypercapnia.
3. Both the composition of inspired air and the effectiveness of alveolar ventilation affect alveolar PO2.
4. Changes in alveolar surface area, in diffusion barrier thickness,
and in fluid distance between the alveoli and pulmonary capillaries can all affect gas exchange efficiency and arterial PO2 .
(Fig. 18.3)
5. The amount of a gas that dissolves in a liquid is proportional to the
partial pressure of the gas and to the solubility of the gas in the liquid. Carbon dioxide is 20 times more soluble in aqueous solutions
than oxygen is. (Fig. 18.4)
Gas Transport in the Blood
Respiratory: Gas Transport
6. Gas transport demonstrates mass flow and mass balance. The Fick
equation relates blood oxygen content, cardiac output, and tissue
oxygen consumption. (Fig. 18.6)
7. Oxygen is transported dissolved in plasma (62%) and bound to hemoglobin (798%). (Fig. 18.5)
8. The PO2 of plasma determines how much oxygen binds to hemoglobin. (Fig. 18.8)
9. Oxygen-hemoglobin binding is influenced by pH, temperature, and
2,3-diphosphoglycerate (2,3-DPG). (Fig. 18.9)
10. Venous blood carries 7% of its carbon dioxide dissolved in plasma,
23% as carbaminohemoglobin, and 70% as bicarbonate ion in the
plasma. (Fig. 18.11)
11. Carbonic anhydrase in red blood cells converts CO2 to carbonic acid,
which dissociates into H + and HCO3- . The H + then binds to hemoglobin, and HCO3- enters the plasma using the chloride shift.
Regulation of Ventilation
Respiratory: Control of Respiration
12. Respiratory control resides in networks of neurons in the medulla
and pons, influenced by input from central and peripheral sensory
receptors and higher brain centers. (Fig. 18.13)
13. The medullary dorsal respiratory group (DRG) contains mostly
inspiratory neurons that control somatic motor neurons to the
diaphragm. The ventral respiratory group (VRG) includes the preBötzinger complex with its apparent pacemakers as well as neurons
for inspiration and active expiration. (Fig. 18.14)
14. Peripheral chemoreceptors in the carotid and aortic bodies monitor PO2, PCO2, and pH. PO2 below 60 mm Hg triggers an increase in
ventilation.(Fig. 18.17)
15. Carbon dioxide is the primary stimulus for changes in ventilation.
Chemoreceptors in the medulla and carotid bodies respond to
changes in PCO2.(Fig. 18.17)
16. Protective reflexes monitored by peripheral receptors prevent injury
to the lungs from inhaled irritants.
17. Conscious and unconscious thought processes can affect respiratory
activity.
18
Questions
Level One Reviewing Facts and Terms
1. List five factors that influence the diffusion of gases between alveolus and blood.
2. More than
% of the oxygen in arterial blood is transported bound to hemoglobin. How is the remaining oxygen transported to the cells?
3. Name four factors that influence the amount of oxygen that binds to
hemoglobin. Which of these four factors is the most important?
4. Describe the structure of a hemoglobin molecule. What chemical
element is essential for hemoglobin synthesis?
5. The networks for control of ventilation are found in the
and
of the brain. What do the dorsal and ventral respiratory groups of neurons control? What is a central pattern generator?
6. Describe the chemoreceptors that influence ventilation. What
chemical is the most important controller of ventilation?
7. Describe the protective reflexes of the respiratory system.
8. What causes the exchange of oxygen and carbon dioxide between
alveoli and blood or between blood and cells?
9. List five possible physical changes that could result in less oxygen
reaching the arterial blood.
Level Two Reviewing Concepts
10. Concept map: Construct a map of gas transport using the following
terms. You may add other terms.
•
•
•
•
•
•
•
•
alveoli
arterial blood
carbaminohemoglobin
carbonic anhydrase
chloride shift
dissolved CO2
dissolved O2
hemoglobin
•
•
•
•
•
•
•
•
hemoglobin saturation
oxyhemoglobin
PCO2
plasma
PO2
pressure gradient
red blood cell
venous blood
11. In respiratory physiology, it is customary to talk of the PO2 of the
plasma. Why is this not the most accurate way to describe the oxygen content of blood?
12. Compare and contrast the following pairs of concepts:
(a) transport of O2 and CO2 in arterial blood
(b) partial pressure and concentration of a gas dissolved in a liquid
13. Does HbO2 binding increase, decrease, or not change with decreased pH?
14. Define hypoxia, COPD, and hypercapnia.
15. Why did oxygen-transporting molecules evolve in animals?
16. Draw and label the following graphs:
(a) the effect of ventilation on arterial PO2
(b) the effect of arterial PCO2 on ventilation
17. As the PO2 of plasma increases:
(a) what happens to the amount of oxygen that dissolves in plasma?
