* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 13: Properties of Solutions
Chemical equilibrium wikipedia , lookup
Franck–Condon principle wikipedia , lookup
Acid dissociation constant wikipedia , lookup
Properties of water wikipedia , lookup
Diamond anvil cell wikipedia , lookup
History of electrochemistry wikipedia , lookup
Self-assembled monolayer wikipedia , lookup
Atomic theory wikipedia , lookup
Water pollution wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Reverse osmosis wikipedia , lookup
Depletion force wikipedia , lookup
Liquid–liquid extraction wikipedia , lookup
Equilibrium chemistry wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
Vapor–liquid equilibrium wikipedia , lookup
Solvent models wikipedia , lookup
Size-exclusion chromatography wikipedia , lookup
Electrolysis of water wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Sol–gel process wikipedia , lookup
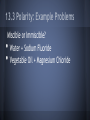
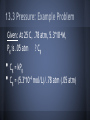
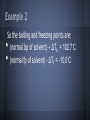
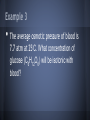
Chapter 13: Properties of Solutions Created By: Nick Brazones, Brian Fike, Maya Hairston, Brian Kuttler, Alex Reardon, Chase Schilling, Moses Suh 13.1 The Solution Process • • A solution is formed when one substance disperses uniformly throughout another. With the exception of gas mixtures, all solutions involve substances in a condensed state. 13.1 Intermolecular Forces • • Molecules or ions of substances in a liquid or solid state experience intermolecular attractive forces. A major factor determining whether a solution forms are the strengths of intermolecular forces between and among the solute and solvent particles. 13.1 Dissolution of an Ionic Solid in Water • NaCl dissolves readily in water because the attractive interactions between the ions and the polar H2O molecules overcome the lattice energy of NaCl. 13.1 Dissolution of an Ionic Solid in Water • • • Water molecules orient themselves to the surface of the NaCl crystals. The positive end of the water dipole is oriented toward the Cl- ions, and the negative end of the water dipole is oriented toward the Na+ ions. The ion-dipole attraction pulls the ions from their positions. 13.1 Dissolution of an Ionic Solid in Water • • • Once separated from the crystal,the Na+ and Cl- ions are surrounded by water molecules. Interactions like this, between solute and solvent molecules, are known as solvation. When the solvent is water, the interactions are also referred to as hydration. catalog.flatworldknowledge.com 13.1 Energy Changes and Solution Formation The overall enthalpy change in forming a solution, ΔHsoln, is the sum of three terms: ΔHsoln = ΔH1 + ΔH2 + ΔH3 ΔH1: Separation of solute molecules (endothermic) ΔH2: Separation of solvent molecules (endothermic) ΔH3: Formation of solute-solvent interactions (exothermic) Remember: breaking bonds takes energy http://wps.prenhall.com/wps/media/objects/3312/3391718/blb1301.html Enthalpy Changes http://wps.prenhall.com/wps/media/objects/3312/3391718/blb1301.html 13.1 Energy Changes and Solution Formation • Ionic solutes like NaCl do not dissolve in non-polar solvents such as Gasoline, because the non-polar hydrocarbon molecules would experience only weak attractive interactions with the ions. 13.1 Energy Changes and Solution Formation • Polar liquids, such as water, do not form solutions with non-polar liquids like gasoline, because the water molecules experience strong hydrogen-bonding interactions. Solution Formation, Spontaneity, and Disorder If you let go of a book, it falls to the floor because of gravity. At its initial height, it has a higher potential energy than when it is on the floor. Unless it is restrained, the book falls; and as it does, potential energy is converted into kinetic energy. When the book strikes the floor, the kinetic energy is converted largely into heat energy, which is dispersed throughout the surroundings. The book has lost energy to its surroundings in the process. This fact leads us to the first basic principle identifying spontaneous processes and the direction they take. Solution Formation, Spontaneity, and Disorder First Basic Principle: • Processes in which the energy content of