(b) what happens to the amount of oxygen that binds to hemoglobin?
657
Gas Exchange and Transport
Level Three Problem Solving
20. Marco tries to hide at the bottom of a swimming hole by breathing
in and out through two feet of garden hose, which greatly increases
his anatomic dead space. What happens to the following parameters
in his arterial blood, and why?
(a) PCO2
(c) bicarbonate ion
(b) PO2
(d) pH
21. Which person carries more oxygen in his blood?
(a) one with Hb of 15 g >dL and arterial PO2 of 80 mm Hg
(b) one with Hb of 12 g >dL and arterial PO2 of 100 mm Hg
22. What would happen to each of the following parameters in a person
suffering from pulmonary edema?
(a) arterial PO2
(b) arterial hemoglobin saturation
(c) alveolar ventilation
23. In early research on the control of rhythmic breathing, scientists
made the following observations. What hypotheses might the researchers have formulated from each observation?
(a) Observation. If the brain stem is severed below the medulla, all
respiratory movement ceases.
(b) Observation. If the brain stem is severed above the level of the
pons, ventilation is normal.
(c) Observation. If the medulla is completely separated from the
pons and higher brain centers, ventilation becomes irregular
but a pattern of inspiration/expiration remains.
24. A hospitalized patient with severe chronic obstructive lung disease
has a PCO2 of 55 mm Hg and a PO2 of 50 mm Hg. To elevate his blood
oxygen, he is given pure oxygen through a nasal tube. The patient
immediately stops breathing. Explain why this might occur.
25. You are a physiologist on a space flight to a distant planet. You find
intelligent humanoid creatures inhabiting the planet, and they willingly submit to your tests. Some of the data you have collected are
described below.
100
Pigment saturation, %
90
80
70
60
50
40
30
20
10
20
40
60
80
PO2 (mm Hg)
100
The graph above shows the oxygen saturation curve for the
oxygen-carrying molecule in the blood of the humanoid named Bzork.
658
Bzork’s normal alveolar PO2 is 85 mm Hg. His normal cell PO2 is
20 mm Hg, but it drops to 10 mm Hg with exercise.
(a) What is the percent saturation for Bzork’s oxygen-carrying
molecule in blood at the alveoli? In blood at an exercising cell?
(b) Based on the graph above, what conclusions can you draw
about Bzork’s oxygen requirements during normal activity and
during exercise?
26. The next experiment on Bzork involves his ventilatory response to
different conditions. The data from that experiment are graphed below. Interpret the results of experiments A and C.
PO2 = 50 mm Hg
PO = 85 mm Hg
2
Alveolar ventilation
18. If a person is anemic and has a lower-than-normal level of hemoglobin in her red blood cells, what is her arterial PO2 compared to normal?
19. Create reflex pathways (stimulus, receptor, afferent path, and so on) for
the chemical control of ventilation, starting with the following stimuli:
(a) Increased arterial PCO2
(b) Arterial PO2 = 55 mm Hg
Be as specific as possible regarding anatomical locations. Where
known, include neurotransmitters and their receptors.
A
B
PO2 = 85 mm Hg
C
Subject drank
seven beers
Plasma PCO2
27. The alveolar epithelium is an absorptive epithelium and is able to
transport ions from the fluid lining of alveoli into the interstitial
space, creating an osmotic gradient for water to follow. Draw an
alveolar epithelium and label apical and basolateral surfaces, the
airspace, and interstitial fluid. Arrange the following proteins on
the cell membrane so that the epithelium absorbs sodium and water: aquaporins, Na + -K + -ATPase, epithelial Na + channel (ENaC).
(Remember: Na + concentrations are higher in the ECF than in the
ICF.)
Level Four Quantitative Problems
28. You are given the following information on a patient.
Blood volume = 5.2 liters
Hematocrit = 47%
Hemoglobin concentration = 12 g>dL whole blood
Total amount of oxygen carried in blood = 1015 mL
Arterial plasma PO2 = 100 mm Hg
You know that when plasma PO2 is 100 mm Hg, plasma contains
0.3 mL O2 >dL, and that hemoglobin is 98% saturated. Each hemoglobin molecule can bind to a maximum of four molecules of
oxygen. Using this information, calculate the maximum oxygencarrying capacity of hemoglobin (100% saturated). Units will be
mL O2/g Hb.
29. Adolph Fick, the nineteenth-century physiologist who derived
Fick’s law of diffusion, also developed the Fick equation that relates
oxygen consumption, cardiac output, and blood oxygen content:
O2 consumption = cardiac output : (arterial oxygen content
- venous oxgen content)
A person has a cardiac output of 4.5 L >min, an arterial oxygen content of 105 mL O2 >L blood, and a vena cava oxygen content of 50
mL O2 >L blood. What is this person’s oxygen consumption?
30. Describe what happens to the oxygen-hemoglobin saturation curve
in Figure 18.9a when blood hemoglobin falls from 15 g >dL blood to
10 g>dL blood.
Gas Exchange and Transport
Answers
Answers to Concept Check Questions
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
(a) electron transport system (b) citric acid cycle
The PO2 of the alveoli is constantly being replenished by fresh air.
720 mm Hg * 0.78 N2 = 561.6 mm Hg
Air is 21% oxygen. Therefore, for dry air on Everest, PO2 = 0.21 *
250 mm Hg = 53 mm Hg. Correction for PH2O : PO2 = (250 mm Hg 47 mm Hg) * 21 = (203 mm Hg) * 0.21 = 43 mm Hg.
Blood pools in the lungs because the left heart is unable to pump all
the blood coming into it from the lungs. Increased blood volume
in the lungs increases pulmonary blood pressure.
When alveolar ventilation increases, arterial PO2 increases because
more fresh air enters the alveoli. Arterial PCO2 decreases because
the low PCO2 of fresh air dilutes alveolar PCO2. The CO2 pressure
gradient between venous blood and the alveoli increases, causing
more CO2 to leave the blood. Venous PO2 and PCO2 do not change
because these pressures are determined by oxygen consumption
and CO2 production in the cells.
False. Plasma is essentially water, and Figure 18.4 shows that CO2
is more soluble in water than O2.
The other factor that affects how much of each gas dissolves in the
saline solution is the solubility of the gas in that solution.
Yes. Hemoglobin reaches 100% saturation at 650 mm Hg. At sea
level, atmospheric pressure is 760 mm Hg, and if the “atmosphere”
is 100% oxygen, then PO2 is 760 mm Hg.
The flatness at the top of the PO2 curve tells you that hyperventilation causes only a minimal increase in percent saturation of
arterial Hb.
11. As the PO2 falls, more oxygen is released. The PO2 of venous blood
leaving the muscle is 25 mm Hg, same as the PO2 of the muscle.
12. An airway obstruction would decrease alveolar ventilation and increase arterial PCO2. Elevated arterial PCO2 would increase the H +
concentration in the arterial blood and decrease pH.
Answers to Figure and Graph Questions
Figure 18.4: Oxygen is 2.85 mL >L blood and CO2 is 28 mL>L blood.
Figure 18.5: O2 crosses five cell membranes: two of the alveolar cell,
two of the capillary endothelium, and one of the red blood cell.
Figure 18.9: 1. (a) When PO2 is 20 mm Hg, Hb saturation is 34%.
(b) Hemoglobin is 50% saturated with oxygen at a PO2 of 28 mm
Hg. 2. When pH falls from 7.4 to 7.2, Hb saturation decreases by
13%, from about 37% saturation to 24%. 3. When an exercising
muscle cell warms up, Hb releases more oxygen. 4. Loss of 2,3-DPG
is not good because then hemoglobin binds more tightly to oxygen at the PO2 values found in cells. 5. The PO2 of placental blood is
about 28 mm Hg. 6. At a PO2 of 10 mm Hg, maternal blood is only
about 8% saturated with oxygen.
Figure 18.13: 1. pons; 2. ventral respiratory group; 3. medullary chemoreceptor; 4. sensory neuron; 5. carotid chemoreceptor; 6. somatic
motor neuron (expiration); 7. aortic chemoreceptor; 8. internal
intercostals; 9. abdominal muscles; 10. diaphragm; 11. external intercostals; 12. scalenes and sternocleidomastoids; 13. somatic motor
neuron (inspiration); 14. dorsal respiratory group; 15. limbic system; 16. higher brain centers (emotions and voluntary control)
Figure 18.15: One breath takes 5 seconds, so there are 12 breaths>min.
18
Answers to Review Questions
Level One Reviewing Facts and Terms
1. Pressure gradients, solubility in water, alveolar capillary perfusion, blood
pH, temperature.
2. 98%. Remainder is dissolved in plasma.
3. PO2, temperature, pH, and the amount of hemoglobin available for binding
(most important).
4. Four globular protein chains, each wrapped around a central heme group
with iron.
5. Medulla and pons. Dorsal—neurons for inspiration; ventral—neurons for inspiration and active expiration. Central pattern generator—group of neurons
that interact spontaneously to control rhythmic contraction of certain muscle
groups.