the system decreases tend to occur spontaneously. Solution Formation, Spontaneity, and Disorder Suppose we could suddenly remove a barrier that separates 500 mL of CCl4 from 500 mL of C6H14. Before the barrier is removed, each liquid occupies a volume of 500 mL. When equilibrium has been established after the barrier has been removed, the two liquids together occupy a volume of 1000 mL. Formation of a homogenous solution has increased the degree of dispersal, or randomness (entropy), because the molecules of each substance are now mixed and distributed in a volume twice as large as that which they individually occupied before mixing. Solution Formation, Spontaneity, and Disorder Second Basic Principle: • Processes occurring at a constant temperature in which the randomness or dispersal in space (entropy) of the system increases tend to occur spontaneously. Solution Formation, Spontaneity, and Disorder Third Basic Principle: • In most cases, formation of solutions is favored by the increase in entropy that accompanies mixing. Solution Formation, Spontaneity, and Disorder • Consequently, a solution will form unless solute-solute or solvent-solvent interactions are too strong relative to the solute-solvent interaction. Solution Formation and Chemical Reactions • • Be careful to distinguish the physical process of solution formation from chemical reactions that lead to a solution. For example, Ni(s) + 2 HCl(aq) → NiCl2(aq) + H2(g) Solution Formation and Chemical Reactions Ni(s) + 2 HCl(aq) → NiCl2(aq) + H2(g) The chemical form of the substance being dissolved is changed from Ni to NiCl2; if the solution were evaporated, NiCl2, not Ni would be recovered 13.2 Saturated Solutions and Solubility • • • As a solid solute dissolves, the concentration of the solute in the solution will increase. If the concentration of the solute gets high enough, the particles may collide back together. This process is known as crystallization. catalog.flatworldknowledge.com 13.2 Saturated Solutions and Solubility • • • www.prevor.com Solute + Solvent Solution The forward reaction is when the solvent is dissolving. The reverse reaction is when the solute is crystallizing back together. 13.3 Factors Affecting Solubility http://www.engineeringarchives.com/les_matsci_unlimitedandlimitedsolubility.html 13.3 Polarity: Like Dissolves Like • • Non-Polar substances tend to dissolve in non-polar liquids. Polar and ionic substances tend to dissolve in polar liquids. 13.3 Polarity: Like Dissolves Like • • Miscible: Pairs of liquids that dissolve completely in each other Ex: Water + Acetone Immiscible: Paris of liquids that do not dissolve significantly in each other Ex: Water + Oil http://library.thinkquest.org/CR0215471/oil_spills.htm • • 13.3 Polarity: Why Water is the Universal Solvent Water is polar which allows it to dissolve polar and ionic solutions, which are the majority of solutions. Water has the ability to make hydrogen bonds. http://en.wikipedia.org/wiki/Hydrogen_bond 13.3 Polarity: Example Problems Miscible or Immiscible? Water + Sodium Fluoride Vegetable Oil + Magnesium Chloride • • 13.3 Pressure: Trends http://unit4lewiswain.blogspot.com/ • • The solubility of a gas increases as pressure of the gas increases. Does not affect the solubility of solid or solutions 13.3 Pressure: Henry’s Law • • • • The relationship between pressure and solubility of a gas is expressed as: Cg = kPg Cg is the solubility of the gas (molarity). k is a constant that changes with the units of volume and temperature (mol/ L*atm). Pg is the partial pressure of the gas over the solution. 13.3 Pressure: Example Problem Given: At 25 C, .78 atm, 5.3*10-4M, Pg is .05 atm ? Cg • • Cg = kPg Cg = (5.3*10-4 mol/L)/.78 atm (.05 atm) Pressure: Example Problem Answer 3.4 x 10-5 M 13.3 Temperature: Trends • • As temperature increases, the solubility of most solids in water increases. Exceptions do apply* Ce2(SO4)3 As temperature increases, the solubility of gases in water decrease. Ex: Boiling water releases gas molecules 13.3 Temperature: Solubility Chart http://www.kentchemistry.com/links/Kinetics/SolubilityCurves.htm http://www.barrymitzman.com/barry_mitzman/2012/01/mms-go-to-the-super-bowl.html 13.4 Ways of Expressing Concentration Maya and Moses 13.4 Mass Percentage ● “soln” represents solution 13.4 Mass Percentage Problem A solution is made by dissolving 13.5 g of glucose (C6H12O6) in .100 kg of water. What is the mass percentage of solute in this solution? 13.4 Part Per Million (ppm) and Part Per Billion (ppb) • • • • • Diluted 1 ppm =106 grams = 1 milligram (mg) Parts per billion (ppb) 1 ppb =109 grams = 1 microgram (µg) Extremely diluted 13.4 Ppm And Ppb Problems A 2.5 g sample of groundwater was found to contain 5.4 µg of Zn2+. What is the concentration of Zn2+ in parts per million? What is the concentration of Zn2+ in parts per billion? 13.4 Mole Fraction ● Symbol X is commonly used for mole fraction, with a subscript to indicate the component of interest. ● Example: XNACL ● Mole fraction of Sodium chloride (Table Salt) 13.4 Molarity and. Molality ● Molarity (M): ● Molarity changes with temperature because the expansion or contraction of the solution changes its volume. • • Molality (m): Molality does not vary with temperature because mass does not vary with temperature. Thus, molality is often the concentration unit of choice when a solution is to be used over a range of temperatures. 13.4 Molarity Problem A solution contains 5.0 g of toluene (C7H8) and 225 g of benzene and has a density of 0.876 g/mL. Calculate the molarity of the solution. 13.4 Mole Fraction and Molality Problem An aqueous solution of hydrochloric acid contains 36% HCL by mass. Calculate the mole fraction of HCL in the solution. Calculate the molality of HCL in the solution. 13.4 http://wps.prenhall.com/wps/media/objects/3312/3391718/imag1304/AAAVTAQ0.JPG 13.5 Colligative Properties • Physical properties of solutions that depend on the quantity but not the kind of solution particles Vapor pressure o Boiling point o Freezing point o Osmosis o 13.5 Lowering the Vapor Pressure • • • The pressure exerted by the vapor o A substance with no measurable vapor pressure is nonvolatile whereas one that has a vapor pressure is considered volatile. Adding a nonvolatile solute to a solvent always lowers the vapor pressure o Expressed by Raoult’s Law An ideal solution obeys Raoult’s Law http://wps.prenhall.com/wps/media/objects/3312/3391718/blb1305.html 13.5 Raoult’s Law • PA=XAP◦A o PA -Partial pressure exerted by solvent vapor o XA -product of the mole fraction of the solvent in the solution o P◦A -vapor pressure of the pure solvent Example 1 • Calculate the vapor pressure for a solution prepared by dissolving 185.0g sucrose (mol. wt.=342.3) in 643.5 cm3 of water. the density of water is 0.9971g/cm3 and the vapor pressure is 23.76 torr. Example 1 First determine the number of moles of sucrose: Example 1 Next determine the moles of water: Example 1 The mole fraction of water in the solution is: Example 1 Then: The vapor pressure has been lowered by 0.30 torr. 13.5 Boiling-Point Elevation • • The boiling point of a solution is higher than that of a pure liquid. ΔTb=Kbm o ΔTb- boiling point of pure solvent o Kb- molal boiling-point-elevation constant o m- molality 13.5 Freezing-Point Depression • • The freezing point of the solution is lower than that of the pure liquid. ΔTf=Kfm o ΔTf - freezing point of pure solvent o Kf- molal freezing-point-depression constant o m- molality Example 2 • Calculate the boiling point and freezing point of a 25.0 mass % solution ethylene glycol (C2H6O2) in water. Example 2 First calculate the molality of the solution: Example 2 Calculate the changes in freezing and boiling point: Example 2 So the boiling and freezing points are: (normal bp of solvent) + ΔTb = 102.7◦C • • (normal fp of solvent) - ΔTf = -10.0◦C 13.5 Osmosis • • Some materials are semipermeable; they allow some molecules to pass through but not others. Osmosis- the net movement of solvent molecules from the less concentrated solution to the more concentrated solution 13.5 Osmotic Pressure • • Osmotic pressure (π) is the pressure required to prevent osmosis Follows a law similar to the ideal-gas law o o πV=nRT π=MRT where M is the molarity of the solution 13.5 Osmotic Pressure • • If two solutions have the same osmotic pressure, no osmosis will occur and the solutions are isotonic. If one solution has a lower osmotic pressure, it is