6. Medullary chemoreceptors increase ventilation when PCO2 increases. Carotid
and aortic chemoreceptors respond to PCO2, pH, and PO2 6 60 mm Hg. PCO2
is most important.
7. They include irritant-mediated bronchoconstriction and the cough reflex.
8. Partial pressure gradients
9. Decreased atmospheric PO2, decreased alveolar perfusion, loss of hemoglobin, increased thickness of respiratory membrane, decreased respiratory
surface area, increased diffusion distance.
Level Two Reviewing Concepts
10. Start with Figure 18.10.
11. Most oxygen is bound to hemoglobin, not dissolved in the plasma.
12. (a) Most O2 is transported bound to hemoglobin, but most CO2 is converted
to bicarbonate. (b) Concentration is amount of gas per volume of solution,
measured in units such as moles per liter. While solution partial pressure and
concentration are proportional, concentration is affected by the gas solubility, and therefore is not the same as partial pressure.
13. decrease
14. Hypoxia—low oxygen inside cells. COPD—chronic obstructive pulmonary
disease (includes chronic bronchitis and emphysema). Hypercapnia—
elevated CO2.
15. Oxygen is not very soluble in water, and the metabolic requirement for
oxygen in most multicellular animals would not be met without an oxygentransport molecule.
16. (a) x-axis—ventilation in L>min; y-axis—arterial PO2, in mm Hg. See Figure
18.9. (b) x-axis—arterial PCO2 in mm Hg; y-axis—ventilation in L>min. As
arterial PCO2 increases, ventilation increases. There is a maximum ventilation
rate, and the slope of the curve decreases as it approaches this maximum.
17. (a) increases (b) increases
659
Gas Exchange and Transport
18. Normal, because PO2 depends on the PO2 of the alveoli, not on how much Hb
is available for oxygen transport.
19. (a) See Figure 18.17. (b) See Figure 18.13.
Level Three Problem Solving
20. Increased dead space decreases alveolar ventilation. (a) increases (b) decreases (c) increases (d) decreases
21. Person (a) has slightly reduced dissolved O2 but at PO2 = 80, Hb saturation
is still about 95%. If oxygen content is 197 mL O2>L at PO2 = 100 and 98%
saturation, then oxygen content at PO2 = 80 mm Hg and 95% saturation
is 190 mL O2>L blood 1197 * 10.95/0.9822, with Hb constant. Person (b)
has reduced hemoglobin of 12 g>dL, but it is still 98% saturated. So, oxygen
content would be 157.6 mL O2>L blood 1197 * 112/1522. The increased PO2
did not compensate for the decreased hemoglobin content.
22. (a) decrease (b) decrease (c) decrease
23. (a) Respiratory movements originate above the level of the cut, which could
include any area of the brain. (b) Ventilation depends upon signals from the
medulla and/or pons. (c) Respiratory rhythm is controlled by the medulla
alone, but other important aspects of respiration depend upon signals originating in the pons or higher.
24. With chronic elevated PCO2, the chemoreceptor response adapts, and CO2 is no
longer a chemical drive for ventilation. The primary chemical signal for ventila-
tion becomes low oxygen (below 60 mm Hg). Thus, when the patient is given
O2, there is no chemical drive for ventilation, and the patient stops breathing.
25. (a) Alveoli—96%; exercising cell—23% (b) At rest Bzork only uses about
20% of the oxygen that his hemoglobin can carry. With exercise, his oxygen
consumption increases, and his hemoglobin releases more than 3>4 of the
oxygen it can carry.
26. All three lines show that as PCO2 increases, ventilation increases. Line A shows
that a decrease in PCO2 potentiates this increase in ventilation (when compared
to line B). Line C shows that ingestion of alcohol lessens the effect of increasing
PCO2 on ventilation. Because alcohol is a CNS-depressant, we can hypothesize
that the pathway that links increased PCO2 and increased ventilation is integrated in the CNS.
27. Apical—faces airspace; basolateral—faces interstitial fluid. Apical side has
ENaC and aquaporins; basolateral side has aquaporins and Na+-K+-ATPase.
Na+ enters the cell through ENaC, then is pumped out the basolateral side.
(Cl- follows to maintain electrical neutrality.) Translocation of NaCl allows
water to follow by osmosis.
Level Four Quantitative Problems
28. 1.65 mL O2>gm Hb
29. 247.5 mL O2>min
30. Nothing. The percent saturation of Hb is unchanged at any given PO2. However, with less Hb available, less oxygen will be transported.
Photo Credits
CO: Jorge Bernardino de la Serna, MEMPHYS-Center for Biomembrane
Physics, University of Southern Denmark
660
Biotechnology: Photomick/iStockphoto.com.