hypotonic with respect to the more concentrate, or hypertonic, solution. Example 3 • The average osmotic pressure of blood is 7.7 atm at 25◦C. What concentration of glucose (C6H12O6) will be isotonic with blood? Example 3 • • • π=MRT M=π/RT 7.7atm/(0.0821)(298 K) = 0.31 M 13.5 Determining Molar Mass • • The colligative properties of solutions can be used to determine molar mass. Any of the four colligative properties can be used. 13.6 Colloids • • • Colloids are the intermediate types of dispersions between solutions and heterogenous mixtures. The size of a colloid is one that is larger than a molecule, but not large enough for the components to be separated by gravity. They can be found in any of the three natural states of matter. Examples of Colloids • • Some examples of colloids are fog, smoke, whipped cream, milk, paint, marshmallow, butter, lotion, and ruby glass. Milk is just a mixture of fat droplets suspended in water. http://www.absoluteastronomy.com/topics/Colloid 13.6 Tyndall Effect • • The diameter of a colloid particle can range between 5nm to 1000 nm. Despite their size, they are still large enough to scatter light. http://silver-lightning.com/tyndall/ 13.6 Hydrophilic vs. Hydrophobic • • The most important colloids are ones where the dispersing medium is water. The colloids are either hydrophilic or hydrophobic. 13.6 Hydrophilic • • • A hydrophilic colloid is similar to large molecules in the human body. The hydrophobic groups are in the inside of the molecule, while the hydrophilic groups are on the outside of the molecule, keeping it suspended in water. They usually contain either oxygen or nitrogen and carry a charge. 13.6 Hydrophobic • • A hydrophobic colloid has to be stabalized, because if not, the molecules will separate in the water. They can be stabilized by adsorption of ions on their surface, or the adhesion of the ions to its surface. 13.6 Hydrophobic Cont. • • The ions on the outside of the molecule are hydrophilic, so they interact with the water and keep it suspended. The repulsion between the ions and the colloid particles keep the molecules from colliding and getting larger. http://www.life-treasure.com/technology.html Example of Suspension http://www.zazzle.com/chemists_are_frequently_in_suspension_humor_card-137220125589000317 Other Cases • • Hydrophobic colloids can also be stabilized with hydrophilic groups on their surfaces. For example: o Oil is a hydrophobic substance, so when in water, it forms an oil slick. o But in the presence of sodium stearate, a substance with one hydrophilic end and one hydrophobic end, the oil is stabilized. o The hydrophobic end of the stearate interacts with the oil, while the hydrophilic end interacts with the water, suspending the molecule in water. 13.6 Emulsion • Emulsion is when a suspension of one liquid is formed in another liquid. o Example: process that occurs in the small intestine o In digestion, bile is released, and one specific component in it emulsifies the fats, which helps the absorption of them through the intestine wall. o The component in bile is an emulsifying agent. 13.6 Removal of Colloids • • • Sometimes, colloids must be removed from the dispersing medium. They are too small to be removed by filtration, so they must be removed by coagulation. After coagulation, the colloids will be large enough to filter, or they can settle out of the dispersing medium. 13.6 Coagulation • • Coagulation is when the colloids collide to create larger molecules. It can occur by either heating the mixture or adding electrolytes 13.6 Heating • • When heated, the speed of the particles increase, allowing them to collide. After they collide, the particles stick together and grow larger. 13.6 Adding Electrolytes • • • When an electrolyte is added, it removes the charge on the surface of the particles. The neutral charge eliminates the electrostatic repulsion between the particles. With no repulsion, they are allowed to collide and grow larger. 13.6 Semi Permeable Membranes • Another way to separate ions from a colloid is by using semi permeable membranes. o • Ions can pass through the membrane, but the colloids cannot. This separation is otherwise known as dialysis, and is used to purify blood in the kidney.